
How many valence electrons does Boron have? There are two ways to find out the valence electron of the boron. The first is just to look at the periodic table. Boron is a member of group number 13 in the periodic table also known as the 3A or IIIA. Boron is the lightest element in this group having atomic number 5. Boron has three valence electrons.
The second method to find the valence electrons of the boron is to write the electronic configuration of the boron. To write the electronic configuration of elements, it’s very important to understand the brief concept of shell and subshell.
How do you Find the Number of Electrons in a Shell?
The electrons revolve around the nucleus in a fixed circular path called the shell. Each shell contains a different number of electrons. The simple formula to calculate the number of electrons in a shell is 2n2 where n is the number of shells 1,2,3 and so on.
How many electrons in K shell?
For K, n=1
2(1)2 = 2 electrons
How many electrons in L shell?
For L, n=2
2(2)2 = 8 electrons
How many electrons in M shell?
For M, n=3
2(3)2 = 18 electrons
How many electrons in N shell?
For N, n=4
2(4)2 = 32 electrons
How Many Subshells are in a Shell?
Shells consist of subshells known as s, p, d, and f. The K shell has only one shell known as s-subshell. The L shell has two subshells known as s and p subshells. The M shell has three subshells known as s, p, and d subshells. The N shell has four subshells known as s, p, d, and f subshells. The s, p, d, and f subshell contains the maximum number of electrons of 2, 6, 10, and 14 respectively.
How many valence electrons does Boron have?
Boron has 5 protons and 5 electrons. The protons are present inside the nucleus but electrons revolve around the nucleus. Now we need to do the electronic configuration of boron. The electronic configuration of boron is 1s2, 2s2,2p1.
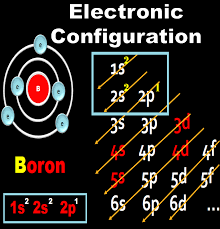
Now you have to find the valency shell, to find the valency shell you have to take the principal quantum number. In case of boron (1s2, 2s2,2p1), the principal quantum number (represented as n) is 1 and 2.
n = 1, 2
To find the valance electron, you have to consider the highest value of n or principal quantum number. In the case of boron, the highest value of n is 2. So, in n= 2, you get 2s2 and 2p1.This means that the boron has three valence electrons.



Leave a Reply