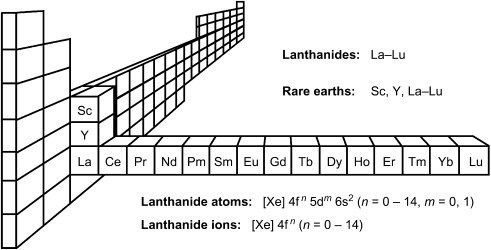
The lanthanides are the 14 elements series from cerium (Z=58) to lutetium (Z=71) in the periodic table. These elements are called lanthanides because these elements are placed after lanthanum in the 3rd group and 6th period of the periodic table. Lanthanides are present in the 6th Period of the periodic table so these elements have 6 shells in their electronic configuration. The last electrons in these elements enter the 4f orbital. All 15 elements from lanthanum to lutetium have similar properties.
The study of lanthanides consists of the study of all the 15 elements from lanthanum to lutetium together. Lanthanum is the prototype of Lanthanide. Lanthanides are also called lanthanones or rare earth metals. The name rare earth was given to them because these elements were originally extracted from the oxides for which ancient name was earth and which were considered to be rare.
The term rare earth is avoided now because many of these elements are no longer rare and now they are abundant. Lanthanides are also known as the first inner transition elements. In the lanthanide series, 4f orbital is in the process of filling. In the lanthanide series, there are two elements, one has zero electrons in 4f (lanthanum) and another has completely filled 4f subshell (lutetium) but we considered these two elements in the lanthanide series or 4f series because their properties are similar to lanthanides.
Why Lanthanides are Placed at the bottom of the Periodic Table?
The lanthanides are f-block elements located at the bottom of the periodic table. The lanthanide series is started from between barium (Z=56) and hafnium (Z=72).
Moseley (the scientist who discovered atomic numbers) proved that the properties of barium are very similar to calcium and strontium. It also proved that hafnium is similar properties to zirconium, that’s why barium must be placed below strontium and hafnium below zirconium. The properties of lanthanum are very similar to yttrium so we placed lanthanum just below the yttrium and a series of 14 elements is placed at the bottom of the periodic table. The series of 14 elements is started from cerium and ends at lutetium. Since all the 15 elements of the Lanthanides resembles one another in many respects so these must be placed in the same group.
What is the Occurrence of Lanthanide Elements?
The lanthanides are not scare in the earth’s crust as the name rare earth is implied. Potentially their sources are unlimited, each lanthanide’s minerals contain all lanthanides except promethium (very unstable not found in minerals) which is an unstable radioactive element.
Important Minerals of Lanthanides
Remember that the lanthanides having atomic numbers 57 to 63 is known as light lanthanides and the atomic numbers from 64 to 71 are called heavy lanthanides. There are three major minerals of lanthanides:
- Monazite
- Xenotime
- Euxenite
1. Monazite
It contains a mixture of light lanthanides (La57 to Eu63). It contains 50 to 70% light lanthanides.
- Thorium oxide, ThO = 3-10%
- Silicon oxide, SiO2 = 1-2%
- Phosphorous oxide, P2O5 = 22-30%
- Zirconium oxide, ZrO
- Titanium oxide, TiO2
- Traces of Uranium
2. Xenotime
It is a mixture of phosphates of heavier lanthanides (Gd64 to Lu71). It contains 54-65% heavy lanthanides.
- Thorium oxide, ThO2 = 3%
- Uranium oxide, U2O8 = 3.5%
- Zirconium oxide, ZrO2 = 2.3%
3. Euxenite
In this mineral, light and heavy lanthanides are in comparable quantity. It also has phosphates of lanthanides.
Oddo-Harkins Rule
Oddo-Harkins states that the elements (lanthanides) having even atomic number is more abundant as compared to the elements having odd atomic number. Even is 2 to 7 times more abundant than the odd atomic number lanthanides.
What are the Properties of the Lanthanides?
1. Oxidation State
All Lanthanide exhibits a common stable oxidation state of +3. In addition, some lanthanide shows + 2 and + 4 oxidation state also in solution or in solid compounds.
