
Osmosis is an important colligative property of solution and was first reported by Abbe Nollet in 1748. Osmosis does not occur when a cell is in an isotonic solution it is because the isotonic solution means the number of particles inside and outside of the cell is equal. (no osmosis and no diffusion is occurring)
To understand the term osmosis, first, we discuss diffusion.
Diffusion:
“The net moment of any substance from the region of their higher concentration to their region of lower concentration”
Consider two solutions ”A” and ”B”, solution ”A” with concentration ”CA ” and solution ”B” with concentration ”CB ”. let we assume that the concentration of ”A” is greater than ”B” means the particles of solute in solution ”A” is greater than solution ”B” but the concentration of solvent is greater in solution ”B”.
- More the solution concentration means more the solute particles in that solution.
- lessor the solution concentration means more the solvent particles in that solution.
- In chemistry, the priority of the term ”concentration” is given to solute.
Osmosis:
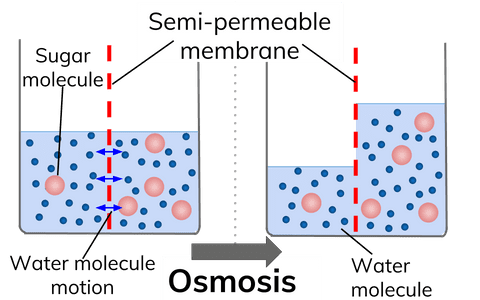
Abbe Nollet observed that when a solution is separated from a pure solvent by a suitable membrane, there is a spontaneous flow of solvent into the solution. This phenomenon of the spontaneous flow of solvent through a membrane into a solution or form a dilute solution to a concentrated is termed osmosis. (Greek osmos = a push)
In a semi-permeable membrane, pores are very small which permits to pass only one thing (solvent) because the particles of solute are slightly larger than solvent that cannot permit to pass through SPM. The movement of particles is stopped until the concentration is the same on both sides (attain equilibrium).
”The movement of solvent molecules from their higher concentration to their lower concentration through a semi-permeable membrane is called Osmosis”
OR
”The movement of solvent molecules from a lower concentration of solution to higher concentration through a semi-permeable membrane is called Osmosis”
Difference between Osmosis and Diffusion:
| Osmosis | Diffusion |
| There is the flow of solvent into the solution through a semi-permeable membrane. | There is the flow of both the solvent and solute and no semi-permeable membrane is required. |
| Solvent flows from a solution of lower concentration to a solution of higher concentration. | The solution flows from higher concentration to lower concentration until equilibrium in concentration is achieved. |
Semi-permeable membrane:
It is a special type of membrane having small pores and allow to pass only solvent particles no solute particles.
Examples:
- Animal membrane
- Cellophane
- Copper ferrocyanide Cu2 [Fe(CN)6 ]
Osmotic pressure:
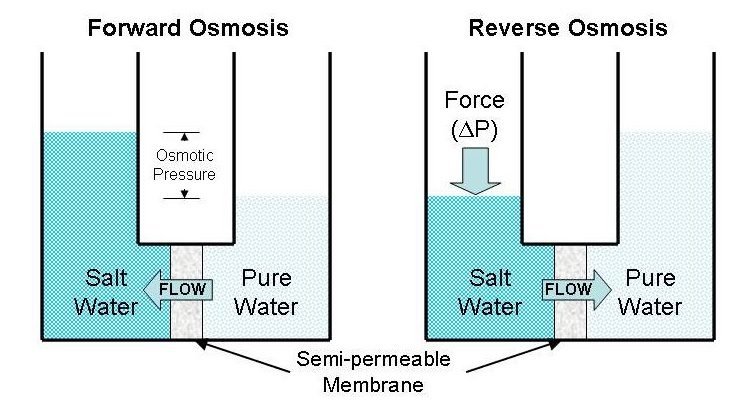
By applying certain pressure to the solution osmosis can be prevented. The minimum pressure required to prevent osmosis is known as osmotic pressure. The osmotic pressure of a solution is a colligative property and at a given temperature its magnitude depends only on the concentration of solute. The membrane used (SPM) depends on the nature of solute and solvent.
”The net movement of solvent molecules from solvent to the solution can be stopped by applying some extra pressure over solution side is called Osmotic pressure”
OR
”The extra pressure over solution side which stops osmosis is called Osmotic pressure”
Symbol:
The symbol used for osmotic pressure is ”π”
Experimentally:
π ∝ M —–(1)
π ∝ T —–(2)
Combine (1) and (2)
π ∝ MT
π = MRT
Where,
- π = Osmotic pressure
- M = Molarity of solution
- T = Absolute temperature (in K)
- R = Solution constant
The value of R is same as universal gas constant (0.0821 atm l/mol K)
Note:
π = MRT is same as gas law
π = MRT
π = n/vRT
PV = nRT is a gas law.
1atm = 1.01 × 105 Pa = 1.01 × 105 N/m2 ≅ 1.01 bar = 760 mmHg = 760 Torr
Reverse Osmosis:
- Reverse osmosis is a non-spontaneous process that needs some external pressure.
- if we apply more pressure on the solution side, RO start.
Isotonic solutions:
Solutions in which no flow of solvent molecules through a semi-permeable membrane are called isotonic solutions.
Let us consider two solutions of different concentrations having the same solvent and separated by a semi-permeable membrane. The solvent molecules will flow from the solution of low osmotic pressure toward the solution having high osmotic pressure. This process will continue until both the solution attains the same osmotic pressure. Such a pair of solutions will say to be isotonic with each other. For example, a 6% solution of urea and 18% solution of glucose is isotonic with each other, because they have the same osmotic pressure.
