
In this blog, we will determine the number of valence electrons in the phosphorus atom. This blog will provide you with detailed information about phosphorus and its valence electrons. We will also discuss its properties, applications, and some disadvantages. After exploring this blog, you will be able to know some basics and a detailed overview of phosphorus. Let’s start by discussing ing history and discovery of phosphorus.
History and Discovery of Phosphorus
The word phosphorus is derived from the Greek word (φῶς = light, φέρω = carry) which means light carrier.
It was initially discovered in 1669 by a German chemist Henning Brand. He took an experiment with urine. He attempted to create the fabled philosopher’s stone through the distillation of some salts by evaporating urine. In this experiment, Brand got a white material that was glowing in the dark and burned nicely. It was named phosphorus mirabilis.
In 1680 Robert Boyle was the first man who use phosphorus to ignite sulfur-tipped wooden splints.
Later on in 1777, it was acknowledged as an element by Antoine Lavoisier.
Introduction
Phosphorus is an element present in group 15 (5A) and in the third period with the symbol P, atomic number 15, and atomic mass 30.973. There are two major forms of phosphorus, white phosphorus, and red phosphorus. Phosphorus is not found freely in nature because it is highly reactive.
How we can determine the valence electrons in Phosphorus?
We can use two methods to determine valence electrons in phosphorus atoms.
- Determining with the help of the periodic table
- Determining by its electronic configuration
Determining with the help of the periodic table
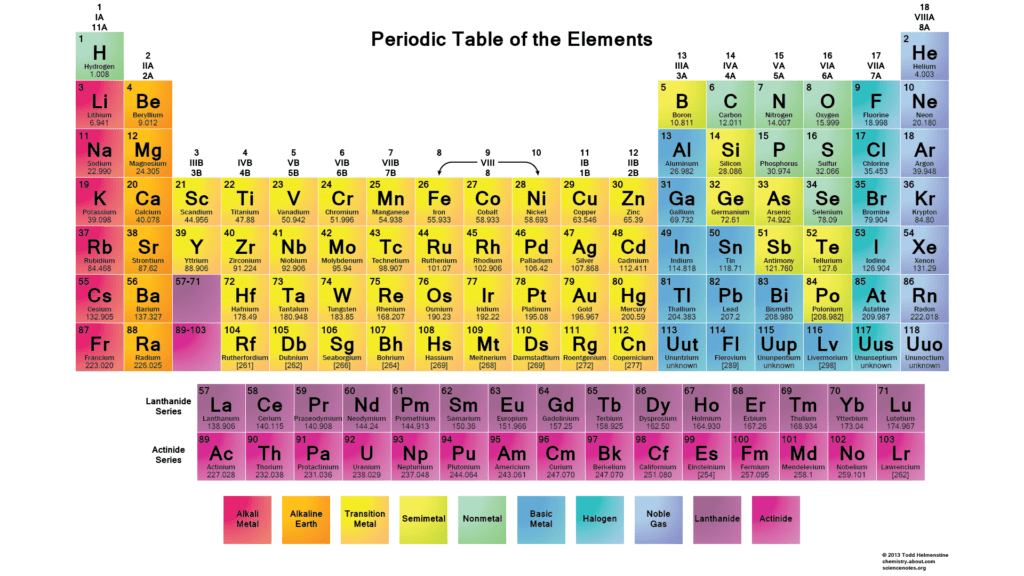
As we can see Phosphorus lies in group 15 (also called group 5A). As every element present in this group will have five valence electrons, phosphorus will also have five valence electrons. The atomic number of phosphorus is 15 which means it has 15 protons since these all are neutral, the number of protons is equal to the number of electrons (15). The atomic mass of phosphorus is 30.97.