2. Atomic and Ionic radii
In the lanthanides, with increasing atomic number, the atomic and ionic radii decrease.
3. Color of Lanthanides
The lanthanides are silvery-white but many of the trivalent ions of lanthanides are colored in the solid-state as well as in solution the color of lanthanides arises due to the absorption in the visible region of the spectrum resulting in f-f transition because they have partially filled orbitals.
4. Magnetic Property
Ions that contain all paired electrons are diamagnetic while those containing unpaired electrons are paramagnetic in nature.
5. Ionization Enthalpies
When we move from left to right in the lanthanide series, the value of ionization energy is increased. The first ionization enthalpy of lanthanides is of the order of 600 KJ/mol and the second ionization energy is about 1200 KJ/mol.
6. Basic Character
All the lanthanides form hydroxides having formula Ln(OH)3. These hydroxides are ionic and basic in nature. They are stronger base than Al(OH)3 but weaker than Ca(OH)3.
7. Chemical Reactivity
All the lanthanides are highly electropositive metals and have almost similar chemical reactivity. The lanthanides are more reactive than d-block elements.
8. Chemical Properties
Some of the chemical properties of lanthanides are given below:
- All lanthanides are silvery-white salt metals that transit readily on exposure to air.
- The hardness of lanthanides increases with the increasing atomic number.
- They have a typical metallic structure and a good conductor of heat and electricity.
- The earlier members of the series are quite reactive similar to the calcium but with the increasing atomic number, they behave like aluminum.
- All these metals combine with hydrogen when heated while they liberate hydrogen from dilute acid.
- They are also combined with halogen and all lanthanides react with water also.
- They react with nitrogen to form nitrides of the formula LnN.
- When the metal is heated with carbon, they form carbides of the formula Ln3C, Ln2C3, and LnC3.
What are the Similarities Between Actinides and Lanthanides?
- In both series, the extra electron enters (n-2)f orbital.
- The element of both series shows a +3 oxidation state.
- The nitrates, perchlorates, sulfates of trivalent actinides as well as lanthanides are soluble but the hydroxides, fluorides, and carbonates are insoluble in water.
- In the absorption spectra of elements of both the series, sheep line-like bands appear due to f-f transition.
What are the Dissimilarities Between Actinides and Lanthanides?
| Lanthanides | Actinides |
|---|---|
| The binding energy of 4f is higher. | The binding energy of 5f is lower. |
| The 4f electrons have a greater shielding effect. | The 5f electrons have a lower shielding effect. |
| The maximum oxidation state exhibited by lanthanides is +4. | Due to lower binding energy, they show higher oxidation states such as +4, +5, +6. |
| The additional electrons enter the 4f orbital. | The additional electrons enter the 5f orbital. |
| They do not form oxocations. | They form oxcations such as UO22+, NPO2+, PuO2+. |
| Expect Pm, all the remaining lanthanides are non-radioactive. | All actinides are radiactive. |
| They do not form complexes easily. | They form complexes easily. |
| They are paramagnetic but magnetic properties can be easily explained. | They are also paramagnetic but it is difficult to explain magnetic properties. |
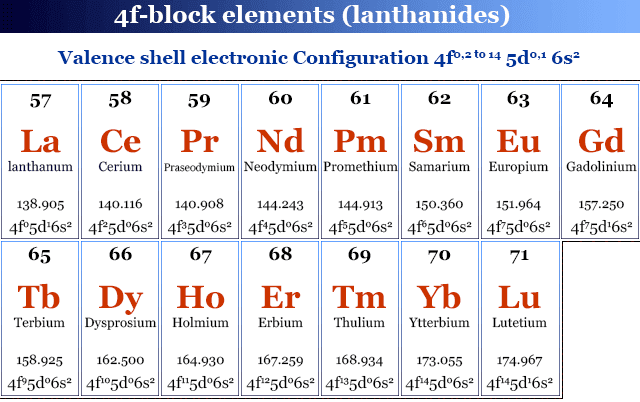
Electronic Configuration of Lanthanides



Leave a Reply