
The equation E=nhv is used to find the energy of the photon. Energy can also be released or absorbed in small discrete packages that mean energy is quantized. Each package is termed ”quantum”. Einstein expanded on this theory with Planck’s to show that radiant energy is also quantized and the quantum of radiant energy is called a photon. So, Einstein shows that light behaves not only as a wave but also as a particle or photon and each photon of radiant has energy is directly related to the frequency of that radiant determined by planks:
E=hv
where,
- E=Energy of photon
- P=Plank’s constant
- V=frequency
we can also explain the above equation as,
E=nhv
where,
- n=no. of photons
It is important to note here that n is the actual number of photons (not the number of moles)
What is the energy associated with a mole of photons of red light (714nm)?
E=nhv —— (1)
As
c=λv
v=c/λ
v=3.00×108 ms-1 / 715nm
v=3.00×108 ms-1 / 715nm × 109 nm / 1m
v=4.20×1014 s-1
put in eq (1)
∴ 1 mole of photon contains 6.02 ×1023 photons (Avogadro’s number is equal to 1 mole)
E=1mole × 6.02×1023/mol × 6.62×10-34 × 4.20×1014 s-1
E=1.68×105 J
So, one mole of a photon of red light at 750nm will have energy equals E=1.68×105 J
Electromagnetic radiation like light, X-rays, etc. can produce the phenomenon of interference, diffraction, and polarization due to their wave nature. But under certain circumstances, they can produce the photoelectric effect and Compton effect which explains the particle nature. It means that electromagnetic radiations have dual nature, wave as well as particle nature.
Photoelectric Effect:
The photoelectric effect is an example of the interaction between radiation and matter.

If the light of suitable frequency is made to fall over a metal surface, it ejects electrons from the metal surface is known as the Photoelectric effect and the electrons ejected from the metal surface during the photoelectric effect are called Photoelectrons.
When light is shining over the cathode surface, the electrons are emitted from the cathode or metal surface and the anode attracts the photoelectrons. The cathode and anode are connected to the battery and attached to the voltmeter to measure the potential. An ammeter is also attached to measure the current due to photoelectrons which are known as photoelectric current.
Problems that can’t explain by classical theory:
Classical physics fails to explain three main problems:
- Intensity problem
- Frequency problem
- Time delay
Intensity problem:
According to classical physics,
Energy? (Amp)2
- Dim light means low amplitude. If we use dim light, both the intensity and energy are low.
- Bright light means high amplitude. If we use bright light instead of dim light, the more the intensity of light, more the energy.
According to classical physics, when we use dim light in the photoelectric effect, the kinetic energy of photoelectrons is low. But when we use a bright light, the kinetic energy of photoelectrons is high.
Condition:
The light (bright or dim) is monochromatic means the same wavelength of light is used.
It is important to note here, How can we measure the kinetic energy of photoelectrons? We measure the kinetic energy of photoelectrons by reversing the polarity of the battery. Now the photoelectric current will be reduced. The photoelectric current does not drop to zero immediately because the photoelectrons are emitted from the metal surface at different speeds. Some will react to the cathode even though the potential difference opposes their motion. However, if we make the reversed potential difference large enough Vs (stopping potential) at which the photoelectric current drop to zero.
Electrons move due to their kinetic energy. Anode(+) attracts the electrons while cathode(-) repeal the electrons. We measure the kinetic energy of electrons on the basis of repulsion so, when we increase the negative potential of the cathode until no electrons reach the cathode (-ve plate) and the value of the potential at that time is known as stopping potential. Mathematically, the maximum kinetic energy of photoelectrons is given as:
Kmax = eVs
where,
- Vs= stopping potential
- e= charge of an electron
As we know,
- Dim light → low Intensity → low kinetic energy
- Bright light → high intensity → high kinetic energy
By conducting an experiment,
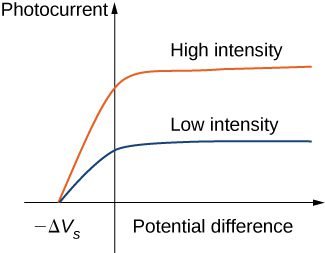
The value of stopping potential for both bright light (high intensity) and dim light (low intensity) is the same. It means the kinetic energy of the dim light electrons and bright light electrons is the same. So, Classical theory fails to explain the intensity problem.
Frequency problem:
Photoelectrons are ejected from the given metal surface when the frequency of incident light is equal to or greater than a particular value, called threshold frequency f°, whatever the intensity of the light may be.
The emission of photoelectrons will not take place from a given metal surface if the frequency of incident light is less than the threshold frequency, whatever the intensity of the light may be.
Threshold frequency:
The minimum frequency of incident light that produces a photoelectric effect is known as the Threshold frequency f°.
- The threshold frequency f° depends upon the nature of the metal surface.
