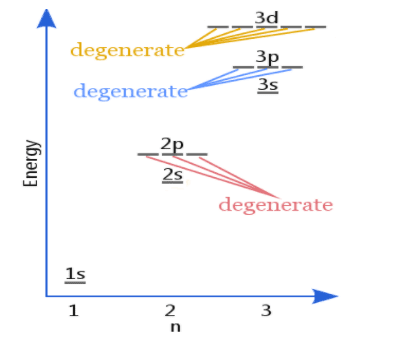
Orbitals that have equal energies are said to be degenerate orbitals. The number of orbitals having the same energy is called degeneracy. In the given content we study about. Orbitals, electronic configuration, ways to write electronic configuration, and related rules and principles are also disscused here. The rules to write electronic configuration help us to understand how to fill electrons in orbitals. Hund’s rule gives information about the stability of degenerate orbitals.
Orbital:
Orbital is a function describing the location and wave-like behavior of electrons in an atom. This function helps to find the probability of finding electrons around the nucleus in an atom.
Electronic configuration:

The distribution of electrons in shells and subshells and orbitals in the atom of an element is called electronic configuration.
Valence shell electronic configuration:
The distribution of electrons in various subshells and orbitals of the valence shell of an atom is called valence shell electronic configuration.
- The electronic configuration of an atom is written in terms of nlx notation .
- where l=0 (s-subshell),1(p-subshell),2(d-subshell)and 3(f-subshell).
- The super script x indicate number of electrons in subshell.
- ‘n’ is written left to ‘l’ indicate the number of shells.
Symmetrical electronic charge distribution:
The distribution in which electronic charge is distributed uniformly in orbitals. This is called uniform or symmetrical electronic charge distribution.
Unsymmetrical electronic charge distribution:
The distribution in which electronic charge is not uniformly distributed. This distribution is called unsymmetrical electronic charge distribution.
This distribution of electronic charges depends on;
- Shape of orbitals.
- The way in which electrons are distributed in various orbitals.

s-orbital:
s-orbital is spherical in shape.The electronic charge distribution in this orbital is uniform in all directions .whether orbital is singly filled or doubly filled.
p-orbital:
The distribution of electronic charge in an electronic configuration having three p-orbitals.Either half-filled (Px1,Py1 ,Pz1) or filled (Px ,Py2 ,Pz2 ) is symmetrical.The configuration p1 ,p2 ,p4 and p5 is unsymmetrical distribution.
d-orbital:

The distribution of charge in electronic configuration having five d-orbitals.Either half filled (d1xy,d1yz,d1xz,d1z2 and d1x2y2) or completely filled( d2xy,d2yz,d2xz,d2z2 and d2x2y2) is symmetrical. The configuration d2 ,d3 ,d4 ,d6 ,d7,d8 and d9 have unsymmetrical charge distribution.
Relation between electronic charge distribution and configuration and its stability:
It is observed that electronic arrangement having symmetrical distribution has lower energy and higher stability. As compared to an electronic arrangement having an unsymmetrical distribution of charge has less stability. So half-filled and filled orbitals having symmetrical distribution are more stable than others.
For example:
Three p-orbitals, the px orbital is symmetrical along the x-axis. And py and Pz are along the y and z-axis respectively. If we place one electron in px the electronic charge would be concentrated in the x-direction only. As the electronic charge would be concentrated in the y-direction in the case of Py1.In px and py the electronic charge is concentrated in the xy-plane mainly. A similar electronic charge would be concentrated more along x-axis for Px2 , Py1 , Pz1 . Thus in all p-configurations ( p1 ,p2 ,p4 and p5 ) charge is not distributed in all directions and hence all these configurations are less stable.
Rules for writing electronic configuration:
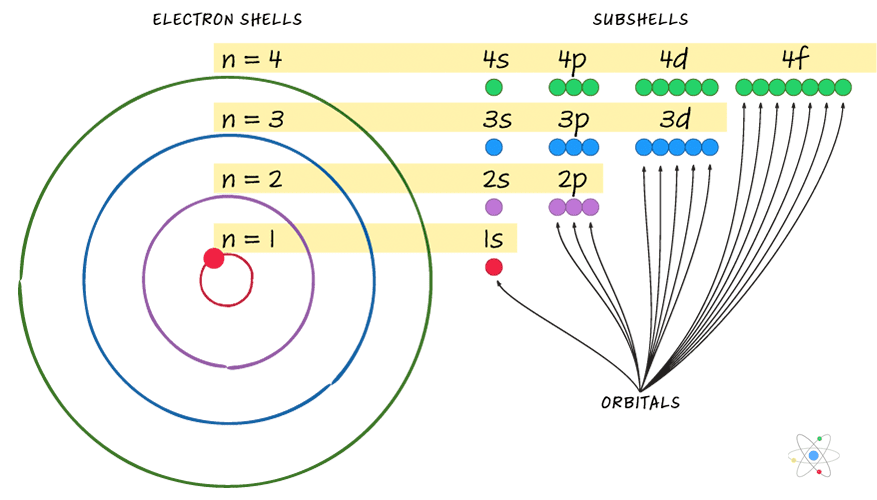
Rules for writing electronic configuration of an atom of an element are as follow:
1.Maximum number of electrons in a shell:
According to the Bohr-Bury scheme, the maximum number of electrons that a shell can hold is equal to 2n2 .
Where n=number of shell or principal quantum number.
Thus according to Bohr bury scheme number of electrons in shells is as;
- 1st: (n=1)
- 2nd : (n=2)
- 3rd : (n=3)
- 4th :(n=4)
shell is equal to
- 2×12 =2
- 2×22 =8
- 2×32 =18
- 2×42 =32
2.Maximum number of electrons in sub-shell:
The maximum number of electrons in a subshell is equal to;
2(2l +1)
where ‘l’ is azimuthal quantam number and l=0,1,2 and 3 for s,p,d and f sub-shells respectively.
Thus maximum number of electrons in sub shells;
s(l=0)
p(l=1)
d(l=2)
f(l=3)
putting these values in expression 2(2l+1) so
for s: 2(2×0+1)=2
for p:2(2×1+1)=6
for d:2(2×2+1)=10
for f: 2(2×3+1)=14
3.Aufbau Principle:
Aufbau is a German word that means building up or construction.
That’s why this principle is also known as the building up principle or construction principle.
According to this principle:
Orbitals are filled up with electrons in increasing order of their energies.
The relative order of various orbitals of an atom is given as;

Limitations of Aufbau Principle:
There were few elements in which extra electrons when entered in orbitals don’t follow Aufbau Principle.
- Lanthanum(At.no.57): According to Aufbau Principle electronic configuration of Barium (At.no.56)is (Xe)6s2 .Now after completion of 6s orbital at Ba.The next electron according to Aufbau Principle enter 4f-orbital in La atom to give (xe)4f1 5d0 6s2 electronic configuration .But it is observed that extra electron enters 5d orbital instead of 4f orbital to give (xe)4f1 5d0 6s2 at La atom.
- Actinium(At.no.89): According to Aufbau principle electronic configuration of Ra (z=88) atom is (Rn) 7s2 .Now after 7s-orbital has been completely filled at Ra.The next electron according to Aufbau Principle must enter 5f-orbital in Ac atom .But this electron enters the 6d-orbital instead of 5f-orbital in Ac atom.
4.(n+l) Rule:
The relative order of energy of different orbitals can be found with help of the (n+l) rule.
According to this rule:
The orbitals having the lowest value of (n+l) have the lowest energy and hence filled up first with electrons.
When two orbitals having the same value of (n+l). Then the orbital with the lower value of n is of lower energy and filled up first with electrons.
Using this rule sequence in which orbitals are filled can be found.
For example:
The value of (n+l) for 4s (4+0) is less than for 3d (3+2) .Hence 4s is filled first than 3d .
5. Pauli’s Exclusion Principle:

This principle states that:
The values of all four quantam numbers for two electrons residing in same orbital like s,px,py,pz,dxy,dyz,dxz,dx2 y2 ,dz2 etc.Cannot be same.
- For two electrons residing in same orbital the value of three quantam number is same but the fourth quantam number that is spin quantam number (ms ) for two electrons must be different.
- For one electron spin quantam number :ms will be +1/2 and for other it will be -1/2.
- Two electrons residing same orbital must have opposite spin(↑↓).
- Opposite spin gives less energy to system.And according to thermodynamics a state with less energy is more stable.
For example:
The two different arrangements for 2s orbital as :
| Arrangement A | ↑ n=2 l=0 ml=0 ms=+1/2 | ↓ n=2 l=0 ml=0 ms=-1/2 |
| Arrangement B | ↑ n=2 l=0 ml=0 ms=+1/2 | ↓ n=2 l=0 ml=0 ms=+1/2 |
Applications of Pauli’s Exclusion Principle:
By using this rule the maximum number of electrons accommodated by orbitals, shells and subshells can be calculated:
1st shell (L-shell with n=1):
The value of l and mise calculated as
l=0
ml=0
ms =+1/2 or -1/2
So these values give two sets of values for four quantum numbers. And this shell contains a maximum of two electrons that are in 1st orbital.
2nd shell (L-shell with n=2):
This will eight sets of quantum numbers that show the second shell consist of eight electrons in which two electrons are in 2s-subshell while the remaining six are in 2p-subshell.And these 2p subshell are distributed as 2px,2py,2pz orbitals as 2Px2 ,2Py2 ,2Pz2 .
Third shell (M and N shells with n=3 and n=4):
3rd shell accomodate 18 electrons(n=3)
2 in 3s-sub shell,6 in 3p-subshell and 10 in 3d-subshell.
4th shell accomodate 32 electrons(n=4)
2 in 4s-subshell,6 in 4p-subshell,10 in 4d-subshell and 14 in 4f-subshell.
Following table helps to understand how Pauli’s exclusion principle helps to find number of electrons in orbitals:
| Main shells | Combination number | Values of n,l,ml ,ms | Electron spin | Total electrons in orbitals and sub-shells | Total electrons in main shell |
| 1st shell k-shell (n=1) | 1 2 | n=1,l=0,ml=0,ms=+1/2 n=1,l=0,ml=0,ms=-1/2 | ↑ ↓ | 2(1s2)(↑↓) | 2or 2×12=2 |
| 2nd shell L-shell n=2 | 1 2 | n=2,l=0,ml=0,ms=+1/2 n=2,l=0,ml=0,ms=-1/2 | ↑ ↓ | 2(2s2)(↑↓) | 2 × 22 =8 |
| 3rd shell M-shell n=3 | 3 4 | n=2,l=1,ml=0,ms=+1/2 n=2,l=1,ml=0,ms=-1/2 | ↑ ↓ | 2(2pz2)(↑↓) | 2 × 32 =18 |
| 4th shell N-shell n=4 | 5 6 | n=2,l=1,ml=+1,ms=+1/2 n=2,l=1,ml=+1,ms=-1/2 | ↑ ↓ | 2(2py2)(↑↓) | 2×42 =32 |
| 5th shell O-shell n=5 | 7 8 | n=2,l=1,ml=-1,ms=+1/2 n=2,l=1,ml=-1,ms=-1/2 | ↑ ↓ | 2(2px2)(↑↓) | 2×52 =50 |
Hund’s Rule for maximum multiplicity:

Hund’s Rule helps to understand the concept of degenerate orbitals.
According to this rule:
Firstly in the case of degenerate orbitals the orbitals with the same energy, if two electrons opposite spin is placed in same orbital the electrostatic repulsion becomes high as compared when they are placed in seprate orbital repulsion becomes low.
So for least repulsion it is necessary electrons in degenerate orbitals must be placed separately until it is necessary to place them together in the same orbitals.
The most stable arrangement is one in which there are unpaired electrons present.
For example :
Stable electronic arrangement for p2 configuration. There are many ways to place electrons in p -orbital three of them are :
A:
| ↑↓ |
B:
| ↑ | ↑ |
C:
| ↑ | ↓ |
Exchange energy is the energy decrease per exchange pair is called exchange energy.
- In above example there one exchange pair in arrangement B and nothing in C.
- There is a loss of energy in B in form of exchange energy.
- The arrangement B with less energy is more stable than C.
Determination of exchange pair of electrons:
The electronic configuration p3 has twenty electronic arrangements three of which are as:
1.
| 1 | 2 | 3 |
| ↑↓ | ↑ |
2.
| 1 | 2 | 3 |
| ↑ | ↑ | ↑ |
3.
| 1 | 2 | 3 |
| ↑ | ↓ | ↑ |
According to the first part of Hund’s Rule:
- Arrangement 1 is least stable.
- Arrangement 2 has three exchange pairs namely 1,2 and 1,3 and 2,3.
- Arrangement 3 has one exchange pair 1,3.
- The energy of arrangement 2 is decreased by 3E and arrangement 3 by E .
- If E is exchange energy then arrangement 2 has lowest energy and it is most stable one.
According to the second part of Hund’s Rule:
Electronic arrangement with a maximum number of parallel electron spin will be most stable of given electronic configuration. This type of arrangement has maximum exchange pairs and has low energy or the highest stability.
Steps to calculate exchange pairs of electrons:
1.Find out the number of electrons with upward spin. Let the number be m.
And the number of exchange pairs will be as;
mC2 = m!/2(m-2)!
=m×(m-1)×(m-2)…2×1/2×(m-2)×(m-3)…2×1
2.Find out the number of electrons with downward spin. Let the number be n.
The number of exchange pairs of n electrons will be as:
nC2 =n!/2(n-2)!
=n×(n-1)×(n-2)…2×1/2×(n-2)×(n-3)…2×1
3.Total number of exchange pair of electrons with upward and downward spin are:
mC2 + nC2 = [ m!/2(m-2)! ]+[ n!/2(n-2)! ]
=[m×(m-1)×(m-2)…2×1/2×(m-2)×(m-3)…2×1]+[ n×(n-1)×(n-2)…2×1/2×(n-2)×(n-3)…2×1]
For arrangement 1:
The number of exchange pairs will be as;
=2C2 +1C2
=2!/2(2-2)! +0
=2×1/(2×0)! +0
=2×1/2×1 +0
=1
For arrangement 2 :
The exchange pairs of electrons are
=3C2 +0C2
=3!/2(3-2)! +0
=3×2×1/2×1 +0
=3
For arrngement 3 :
The number of exchange pair of electrons are:
=2C2 +1C2
=1
Pairing energy:
The amount of energy required to place two electrons together in same orbital is called pairing energy.The placing of two electrons involves considerable repulsion.
- It is exothermic process.
- Has positive sign and increase energy of system.
- Destabilise the system.
- The overall stability of system depends on pairing energy(P) and exchange energy(E).
For electronic arrangements 1,2,3 for configuration p3 aggregates of pairing and exchange energy are:
-E+P,-3E+0,-E+0 respectively.
Order of stability for these three arrangements:
2 ˃3˃1.
Relative stability of stable electronic arrangement of different electronic configuration:
According to Hund’s Rule :
Configurations p1 ,p2 and p3 have following electronic arrangements:
1.
| ↑ |
2.
| ↑ | ↑ |
3.
| ↑ | ↓ | ↑ |
Comparison of 1,2 and 3:
| characteristic | 1 | 2 | 3 |
| No.of exchange pair | 0 | 1 | 3 1,2 ;1,3 ;2,3 |
| Exchange energy | 0 | -E | -3E |
| Pairing energy | 0 | 0 | 0 |
| stability | Least stable | stable | Most stable |
NOTE: p3 is the most stable configuration because of the maximum exchange energy of all three degenerate orbitals.
The stability order is as follow;
p1 ˂p2 ˂p3 .
Configurations of d1, d2 ,d3 ,d4 and d5 have following electronic arrangement:
1.
| ↑ |
2.
| ↑ | ↑ |
3.
| ↑ | ↑ | ↑ |
4.
| ↑ | ↑ | ↑ | ↑ |
5.
| ↑ | ↑ | ↑ | ↑ | ↑ |
Compaison of 1,2,3,4,5 arrangements:
| characteristic | 1 | 2 | 3 | 4 | 5 |
| No.of exchange pairs | 0 | 1 | 3 | 6 | 10 |
| Exchange energy | 0 | -E | -3E | -6E | -10E |
| Pairing energy | 0 | 0 | 0 | 0 | 0 |
| stability | Least stable | Stable than 1 | Stable than 2 | Stable than 3 | Most stable |
The electronic arrangement consists of half-filled degenerate orbitals that are stable than others.



Leave a Reply