
In this context, we briefly study atomic emission. Emission is something the discharge, release especially the gas or radiations. In analytical chemistry, we study the analysis of matter or the characterization of matter. Analysis of matter is in two forms one is atomic and the other is molecular form.
What is Atomic Emission?
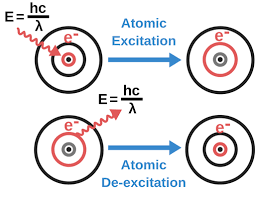
If our sample in which we analyze the analyte is in atoms or atomic form then this is called atomic spectroscopy. In atomic emission spectroscopy, the electrons present in our analyte first get excited by absorbing radiations from the source and then emit the same amount of radiation that is absorbed. This is called ”atomic emission”. And these emitted radiations are measured and this is called atomic emission spectroscopy.
In order to study atomic emission first, we have to take a look at spectroscopy, and Analyte.
What is Spectroscopy?
Spectroscopy is a tool to study the structure of atoms or molecules. In this, there is an interaction of matter with electromagnetic radiations. We get qualitative and quantitative information.
What is Analyte?
We characterize matter in the form of a sample. The sample contains two parts analyte and matrix. The constituents of the sample about which analytical information is required are called Analyte.
Whereas all other contents except that of the analyte are called matrix.
How many Basic Types of Spectroscopy?
There are two basic types of spectroscopy.
- Atomic spectroscopy
- Molecular spectroscopy
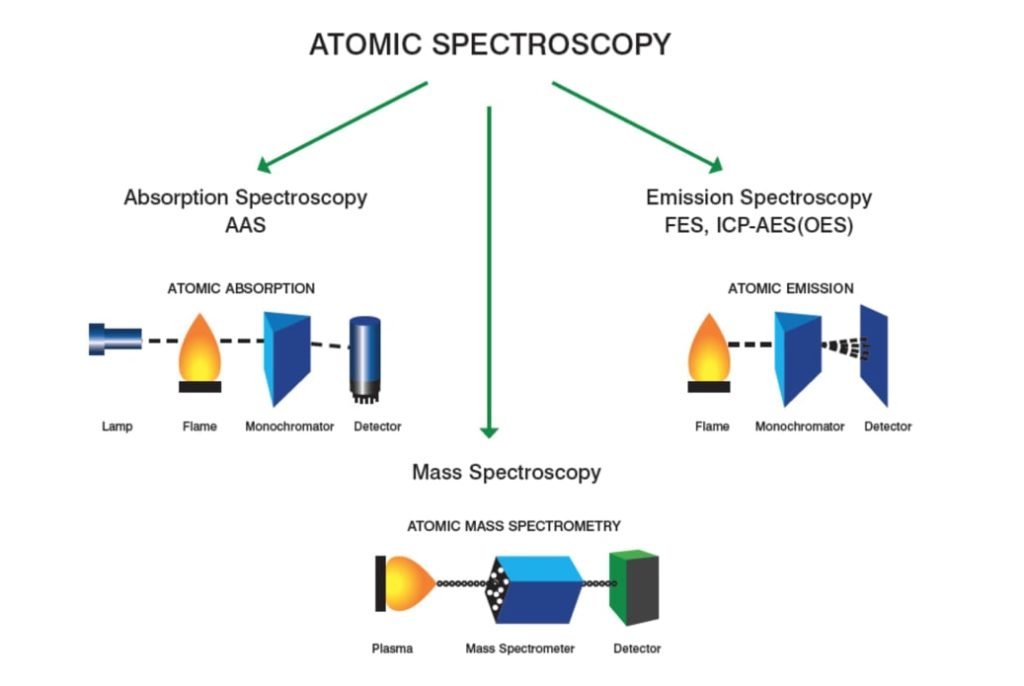
Atomic Spectroscopy:
In this type of spectroscopy, the sample is analyzed in the form of atoms. There are two types of atomic spectroscopy.
- Atomic absorption spectroscopy
- Atomic emission spectroscopy
Atomic emission Spectroscopy:
In atomic emission spectroscopy, the sample is in the form of atoms. The sample first gets excited by absorbing radiations and after deexcitation the sample emits radiations.
The amount of emitted radiations is measured and this is called atomic emission spectroscopy.