- The energy of photoelectrons depends upon the frequency of incident light and is independent of the intensity of light.
Time Delay problem:
According to classical theory, there must be a time interval between the incident light on the metal surface and the ejection of photoelectrons. When we use dim (low intensity) light, electrons are not emitted quickly (time delay to eject the electrons) but when we use bright (high intensity) light, electrons eject from the metal surface immediately. However, the experimental results show that there is no time delay between incident light and the emission of photoelectrons provided that the frequency of light is equal to or greater than the threshold frequency.
These three major problems could not be explained by classical theory. However, the Quantum theory has successfully explained the photoelectric effect which was proposed by Einstein in 1905.
Einstein’s explanation of the photoelectric effect:
In 1905, Einstein proposed the Quantum theory to explain the photoelectric effect, according to which the energy of light is not uniformly spread out, the energy is present in small packets (energy packets) called photons.
The Photon:
- Each photon has energy E=hv and momentum p= hv/c, c is the speed, of light.
- All photons of light of particular frequency v, or wavelength λ, have the same energy E= hv=hc/λ and momentum p=hc/λ=h/λ, whatever the intensity of radiation may be. By increasing the intensity of light of a given wavelength, there is only an increase in the number of photons. Thus, the photon energy is independent of the intensity of radiation.
- Photons are electrically neutral and are not deflected by an electric and magnetic fields.
- In a photon-particle collision, the total energy and momentum are conserved. However, the number of photons may not be conserved in a collision.
- Low intensity means a lesser number of the photon.
- High intensity means more number of the photon.
- Low-intensity doesn’t means low energy.
- High-intensity doesn’t mean high energy.
when we increase the intensity (dim→bright) means the number of photons increases in the beam of light. (one photon for one electron)
When is the energy of a photon is differ?
The energy of the photon differs when a different frequency of light is used. More the frequency, the more the energy of the photon, or vice versa.
On which factor the kinetic energy of light depends:
”Kinetic energy of light depends upon the frequency of incident light”
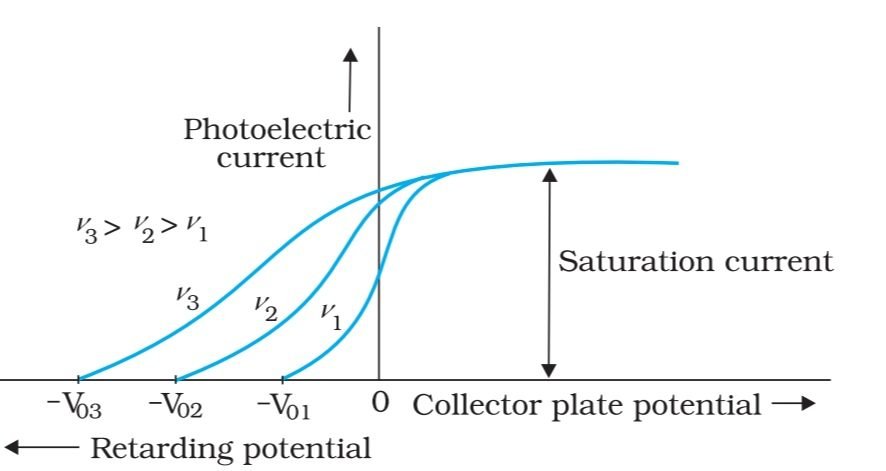
Greater the frequency of light, the greater the kinetic energy of photoelectrons, the more the stopping potential, or vice versa. So, the kinetic energy of incident light depends upon the frequency of light not the intensity of light.
Einstein equation for photoelectric effect:
According to Einstein,
Ephoton = ? + K.Emax
As E=hf
hf = ? + K.Emax
where,
? = work function
Work function:
”The minimum amount of energy required to liberate or eject the electrons from the metal surface”
hf = ? + K.Emax
If the energy of the photon is low, then
hf = ? + 0
? = hf°
Where,
- f°= Threshold frequency
- h= Planks constant
- ?= work function
Why do we need Threshold frequency?
Let us have an atom and we want to emit its valance electron by shining light in the form of a photon.

If the energy of a photon is not equal to the ionization energy of the atom, the electron is not ejected. So, If we want to remove the valance electron, the energy of the photon must equal the ionization energy of an atom that’s why we need threshold frequency for the photoelectric effect.
Compton Effect or Compton Scattering:
Many problems like black body radiation, photoelectric effect, and Compton effect can’t explain by classical theory because matter exhibits the property of both waves as well as particles.
Electromagnetic waves are actually waves but they can behave like particles. Basically, the Compton effect studies the particle behavior of electromagnetic waves. There are two important things in particle behavior. During the interaction, particles transfer both energy and momentum. So, Compton explains the concept of Compton scattering on the basis of quantum theory.
