In this exciting blog, we will discuss Alpha Helix & Beta Pleated sheets, the basic definition of protein, its formation, and how it is sequenced in structure. Basically, there are four structures of proteins. i.e. Primary structure, Secondary structure, Tertiary structure, and quaternary structure. The structure of proteins is basically categorized on the basis of the joining of the polymer of amino acids by a peptide bond.
Two molecules of proteins may be joined together by a peptide bond. The formation of a peptide bond is basically regarded as a condensation reaction. The following techniques can determine the complexity of the structure of a protein. i.e. X-ray crystallography, NMR spectroscopy, and dual polarization interferometry. In number, ten to several thousand amino acids combine to form a typical size of protein structure. Although, it can also be a nano-particle ranging the size between 1-100nm. Proteins structures may be referred to as conformers and rotomers.
Defining proteins
Proteins are the building blocks of amino acids. Proteins are organic molecules that perform many related functions just like defense, transportation, and organization. These are macromolecules that catalyze the biochemical reaction. Proteins are formed when two amino acids undergo a condensation reaction between two amino acids peptide bond is formed. The Amine group of one amino acid and the carboxyl group of another amino acid combine to form a polymer of protein and water (H2O) is released.
H2N-CH2-COOH + H2N-CH2-COOH → H2N-CH2-CO-HN-CH2-COOH + H2O
Structures of Protein
The structure of a protein may be defined as “POLYMER OF AMINO ACID JOINED BY PEPTIDE BOND“
The structure of proteins, their complexity, their fold into a stable 3-D shape, and their confirmations are described on the basis of the sequence of amino acids. Secondary structures of protein can be determined using the dihedral angle of the peptide bond and the tertiary structure by folding of protein chain in space. There are four structures of proteins described below:
01. Primary structure
- It is a hierarchy basic level.
- Primary structures of proteins are basically regular sequences of amino acids in the chain form in linear form.
- In 1973, Chris Anfinsen demonstrated a unique highly ordered structure of proteins and their importance.
- A linear sequence of amino acids is encrypted in DNA.
- Mutation in the genes of DNA causes mutation or disturbance in the structure of the protein.
02. Secondary structure
- The secondary structure may refer to the local spatial confirmations or sequence of the polypeptide backbone chain.
- In the main chain, a hydrogen bonding between the carbonyl oxygen and the amine group is very strong. So, it twists into a right-handed helix.
- These are conformers in which spatial arrangements are changed.
- They may exist in two forms alpha helix and beta pleated sheets proteins.
03. Tertiary structure
The tertiary structure is the third secondary structure of the protein. In this structure, amino acids are arranged in three-dimensional patterns in a single polypeptide chain.
04. Quaternary structure
Quaternary structures have spatial arrangements. In this, two or more polypeptide chains act as a sub-unit, and these are arranged in spatial arrangements. So, these are called Quaternary structures.
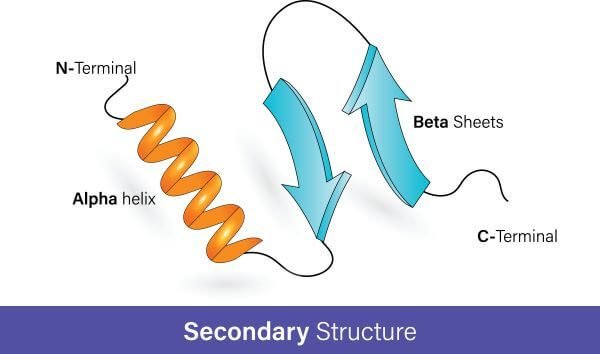
Secondary structure of the protein
Secondary structures of protein may refer to the local spatial confirmation or sequence of the polypeptide backbone chain. It has local folds of the chain due to strong hydrogen bonding and interaction between the amine group and a carboxyl group, forming peptide linkage.
The size of the secondary structure of a protein may depend upon residues in short helices and sheets. Its length may vary from several 3 to 5 residues to over 50 residues. Secondary structures are also defined by the characteristics of the main chain (dihedral angle) and the hydrogen bond between the main chain. The secondary structure of a protein may be in two forms. i.e. alpha helix and beta pleated sheets of protein.
Alpha helix protein:
Linus Pauling was the first scientist who observed the alpha helix structure by x-ray crystallography. It is the most widely common secondary protein. Alpha helix is formed when the backbone of protein due to strong interaction becomes right-handed helical conformation. The right-handed helical conformation has 3.6 residues per turn and also a very strong interaction is present between the main chain amine and a carboxyl group.
mention amine and carboxyl groups also have residue in which hydrogen bonding occurs. In carbonyl (CO), ith residue, and in amine NH (i+4)th residue forms a strong hydrogen bonding between them. Alpha helix may be in the form of a 310 helix. It is the characteristic feature in which a hydrogen bond (a strong interaction) is present by (i) and (i+3) residue.
The standard and optimal angle for the backbone is -57℃ and the optimal dihedral angle is -47℃. But on the other hand, for 310 helices optimal backbone angle is -49℃ and the optimal dihedral angle is -26℃.

