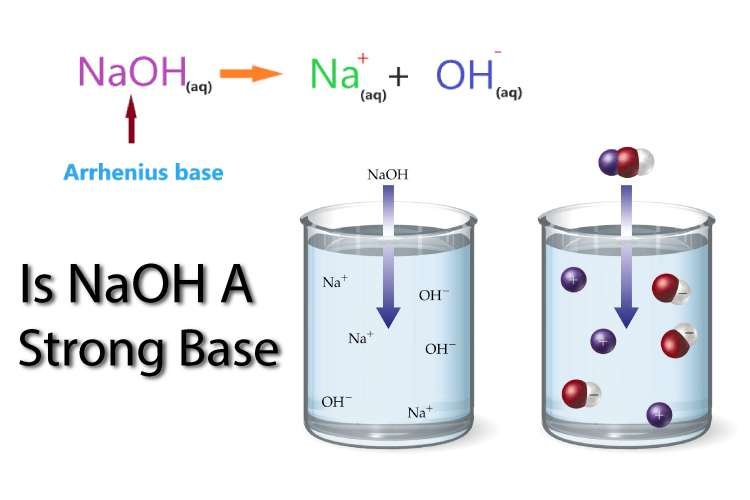
Before discussing is NaOH a strong base or not let’s have a look at basic properties or general information about caustic soda (NaOH). It is also called metal hydroxide or caustic soda. As it is an ionic compound, easily dissolves in water because it is the dielectric property of water to overcome the ionic bond and dissolve NaOH in water. We can also obtain caustic soda by the electrolysis of brine solution.
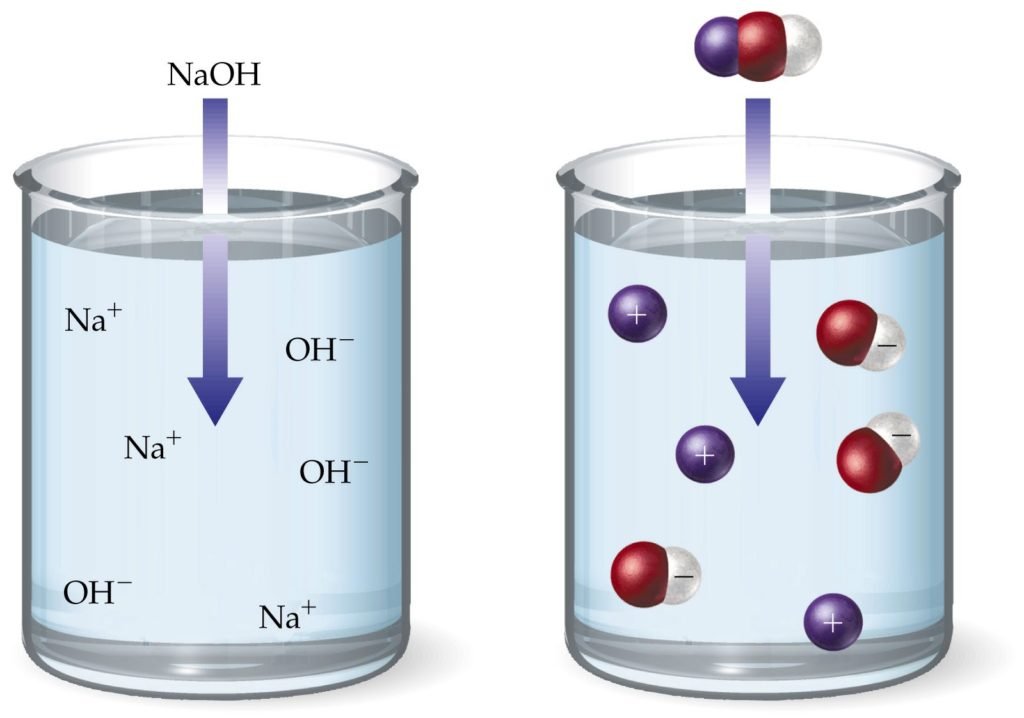
NaOH a strong base or not can easily be understood by knowing a basic difference between a strong and weak base. Strength of base can be categorized on the basis of dissociation in an aqueous medium & gaining of a proton or lone pair. The following table will show a great difference between a strong and weak base.
Difference b/w a Strong and weak base
| STRONG BASE | WEAK BASE |
| 1. A strong base is the base that split up completely 100 % into its ions in an aqueous medium. | 1. A weak base is a base that does not split up 100% completely into its ions in an aqueous medium. |
| 2. It has a pH of more than 7 but less than 14. | 2. It has a pH relatively close to 7. i.e, 8 or 7.9, etc. |
| 3. Strong bases have a higher value of Kb (Dissociation constant for bases) | 3. Weak bases have a lower value of Kb. |
| 4. No reversible reaction occurs due to complete ionization or dissociation. | 4. A reversible reaction occurs due to incomplete dissociation. |
Acid-Base concepts to show NaOH a strong base
There are following three valid concepts that can show you that Is NaOH a strong base or a weak base. These concepts are the Arrhenius concept, Lowry-Bronsted concept, and Lewis concept.
1. Arrhenius’s concept
This concept is only valid in an aqueous medium. So, if we talk about base. That base is said to be a strong base if it will give OH ions in water at dissociation. According to Arrhenius’s concept “THAT BASE WILL BE VERY STRONG BASE WHICH gives OH ion AT DISSOCIATION“. So, NaOH is a strong base because it will dissociate completely and also give OH ions.
NaOH + H2O → Na+ + OH–
So, this theory proves that NaOH is a strong base.
2. Lowry-Brosnted Concept
According to the Lowry-Bronsted concept, a strong base is a substance that can accept a proton. So, when NaOH reacts with the HCl, NaOH accepts a proton from HCl and behaves as a strong base.
NaOH is a strong base due to the presence of OH ions having lone pair. The proton is accepted by the OH ions and in return, OH also shares its electron with hydrogen so that both become stable.

3. Lewis Theory
G. N Lewis proposed a theory representing a brief difference between a strong acid and a strong base. A strong base is a specie that can donate a pair of electrons. When NaOH is dissociated OH ion is separated which means that it is an electron enriched specie.
So, OH in NaOH gives the pair of electrons to the other electron-deficient species so acts as a strong lewis base.
Conclusion
All the above theories explained proves that NaOH is considered a strong base. The strength of the base is determined by the degree of dissociation. More degree of dissociation results in a strong base. It also depends upon the gaining of a proton or the donation of an electron pair to any other electron-deficient specie.
What are the Applications of NaOH??
Sodium hydroxide has many applications:
- It is used to manufacture soap, paper, rayon as well as explosives.
- It is also used for laundering, bleaching, oxide coating, and metal cleaning purposes.
- It is used in electroplating techniques and electrolytic extraction.
- It is a highly versatile substance that can also be obtained as a by-product in the manufacturing of chlorine.
Why NaOH is not considered acid??
NaOH is not considered acid because it is a property of acids to dissociate a proton in an aqueous medium but NaOH did not possess any proton in it. So, it cannot release an H+ ion. Also, it will not satisfy the conditions of the above-explained theories.



Leave a Reply