Explanation:
”The wavelength of scattered X-ray is larger than incident X-ray”
Authur Holly Compton was an American physicist who won the Nobel prize in physics in 1927 for his discovery of the Compton effect. He studies the scattering of X-rays from a graphite target. They shine the X-rays on a graphite target and measure the wavelength of scattered X-rays. He observed that the wavelength of scattered X-rays is larger than incident X-rays. One thing kept in mind is that, the larger the wavelength, the smaller the energy.
E α 1/λ
After scattering, if the wavelength of X-rays increases, it means energy is decreased. Why did the energy of scattered X-rays decrease? When the X-rays interact with matter they transfer their energy and momentum. In the photoelectric effect, only energy is transferred, But in Compton scattering energy, as well as momentum, is transferred.

Compton shines the photon on a graphite target having loosely bounded electrons (free electrons). When a photon interacts with the electron, both the photon and electron scatter in opposite directions and make an angle θ with their original direction. The X-ray photon is scattered at different angles and measured by a detector. After observation from the detector, the wavelength of scattered X-ray photons is greater than the incident photon. The change in wavelength after scattering is called Compton shifting.
∆λ=h/m°c × (1-cosθ) ——- (1)
Where,
- h= planks constant
- m°= rest mass of the electron
- c= speed of the electron
- ∆λ= change in wavelength after scattering
∆λ= λf – λi
Where,
- λf = wavelength after scattering
- λi = wavelength before scattering
Eq (1) becomes,
λf – λi= h/m°c × (1-cosθ)
Compton wavelength:
The factor h/m°c is constant and known as Compton wavelength.
h/m°c = 6.63×10-34 / 9.1×10-31 × 3.00×108
h/m°c = 2.43×10-13 m
Compton shifts at different angles:
At 90°:
∆λ=h/m°c × (1-cosθ)
∆λ=h/m°c × (1- cos 90)
∆λ=h/m°c × (1- 0)
∆λ=h/m°c = Compton wavelength
At 0°:
∆λ=h/m°c × (1-cosθ)
∆λ=h/m°c × (1-cos 0)
∆λ= 0 (no Compton effect)
The photoelectric effect (1905) and Compton effect (1923) confirm the particle nature of electromagnetic radiation.
Wave Nature of Matter:
The wave nature of light shows up in the phenomenon of interference, diffraction, and polarization. On the other hand, with the photoelectric effect and Compton effect which involve energy and momentum transfer, radiation behaves as if it is made up of a bunch of particles called photons.
In 1924, the French physicist Louis Victor de Broglie put forward his hypothesis that moving particles of matter should behave with wave-like properties under suitable conditions. De-Broglie proposed the wavelength λ associated with a particle of momentum p as given:
λ = h/p = h/mv —– (1)
Where,
- m = mass of the particle
- v = speed of particle
- λ = Broglie wavelenght
The above equation is known as the Broglie relation. The validity of this hypothesis can be tested only by experiment. It is interesting to note here that it is also satisfied by a photon. For a photon,
p = hv/c
h/p = c/v = λ
It is clear from eq (1), the heavier the particle (large m) or more energetic particle (large v), the smaller the wavelength λ. For example, the de-Broglie wavelength of a ball of mass 0.12 kg moving with a velocity of 20 m/s is,
p=mv = 0.12 kg × 20m/s = 2.40 kg m/s
λ = h/p
λ = 6.63×10-34 / 2.40 kg m/s
λ = 2.76×10-34 m
This wavelength is so small, Which is the reason why macroscopic objects in our daily life do not show wave-like properties.
Wave character of Subatomic particles:
On the other hand, in sub-atomic particles, the wave character of particles is significant and measurable. Consider an electron of mass m and charge e accelerated from rest through a potential V. The kinetic energy of the electron is given by:
K.E = eV
As
λ= h/p
K.E = 1/2 mv2 = p2 /2m so that,
p = √2 m K.E
p = √2 m eV
by putting the value of p
λ= h /√2 m eV
putting the value of h, m, e we get,
λ = 1.227/√V nm ——- (A)
This shows that matter-wave associated with an electron could be verified by crystal diffraction experiments analogous to X-ray diffraction. In 1929, de Broglie was awarded the Nobel prize in physics for his discovery of the wave nature of electrons.
Davission and Germer Experiment:
In 1928, the wave nature of electrons was first experimentally verified by C.J. Davisson and L.H. Germer and independently by G.P. Thomson. They observed diffraction effects with a beam of electrons scattered by crystals. Davisson and Germer shared a Nobel prize in 1937 for their experimental discovery of the diffraction of electrons by crystals.
