Understanding the endothermic reaction graph involves the understanding of basic definitions of an endothermic reaction. The endothermic and exothermic reaction takes place spontaneously or non-spontaneously. Both reactions are defined on the basis of absorption or releasing of heat. In an endothermic reaction, heat is absorbed from the surroundings into the system. But in an exothermic reaction, heat is evolved from the surroundings to the system.
Surroundings and systems are two opposite things. If we take water in a container. The environment inside the container is called the system and the environment outside the container is called the surroundings. An example of an endothermic reaction is photosynthesis and an example related to an exothermic reaction is respiration.
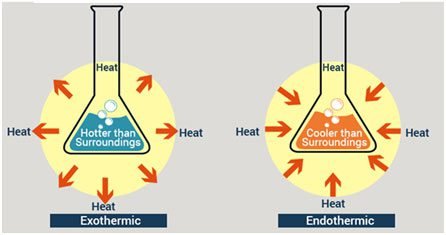
Difference between Endothermic and Exothermic Reaction
The difference between Endothermic and exothermic reactions is as follows in the table:
| ENDOTHERMIC REACTION | EXOTHERMIC REACTION |
| 1. In an endothermic reaction, reactants absorb heat from the surroundings to form products. | 1. In an exothermic reaction, energy is released in the form of heat or light. |
| 2. As the reaction progress, the temperature of a system decreases. | 2. As the reaction proceed or progress, the temperature of the system increases. |
| 3. In this reaction heat or enthalpy of the product is more than that of reactants. | 3. In this reaction heat or enthalpy of reactants is more than that of products. |
| 4. Change in enthalpy by decreasing temperature should be a positive value (∆H = +ve). | 4. Change in enthalpy by increasing the temperature should be a negative value (∆H = -ve) |
| 5. It is non-spontaneous and its energy originated from the environment. | 5. It is a spontaneous reaction and its energy is originated from a system. |
What is an endothermic Reaction????
An endothermic reaction is a reaction in which heat is absorbed from the surroundings to the systems. It is proposed by the scientist Marcellin Berthelot. The term endothermic is derived from two Greek roots “endon” and “therms”. The term “endon” means within and the term ”therms” means hot or warm. So it is also said that an endothermic reaction is a reaction in which heat is absorbed and inside or within the system heart is more and more so the internal system in hot or warm.
In a closed system, there must be the formation or cleavage of bonds. So, if a bond is cleaved it loses a great amount of energy toward the surroundings but also required energy to break this bond, then a reaction is said to be an endothermic reaction. But if a bond is formed in a closed system then it requires great energy to form a bond and also release energy. So, the system release energy to the surroundings. This releasing of energy leads to an exothermic reaction.
For example Electrolysis of water, and photosynthesis. These are the common examples related to an endothermic reaction. In the electrolysis of water, water molecules split up into hydrogen and hydroxyl ions in the presence of an electrical current. So, a great amount of energy is required to break the bond of water. So, the cleavage of the bond leads to an endothermic reaction.
2H2O (l) + Energy → 2H2 (g) + O2 (g)
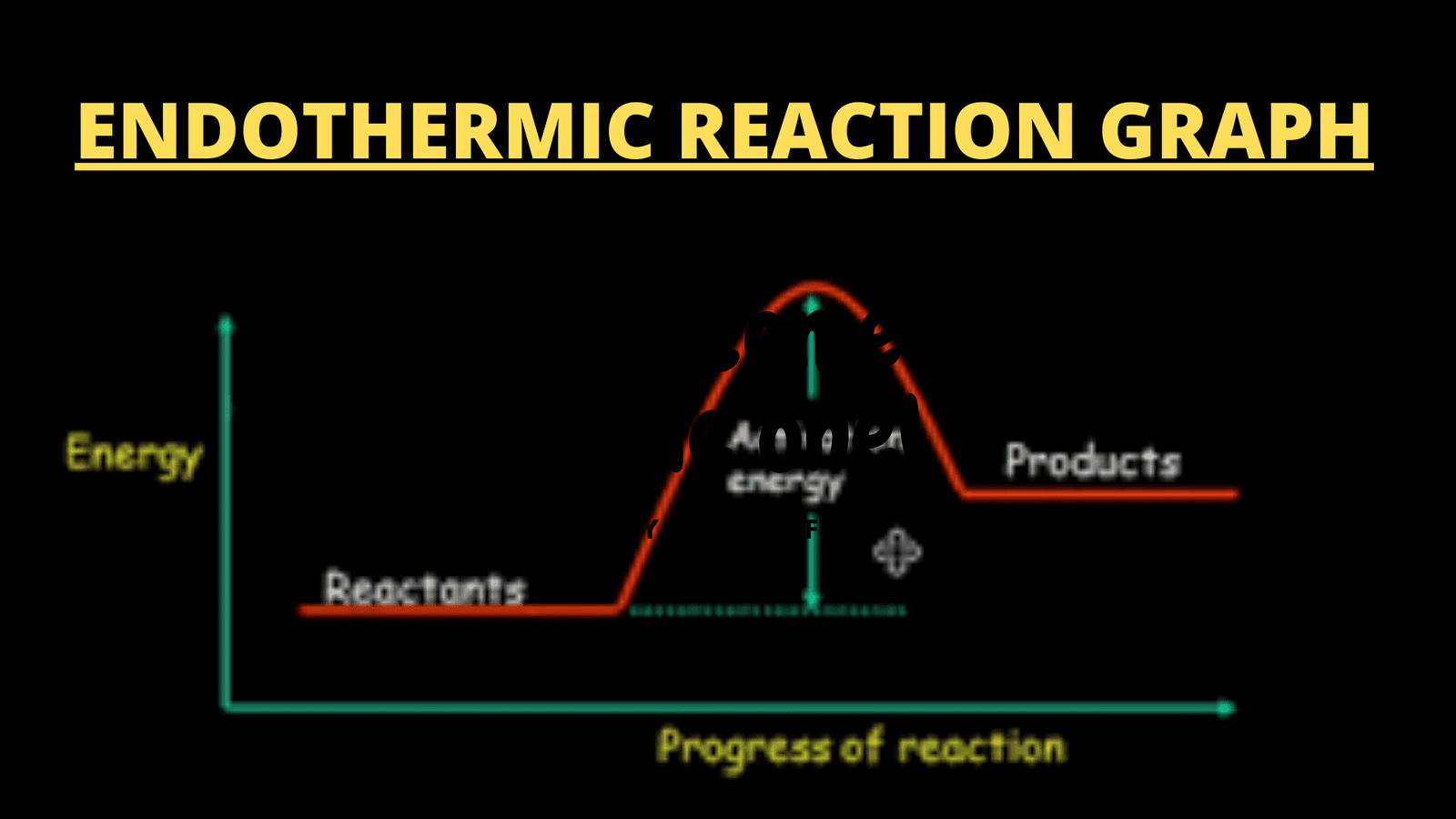
What is Endothermic Reaction Graph????
Energy profile diagram
The graph of any reaction is called an Energy diagram, Energy profile diagram, or potential energy diagram. An energy profile diagram is basically a relationship between a change in energy with the passage of time. It can be represented in the following order:
- Potential energy is represented on the vertical y-axis.
- Time is represented on the horizontal x-axis.
The representation shows two kinds of graphs. One for an endothermic reaction and the other for an exothermic reaction. The releasing or gaining of energy causes a great change in energy. This change in energy occurs regularly by the progression in a reaction from existing reactants to newly formed products.
Enthalpy of a reaction may depend upon energy. Enthalpy represents the heat content of a reaction. When heat energy is absorbed (endothermic bond cleavage) and removed (exothermic bond formation) then there will be a great change in enthalpy. But the question arises how can we measure enthalpy theoretically??
How can we measure enthalpy theoretically??
We can measure enthalpy theoretically by reading a graph and by taking the difference between the enthalpy of reactants before reaction completion and the enthalpy of products after reaction completion. The answer of difference will show a change in enthalpy. As we also know, the value of enthalpy for an endothermic reaction is positive. So, the value becomes positive & reaction must be endothermic.
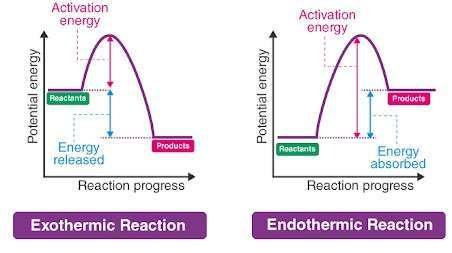
Formation of Transition state & Activation energy
During a reaction, when reactants are proceeding to products, many transition states are formed. So, a difference in energy dominates. In the graph, reactants are shown own low energy state because they have low energy but high stability while the transition state formed as intermediate will be at a high energy state because of its high energy and less stability.
What is Activation Energy & Transition State???
Activation energy may be defined as follows:
- The difference between the energy of reactants in a low energy state and the energy of the transition state (intermediate) in high energy is called Activation Energy.
- It is also defined as the “Minimum energy required to initiate a chemical reaction”.
- The energy required for a reaction to pass the energy barrier and perform the complete transformation of reactants to products is called Activation energy.
A transition state is basically a short-lived state and it is important to know that it has maximum energy. This state is formed when a reaction is at its maximum peak of energy. Maximum energy leads to less stability. So, the transition state has less energy.
What changes occur in the endothermic reaction Graph??
Following are the changes that occur during an endothermic reaction which are then represented on the endothermic reaction graph:
- Heat energy is absorbed from the surroundings into the systems.
- Reactants are transformed into products via a transition state.
- Enthalpy becomes positive greater than zero. i.e, ∆H > 0 or ∆H = +ve
- The energy absorbed is required to reduce the maximum activation energy barrier. So, that product is obtained.
- As we know, endothermic reactions are non-spontaneous. This means to say, this reaction does not occur on its own. It will continue till that when we are applying the condition of heat.
What are the conditions under which Spontaneous endothermic reactions occur??
An endothermic reaction is a non-spontaneous reaction but it will become spontaneous when the following conditions are applied:
- If energy is changed, enthalpy changes which give the -ve value of Gibbs free energy.
- Also, spontaneity in a reaction is enhanced if a change in enthalpy causes an increase in entropy. Entropy is the measure of randomness which is denoted by S.



Leave a Reply