What is ionization energy?
Suppose we have a neutral atom and the force of the nucleus is holding its electrons tightly. We want to remove the valance shell electron. So ionization energy is basically the amount of energy we needed to overcome the nuclear force of attraction.

Definition:
it is the smallest amount of energy absorbed by a gaseous atom to overcome nuclear forces to remove valance shell electrons. After the removal of the electron neutral atom become a positive ion.
Explanation :
We have a Magnesium atom in neutral form. We wish to remove the valance shell electron. As the nuclear forces on the electron hold up the electron. If we want to excite electrons. First, we have to overcome the nuclear force. To overcome nuclear forces, we provide the gaseous atom of Magnesium with energy. When an atom absorbs energy it becomes an unstable atom and excites an electron to attain stability back. So that minimum about of energy that can cause the removal of electrons is known as ionization energy. Ionization energy is also named ionization potential.
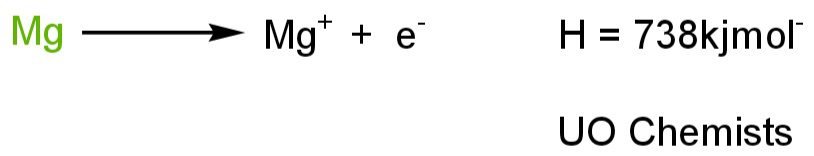
Factors that affect the ionization energy
Here are some factors which cause the increase or decrease in the ionization energy. These are factors on which ionization energy depends. Some factors are given below.
• Atomic Radius
• Nuclear Charge
• Shielding affect
• Nature of orbital
Atomic radius and ionization energy
Atomic radius is the distance from the nucleus to the outermost shell. As the atomic radius decreases ionization energy increases because of its small size more nuclear forces act on electrons and more energy is required to remove valance shell electrons. In the same way, if the atomic radius is large then the atom requires a small amount of energy to remove valance shell electrons. So atomic radius is inversely proportional to ionization energy.
Nuclear Charge and ionization energy
Nuclear charge is the force of attraction by the nucleus on its electrons. The nuclear charge is basically no protons present in the nucleus. It is directly proportional to the ionization energy. As the nuclear charge increases more tightly electrons hold by atoms and it requires more energy to remove valance shell electrons. In the same way, if the nuclear charge is low then it is easy to remove valance shell electrons with a little amount of energy.
Shielding effect and ionization energy
The shielding effect this is affecting the force of attraction of the nucleus by an inner shell on the valance shell. It is basically a screening or blocking of the force of attraction by inner shell electrons. In this case valance shell face less attractive forces of the nucleus. As the shielding effect is high it is easy to remove valance shell electrons so less amount of energy is required. When the shielding effect is low then more attractive forces hold electrons and more ionization energy is required to remove valance shell electrons. The shielding effect is inversely proportional to ionization energy.
Nature of orbitals and ionization energy
Another important factor that affects ionization energy is the nature of the orbital. Orbital from which electron has to remove that’s orbital which can affect the magnitude of energy required to do so.
Periodicity of ionization energy
Periodicity refers to any trend in the periodic table.
Ionization energy along periods
As we go with long periods in the periodic table from left to right. There is an increase in the number of protons or other words, there is an increase in the nuclear forces with which the size of atom decreases as the size of an atom decreases more attractive forces on valance electron. So more energy is required to remove valance electrons. As a result, as ve move with a period in the periodic table ionization energy increases.
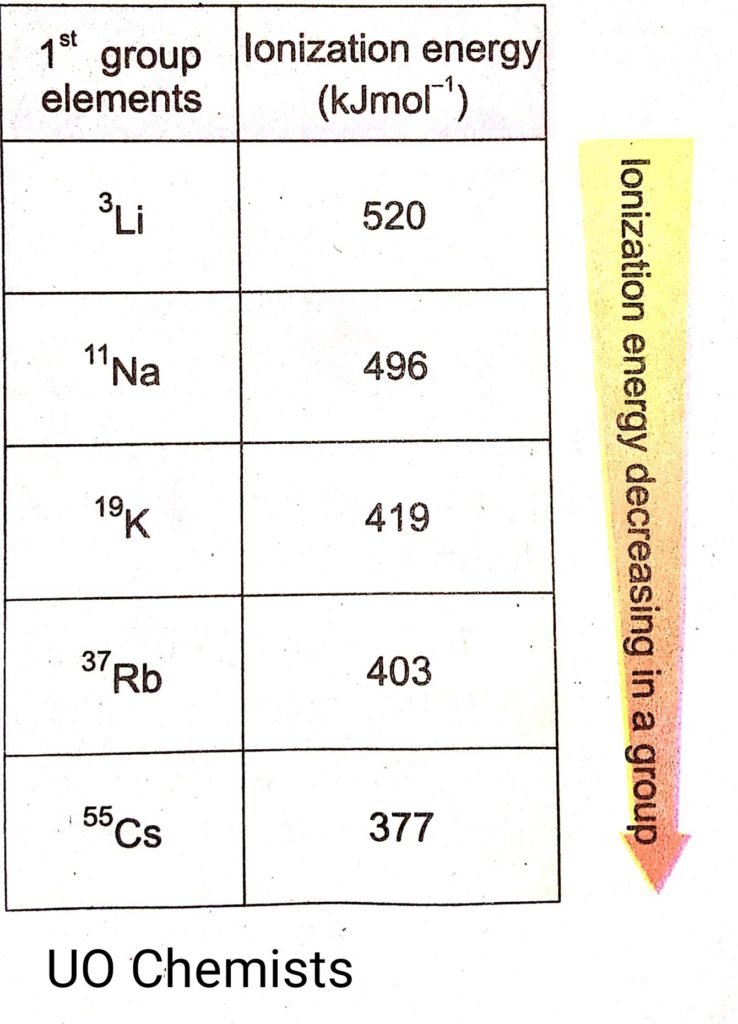
Ionization energy along with groups
In the periodic table as we look from top to bottom size of the atom increases because of the addition of the valance shell. Instead of an increase in nuclear charge ionization energy decreases from top to bottom. This is because of the shielding effect which increases with an increase in valance shell.

First ionization energy :
This the amount of energy absorb or required to remove electrons from neutral atoms.
Second ionization energy :
It is the amount of energy absorb or required to remove the electron from the positive ion.
Why second ionization energy is higher?
Second ionization energy is also greater than first ionization energy because when we provide a neutral atom with the energy it excites an electron and becomes a positive ion. In second ionization we need to remove the second electron from the positive ion which already has more attractive forces on the electron. So it is difficult to remove electrons from positive ions. To overcome strong nuclear forces more energy is required, that’s why second ionization energy is greater than first.
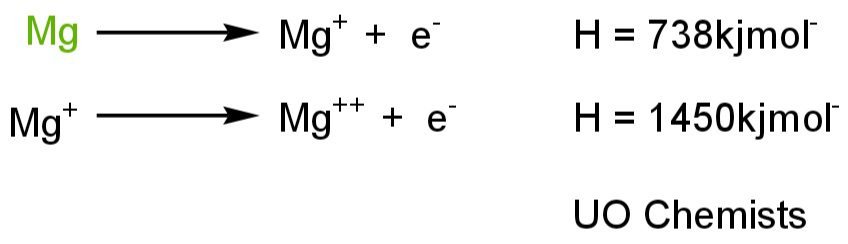



Leave a Reply