
In the world, approximately many things contain Glycosidic linkage in them. Sugar is a monosaccharide that we use in our homes for many purposes. This sugar contains Glycosidic linkage in them. Not only this it is also present in the shells of lobster which is a sea animal. This blog will basically provide a piece of information about what is a glycosidic bond?
Before discussing the Glycosidic linkage, we will have the understand the concept of the Hemiactal or Hemiketal group of saccharides. This would help us to grasp the concept of Glycosidic linkage. So, let’s move on to discuss what is Hemiacetal or Hemiketal??
What is Hemiacetal or Hemiketal???
When an alcohol-containing -OH functional group gets reacted with any carbonyl compound such as aldehyde & ketone results the product is called Hemiacetal or Hemiketal. They have similar general formula R1R2C(OH)OR. In this formula, R1 or R2 may be either hydrogen or any other organic substituent.
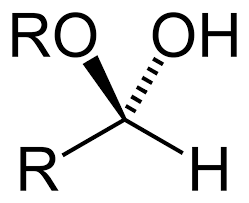
Hemiacetal or Hemiketal are the facts and figures in which there is the addition of alcohol to carbonyl group aldehydes or ketones. Some Hemiacetals are cyclic in nature. These cyclic hemiacetals are known as lactols. They have the same functional group but differ in the number of rings.
Now, the following paragraph will show you how a hemiacetal is formed. It is formed when there is an additional reaction between an aldehyde and a compound containing OH in it.
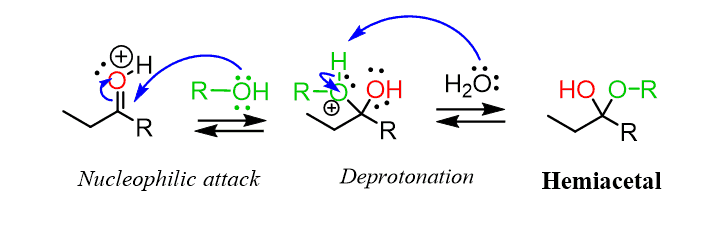
Hemiacetals are also present in the solution of aldehyde in alcohol. Mostly, hemiacetals are sugar in nature.
What is Glycosidic linkage??
Glycosidic linkage is also defined as the covalent linkage that is used to link one ring to another ring.
Glycosidic linkage is basically the covalent linkage between sugar molecules (monosaccharides). As a result, the product obtained is called Glycoside. Glycosidic linkage containing compounds is called Glycoside. Glycosidic linkage can often be formed between hemiacetals or hemiketals and the hydroxyl-containing group. So, a glycoside is formed.

Difference between α & β Glycosidic linkage:
α & β Glycosidic linkage are the distinctions regarding Glycosidic linkage. These distinctions are valuable due to the orientation of the bond. Distinctions of α & β Glycosidic linkage are based on stereocentres and chiral centers. When two carbons in a Glycosidic linkage share the same stereochemistry is called α-Glycosidic linkage.
When two carbons in the Glycosidic linkage share two different stereochemistry that is called β-Glycosidic linkage. In α-Glycosidic linkage, D-sugar moves below the plane and the hydroxyl on the other carbon atom points above the plane. But in β-Glycosidic linkage, there is a reverse configuration. D-sugar points above the plane and hydroxyl on the other carbon atom points below the plane. Alpha 1,4 – Glycosidic linkage is formed in maltose and Beta 1,4 – Glycosidic linkage is formed in Lactose.

N – Glycosidic linkage:
It is very keenly observed that a reaction between second alcohol and hemiacetal or hemiketal may result in the acetal which has a unique glycosidic linkage in it. But if an amine is attacked instead of alcohol then the bond formed between amine and hemiacetal or hemiketal is purely N-glycosidic linkage.
Glycosidic linkage is also present in the structure of ATP (Adenosine triphosphate). ATP structure contains a five-carbon ribose sugar, nitrogenous bases, and phosphate group. The bond by which the nitrogenous base is attached to the five-carbon sugar is called N-Glycosidic linkage.
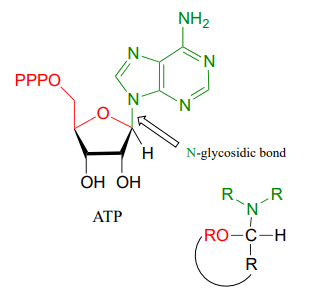
Formation of N-glycosidic bond
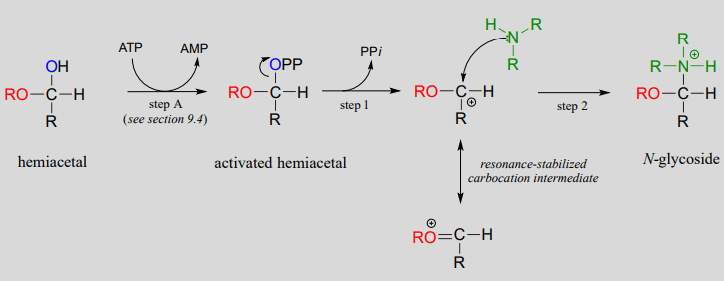
The formation of N-glycosidic linkage is similar to the SN1 nucleophilic substitution reaction. The mechanism of formation of N-glycosidic linkage involves the following steps:
- A hemiacetal is converted into activated hemiacetal by conversion of ATP into AMP.
- This activated hemiacetal by taking inorganic phosphate is converted into resonance stabilized carbocation as an intermediate.
- In the next step, an amine attacked the intermediate formed and results in the formation of N-glycosidic linkage.
What are the uses of Glycosidic linkage??
Following are the uses of Glycosidic linkage:
- Glycosidic linkage is formed in between Amylose which is great food storage for animals.
- It is also present in cellulose. Cellulose is cotton or wood-like material which is present in the stems of plants.
- Chitin is present in the exoskeleton of the beetle, crabs, and lobsters. Chitin has a complex glycosidic linkage.



Leave a Reply