The structural integrity of alpha helix
The stability of the alpha helix may refer to structural integrity. The stability of the alpha helix may depend upon steric configuration. Steric configuration is also called steric hindrance. Different amino acids destabilize the alpha helix.
- Too large or too small R-Group of amino acids destabilizes amino acids.
- Proline destabilizes because of irregular geometry.
Structural integrity and stability also depend upon the dipole moment of the complete helix. Neutralization of dipole moment may occur due to the addition of charged amino acids.
Beta Pleated sheets of Protein
The beta-pleated sheet is the second structure related to the secondary structure. Beta-pleated sheets are mainly composed of beta strands. A beta-strand is basically a segment of a polypeptide chain having a network of hydrogen bonds that possess stretched strands. Beta strands are the structural unit of beta pleated sheets. The network of hydrogen bonding exists between the N-H and C=O groups of backbones. The appearance of beta strands appears pleated from tetrahedral chemical bonding.
The hydrogen bonding is present in i and i+2 residue of the main backbone chain. The “pleated” occurrence in beta-strand causes the distance between two residues to decrease. The distance between these residues is 6Å or 0.6nm. The expected distance between both fully extended residues is 7.6Å.
The beta-strands have great extensions perfectly throughout structures that’s why a twist may possible. The changes or fluctuation (twist formation) are coordinated with the dihedral angle the position of beta-strands may depend upon matching up of -N and -C terminals. If these terminals match up or are present in the same direction then the beta-strand must be parallel and if these both terminals oppose each other in different directions then the beta-strand must be anti-parallel.
Structural motifs of the Beta-pleated sheets
Beta-pleated sheets may be in five forms. These are written as under:
- β- hairpin motif.
- Greek key motif
- β-meander motif
- Psi-loop motif
1. β-hairpin motif
The β-hairpin motif has two opposite anti-parallel strands which are linked together by 2 to 5 residues. It is formed due to the dihedral conformation between two amino acids. i.e. glycine or proline.
2. Greek Key motif

In the Greek Key motif, four adjacent anti-parallel strands are present. Hairpins connect three strands and the fourth strand is opposite to 1st which connects it to the loop.
3. β-meander motif

It is a super secondary protein structure composed mainly of two or more anti-parallel beta-strands linked by each other with hairpin loops.
4. Psi-loop motif

It may contain two anti-parallel strands with which hydrogen bonding connects a strand in between them. It is mainly observed in the aspartic protease family.
Difference between Alpha helix & beta pleated sheets
| ALPHA HELIX PROTEINS | BETA PLEATED SHEETS |
| 1. Amino acids have strong hydrogen bonding which twists the coil to the right side. | 1. Amino acids are present in extended confirmations. |
| 2. Alpha helix may be a single chain polypeptide. | 2. The beta-pleated sheets contain two or more polypeptide strands in their structure. |
| 3. Alpha helix polypeptide is formed when 3.6 amino acid residue winds. | 3. The beta-strand polypeptide is formed by the combination of 3 to 10 amino acid residues. |
| 4. Alkyl groups are oriented outside. | 4. Alkyl group present either inside or outside. |
| 5. Example: Haemoglobin, Myoglobin, etc. | 5. Example: Skin fibers |



Leave a Reply