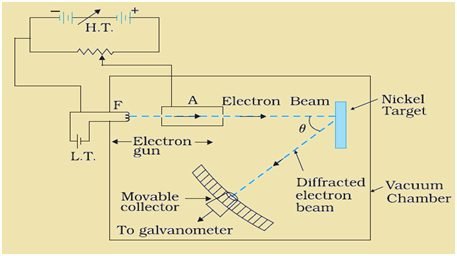
Appratus:
It consists of an electron gun which comprises a tungsten filament F, coated with barium oxide and heated by a low-voltage power supply. Electrons emitted by the filament are accelerated to a desired velocity by applying a suitable voltage from a high-voltage power supply. They are made to pass through a cylinder with fine holes along with its axis, producing a finely collimated beam. The beam is made to fall on the surface of a nickel crystal.
The electrons are scattered in all directions by the atom of the crystal. The intensity of the electron beam, scattered in a given direction, is measured by the electron detector (collector). The detector is moved on a circular scale and is connected to a sensitive galvanometer, which measures the current. The deflection of the galvanometer is proportional to the intensity of the electron beam entering the collector. The apparatus is enclosed in a vacuum chamber.
By moving the detector on the circular scale at different positions, the intensity of the scattered electron beam is measured for different values of the angle of scattering θ which is the angle between the incident and the scattered electron beam. The variation of the intensity (I) of the scattered electrons with the angle of scattering θ is obtained for different accelerating voltages.
Experiment:
The experiment was performed by varying the accelerated voltage from 44V to 68V. It was noticed that a strong peak appeared in the intensity of the scattered electron for an accelerated voltage of 54V at a scattering angle of θ=50°.
The appearance of the peak in a particular direction is due to the constructive interference of electrons scattered from different layers of the regularly spaced atoms of the crystals. From the electron diffraction measurements, the wavelength of matter waves was found to be 0.165 nm.
The de Broglie wavelength λ associated with electrons, using equation (A), for V=54V is given by,
λ = 1.227/√V nm
λ = 1.227/√54 nm
λ = 0.167 nm
Thus, there is an excellent agreement between the theoretical value and the experimentally obtained value of the de Broglie wavelength. Davisson and Germer’s experiment confirms the wave nature of electrons and the de Broglie relation. The de Broglie hypothesis has been basic to the development of modern quantum mechanics.
Problems:
Problem 1:
Q: An electron, an α particle, and a proton have the same kinetic energy. Which of these particles has the shortest de Broglie wavelength?
For a particle, the de Broglie wavelength
λ = h/p
K = P2/√2m
Then,
λ = h/√2mK
As the kinetic energy of an electron, proton, and α particle are the same, the de Broglie wavelength associated with the particle is inversely proportional to the square root of their masses. A proton is 1836 times heavier than an electron and an α particle is four times that of the proton. Hence, the α particle has the shortest de Broglie wavelength.
Problem 2:
Q: What is the de Broglie wavelength associated with (a) an electron moving with a speed of 5.4×106 m/s, and (b) a ball of mass 150 g traveling at 30 m/s?
For the electron,
mass of electron = m = 9.11×10-31 kg
speed of electron = v = 5.4×106 m/s
As, p = mv
p = 9.11×10-31 kg × 5.4×106 m/s
p = 4.92×10-24 kg m/s
de Broglie wavelength is, λ = h/p
λ = 6.63×10-34 Js / 4.92×10-24 kg m/s
λ = 0.135 nm
For the ball,
mass of ball = m =0.150 kg
speed of ball = v = 30 m/s
p = mv
p = 0.150 kg × 30 m/s
p = 4.50 kg m/s
de Broglie wavelength is, λ = h/p
λ = 6.63×10-34 Js / 4.50 kg m/s
λ = 1.74 × 10-34 m
The de Broglie wavelength of electrons is comparable with X-ray wavelengths. However, the ball, it is about 10-19 times the size of the proton.
Problem 3:
Q: What is the de Broglie wavelength associated with an electron, accelerated through a potential difference of 100 volts?
Accelerated potential = 100 V
de Broglie wavelength is, λ = h/p
As, K.E = 1/2 mv2 = p2 /2m so that,
p = √2 m K.E
p = √2 m eV
by putting the value of p
λ= h /√2 m eV
putting the value of h, m, e we get,
λ = 1.227/√V nm
λ = 1.227/√100 nm
λ = 0.123 nm
The de Broglie wavelength is associated with an electron in this case of the order of X-ray wavelengths.
| Physical Quantities | Symbol | Dimensions | Unit | Remarks |
|---|---|---|---|---|
| Planck’s constant | h | [ML2 T-1] | Js | E = hv |
| Stopping potential | V° | [ML2 T-3 A-1] | V | eV° = Kmax |
| Work function | φ° | [ML2 T-2] | J; eV | Kmax = E- φ° |
| Threshold frequency | v° | [T-1] | Hz | v° = φ°/h |
| de Broglie wavelength | λ | [L] | m | λ = h/p |



Leave a Reply