π1 = π2
M1RT1 = M2RT2
We assume that temperature is the same
M1 = M2
Hypotonic solutions:
Hypotonic solutions mean less osmotic pressure. π1 < π2
Hypertonic solutions:
Hypotonic solutions mean more osmotic pressure. π1 > π2
Measurement of Osmotic pressure:
Osmotic pressure is generally measured by the following method:
Berkely and Hartley Method:
It is an accurate method for determining the osmotic pressure of dilute solutions. In this process, the osmosis is prevented by applying pressure to the solution, and the magnitude of this applied pressure gives the osmotic pressure of the solution. Therefore, solvent is placed in horizontal tube A, on the sides of which a thin layer of copper ferrocyanide is deposited. This tube is placed utilizing water-tight joints into a metal jacket B which contains the solution and caries an attachment C through which pressure can be applied.
Tube A is first filled with the solvent through a stop cork up to a defined point in the capillary D. As a result of osmosis, the level of the solvent in the capillary will fall, but by applying pressure through C, it is restored to its initial value. However, This pressure is taken as the osmotic pressure of the solution.

Laws of Osmotic pressure:
Van’t Hoff-Boyle’s Law:
According to this law at a constant temperature, the osmotic pressure of a dilute solution is directly proportional to its concentration.
Mathematically:
π ∝ C
π/C = constant
The concentration is expressed in molar units so that C=1/V when 1 g mole of the solute is present in V dm3 of the solution. Hence
πV = constant
This is known as Van’t Hoff Boyles law.
Van’t Hoff Charles Law:
At constant temperature, the osmotic pressure of a dilute solution is directly proportional to absolute temperature.
Mathematically:
π ∝ T
π/T = Constant
This relation is similar to Charles Law and is known as Van’t Hoff Charles law.
Van’t Hoff Avogadro’s Law:
At constant temperature, solutions contain an equal molar concentration of different solutes having the same osmotic pressure.
Mathematically:
π ∝ n
By combining three laws, we get
π ∝ nT/V
π = n/V.RT
πV = nRT ——(A)
Van’t Hoff equation explains the relation between π, V, and concentration, just as PV = nRT explains the behavior of an ideal gas.
as, C = n/V (no of moles per unit volume)
So, equation (A) can be written as
π = CRT
Determination of Molar Mass from Osmotic Pressure Measurements:
If the mass of a non-volatile and non-electrolytic substance having molar mass M is present in V dm3 of the solution, then
n = m/M
From eq (A),
πV = m/M.RT
M = MRT/ πV
Where,
- m = mass os solute
- T = Absolute temperature
- π = Osmotic pressure
- V = volume
- R = Constant
Problems related to Osmotic Pressure:
Q 1: Calculate the osmotic pressure due to 5% w/v cane sugar solution at 15℃?
Cane sugar = C12H22O11 = M = 342
5% w/v sugar cane means 5 g of sugar cane in 100 ml of solution.
Temperature = 15℃ = 273℃ + 15℃ = 288 K
π = MRT
π = n/V×RT
π = 5/342 /V×RT
π = 5/342 × 1000/100 × 0.0821 × 288
π = 5/342 × 10 × 0.0821 × 288
π = 1182.24/342
π = 3.45%
Q 2: 200 cm3 of aqueous solution contains 1.26 g of a polymer. The osmotic pressure of a solution is 2.6 × 10-3 atm at 300 K. Calculate the molar mass of polymer?
π = MRT
π = n/V×RT
2.6 × 10-3 = 1.26/M × 1000/200 × 0.0821 × 300
2.6 × 10-3 = 1.26/M × 5 × 0.0821 × 300
M = 1.26/ 2.6 × 10-3 × 5 × 0.0821 × 300
M = 155.169/ 2.6 × 10-3
M ≅ 60,000 g/mol
Q 3: A 5% w/v cane sugar solution is isotonic with 0.877% urea solution. if the molar mass of cane sugar is 342, calculate the molar mass of urea?
π1 = π2
M1RT1 = M2RT2
R is general gas constant and temperature is same, we have
M1 = M2
- 5% w/v cane sugar means 5 g of cane sugar is present in 100 ml of solution.
- 0.877% urea mean o.877 g of urea present in 100 ml of solution.
n1/v1 = n1/v1
volume is same 100 ml so,
n1 = n2
mass/M = mass/M
5/342 = 0.877/Murea
Murea = 200.93/5
Murea = 60 g
Osmotic pressure of sucrose solutions at 273 K:
| Concentration (mol/dm3) | Osmotic pressure Π (atm) | π/c |
| 0.1 | 2.59 | 25.9 |
| 0.2 | 5.06 | 25.3 |
| 0.4 | 10.14 | 25.3 |
| 0.6 | 15.39 | 25.6 |
| 0.8 | 20.91 | 26.1 |
| 1.0 | 26.61 | 26.6 |
The osmotic pressure of 1% sucrose solution:
| Temperature (K) | Osmotic pressure Π (atm) | π/T × 102 |
| 273 | 7.085 | 2.594 |
| 283 | 7.335 | 2.591 |
| 293 | 7.605 | 2.595 |
| 298 | 7.729 | 2.594 |
The above experimental data clear that the osmotic pressure of a solution depends both on temperature and concentration.




Leave a Reply