Determining by its Electronic Configuration
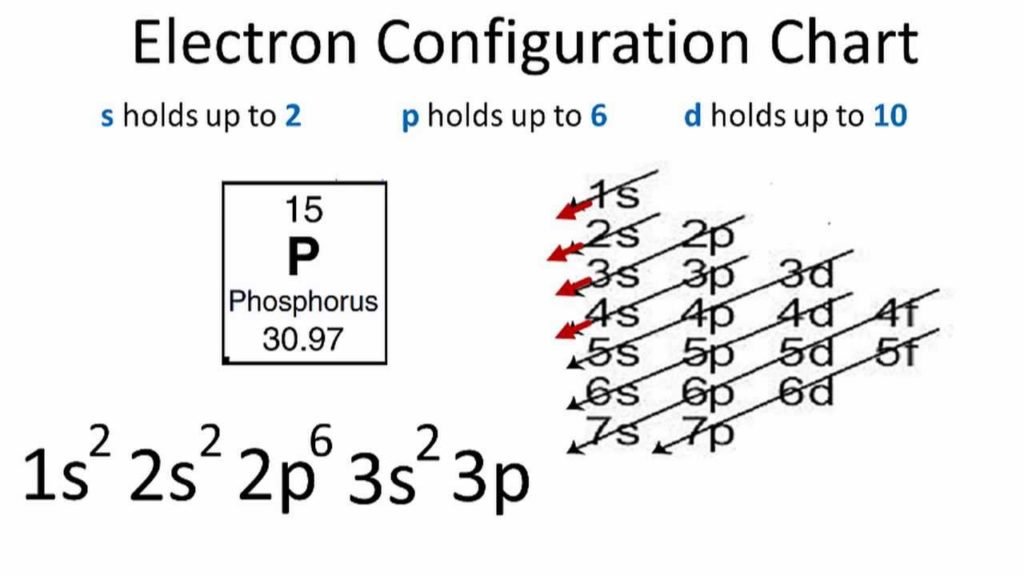
Electronic configuration of phosphorus is: 1s2, 2s2, 2p6, 3s2, 3p3. The outermost orbitals are 3s2, sp3. These outermost orbitals contain 5 electrons (2+3=5 electrons). So, it has 5 valence electrons.
Describe some Properties of Phosphorus?
- It is a highly reactive element.
- White phosphorus is very poisonous and it can spontaneously combust in the air.
- Phosphorus can form different compounds like phosphorus acid, phosphoric acid, and phosphates.
- Its 15 atomic number is 15 and its atomic mass is 30.973 (31).
- It can appear in white, red, and violet colors.
- The boiling point of phosphorus is 553.7 K and the melting point is 317.3 K.
- The heat of fusion of phosphorus is 0.66 KJ/mol.
- The heat of vaporization of phosphorus is 51.9 KJ/mol.
- Phosphorus has different density values for white, red, violet and black phosphorus and these values are 1.823 g/cm3, 2.2 g/cm3, 2.36 g/cm3, and 2.69 g/cm3 respectively.
- The electronegativity of phosphorus is 2.19 on the Pauling scale.
- Phosphorus has a body-centered cubic crystal structure.
- There are three main isotopes of phosphorus 31P, 32P, and 33P. 31P is the most stable among all of these.
Write some uses of Phosphorus?
- Phosphorus is very essential to life.
- Most of our phosphorus comes from phosphate rock and is then converted into phosphoric acid. Approximately Fifty million tonnes are made every year and it is used in multiple fields.
- It is used to make animal feeds.
- It is also used in making rust removers, corrosion preventers, and dishwasher tablets.
- Each molecule of DNA and RNA contains the major part of phosphorus.
- The side of matchboxes is made up of red phosphorus.
- Phosphorus is present in bones. Phosphorus is used in fertilizers.
- It is the 6th most common element in the human body.
- It is the seventh most common element in the earth’s crust.
- It is used in the fertilizer industry.
- Phosphorus compounds are also used in the chemical industry for applications in soft drinks, flame retardants, and pharmaceuticals.
- Phosphorus is a mineral that makes 1% of a person’s total body weight.
- It is needed for the body to make protein for the growth, maintenance, and repair of cells and tissues.
- Phosphorus works with vitamin B.
Disadvantages or Negative Effects of Phosphorus
- Too much use of phosphorus and phosphates in the environment leads algae to bloom in rivers and lakes. The excess algae use up all the dissolved oxygen in the water and fish die.
- Early on, Phosphorus was used in the match head. By overexposing to phosphorus, workers in match factories developed a painful and debilitating deformation of jawbone known as phossy jaw.
- It is highly reactive and can cause damage.
- The white phosphorus is deadly poisonous.
- Access to phosphorus/phosphate in the body can cause diarrhea.
- High phosphate level promotes the progression of kidney disease.
- Excessive soil phosphorus reduces the plant’s ability to take up required micronutrients.
- High phosphorus will interfere with nitrogen absorption, so the plant will lack growth in its stem and foliage, causing stunted growth.
Conclusion
Phosphorus has five valence electrons, as we have determined it. It is an element present in group 15 (5A) with 15 atomic numbers represented by the P symbol. By having five valence and 15 total electrons, It’s electronic configuration is 1s2, 2s2, 2p6, 3s2, 3p3. Different compounds like phosphorus acid, phosphoric acid, and phosphates can be formed from phosphorus. It possesses three main isotopes. Phosphorus has many uses/applications in daily life, in the human body, in fertilizers, in pharmaceuticals, and in plants.



Leave a Reply