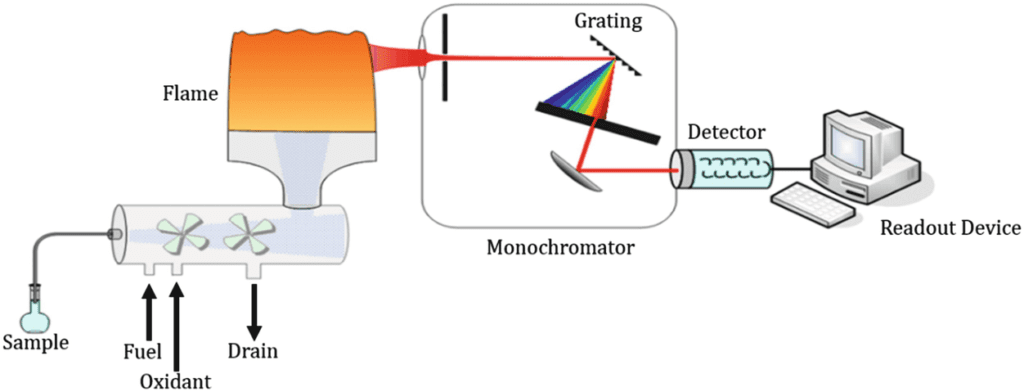
This is a flow sheet diagram of atomic emission spectroscopy.
Source of Atomization:
This is a diagram of flame emission spectroscopy. The sample is mixed with fuel and oxidants. Through drain extra sample is removed. Then sample enters to flame section. Here the sample is converted into atomic form. The sample absorbs the energy of flame by absorbing it and becomes excited. When sampling de-excite it emits particular radiations of light.
Diffraction grating:
And we observe in the diagram that there is a light of emission and another light of the flame. To differentiate between these two lights. These are passed through a diffraction grating with the help of lenses.
Monochromator:
From the grating light pass to the monochromator. The monochromator is a device that is used to scan wavelengths of light in order to select a particular wavelength of light.
The function of a monochromator is to scan the wavelengths of light and only pass out a particular wavelength.
The monochromator is used to enhance the detector response. As we know that generally, we use detectors that are not much sensitive to detect the minor absorption of light or emission of light.
For example, if 100% light is passing from the source and the sample absorbs only 1% light. So there is a chance that the detector does not observe this minor absorption. And by using a monochromator if 10% light is passing. So in this case, if 1% is absorbed then the detector easily detects this minor change.
In flame emission spectroscopy, in place of a monochromator special types of filters are used to select the wavelength of light. The monochromators or filters stop all light and only pass out the emitted light.
Detector:
And the emitted light from the monochromator or filters reaches the detector. The detector gives a response in the form of signals to the readout of the device.
Readout Device:
The detector gives signals to the readout device. The readout device gives information in the form of graphs. The quantity of light is quantitative analysis. And how many wavelengths of light is qualitative analysis. This is all about atomic emission spectroscopy.
Atomic absorption spectroscopy:
The second type of spectroscopy is atomic absorption spectroscopy.
What is atomic absorption?
When the light in the form of radiation strikes an electron it gets excited and absorbs energy. The amount of energy absorbed is measured and this is absorption spectroscopy.
If our sample in which we analyze or characterize the analyte is in the form of atoms then this is called atomic absorption spectroscopy.
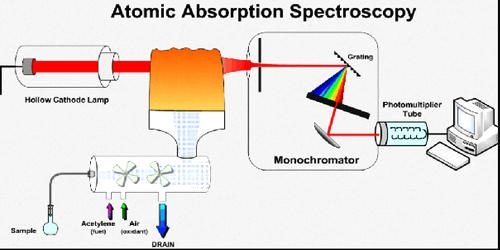
This is a flow sheet diagram of atomic absorption spectroscopy.
Source of atomization:
This is a diagram of flame absorption spectroscopy. This sample is analyzed in the form of atoms. But we cannot measure the emitted light. We will measure the absorbed light.
The light is given out by a hollow cathode lamp. The sample is sucked by a capillary tube. Oxidizing gases are used in burning flame. Fuel gas is also used for burning flame.
After mixing the chamber sample it goes to the slit burner. The shape of the slit burner is longitudinal. In a slit burner, all atoms become excited by absorbing light from the source.
What is the function of a hollow cathode lamp in Atomic emission or atomic absorption?
If we supply more light then there is no excitation of electrons. And if we supply less then there will be no excitation. How do we know the amount of energy? For this problem, we have to use a hollow cathode lamp. A hollow cathode lamp is made of such material that is of our analyte.
In a hollow cathode, lamp electrons are excited electrically. After de-excitation, the amount of emitted energy is measured which is exactly equal to the amount of energy needed for transitions.
Diffraction grating:
Next to the slit burner, there is diffraction grating which is used to differentiate between the light of source or light of emission.
Monochromator:
The monochromator is used to scan a particular wavelength of light. It sends signals to the detector.
Detector:
Detectors give responses or signals to the readout device and data is given out in the form of graphs.
So this is all about the basic principle of atomic absorption spectroscopy.
Which one is best; Atomic or molecular spectroscopy?
For best qualitative analysis atomic spectroscopy is best. But there is a problem that the sample is must be converted into atomic form and the sample cannot be reused for another type of spectroscopy.
On the other hand molecular spectroscopy, there is no use of the heating source. But molecular spectroscopy gives a band spectrum which is difficult to study.
In atomic spectroscopy, there is a line spectrum that is easily studied.



Leave a Reply