This blog will provide you with a brief description of the Ionization constant of acids, and bases. Also, it will discuss the ionic product of water or autoionization of water and their relationship with the ionization constants of acid and base Ka, and Kb. Discussion of ionization constant also requires the discussion about acidic and basic strength. So, we will discuss first the difference between the strong and weak acid and base.
Difference between the strong and weak Acid-Base
We all know that there are many theories and concepts which discussed the nature of acid and base. But still, there is a very difficult situation occurred to discussing the strength of acid and base.
Strong acid and base are the species that ionize completely and furnishes H and OH ions. These have a fast rate of reaction due to a higher degree of dissociation. The higher degree of dissociation may also cause the acid or base stronger. Strong acids and bases have excellent conductivity because they completely dissociate into their ions. Conductivity may depend upon the ions (negative or positive). Those acids and bases are very strong that are polyprotic or polyhydroxy. They have the ability to donate more than one H or OH ion. For example, HCl, H2SO4, NaOH, Ca(OH)2.
Weak acid and base are the species that do not ionize completely into their ions. We will not say that it will never dissociate into ions, it will dissociate but follow the condition of equilibrium. Some ions are dissociated to attain equilibrium conditions so act as weak acids and bases. These are antagonistic to the strong acid and base. They have less degree of dissociation. They also possess a low rate of reaction. For example, Acetic acid CH3COOH.
Ionization Constant for Acids – Ka
Ionization constant Ka for acid is defined as:
“The constant when some of the reactants are converted into products of the weak acid and attain the condition of equilibrium is called Ka“.
We can find out Ka for only weak acid. A necessary condition for Ka is that reaction must follow equilibrium. Ka ionization constant for acid determines the acidic strength of an acid. The larger the value of Ka larger is the acidic strength.
Generally, weak acid has a larger Ka value and strong acid has a smaller ka value. The numerical value of Ka is zero for strong acid this is because after dissociation all the ions are converted into the products and there is no chance for a reaction to come back.
HCl → H+ + Cl–
But in weaker acids, there develops a condition of equilibrium. In that acid, some ions are converted into products but to attain equilibrium they must come back. This acid will not completely dissociate. So,
CH3COOH ⇌ CH3COO– + H+
Consider the following reaction of general acid:
H2O + HA ⇌ H3O+ + A–
A- is the conjugate base of te relative acid and HA is parent acid which is dissolved/ionized incompletely in water.
k = [H3O][A–] / [H2O][HA]
k[H2O] = [H3O][A–] / [HA]
Ka = [H3O][A–] / [HA]
This equation can also be written as:
pKa = -log10Ka = log10[HA] / [A–][H3O]
The following relation is derived from the equation:
Value of Ka ∝ Acidic strength ∝ [H+] in solution
Ionization constant for base – Kb
OH– ion releasing/dissociating bases are really very strong bases. The strength of basis character may depend upon the concentration of OH– ions. Dissociation of OH– ions may result in the strength of the base. More dissociation of hydroxyl ions more strong is base. The ionization constant for base Kb is more likely similar to the Ionization constant of acid Ka.
To determine theoretically the strength of the base we have to calculate its numerical value. The greater the value of Kb greater will be the strength of the base. A base is a species that can accept a pair of electrons. So, if we take an example of NH3 it is a base. The reaction of NH3 in water will furnish OH– ion. There are many exceptions related to Ammonia. Ammonia may act as an acid as well as a Base. So, it is important to know Is NH3 acid or base????
NH3 + H2O ⇌ NH4+ + OH–
Consider a following general reaction:
B– + H2O ⇌ BH+ + OH–
Kc = [BH][OH] / [H2O][B]
Kc[H2O] = [BH][OH] / [B]
Kb = [BH][OH] / [B]
This equation can also be written as:
pKb = -log10kb = log [B] / [BH][OH]
Value of kb ∝ Basic Strength ∝ [OH–] in the solution
Ionic Product of Water
The ionic product of water is also called Autoionization of water. Ionic product means the product of the concentration of hydronium ion and hydroxyl ion. Till 1884, it was proved that ions are responsible for the conductivity of electrolytes including water. So, at that time in 1884, S. Arrhenius write the expressions for the self-ionization of water. But, no condition is given on how the H+ ion is formed.
H2O ⇌ H+ + OH–
Then in 1923, Lowry and Bronsted prove that self-ionization occurs between two water molecules. So, then the explanation of the H+ ion occurs. Water molecule is dissociated into H+ and OH– ion. In this H ion having no electron can not exist in the solution so it will react with another water molecule to form the hydronium ion. So, it will give the expression:
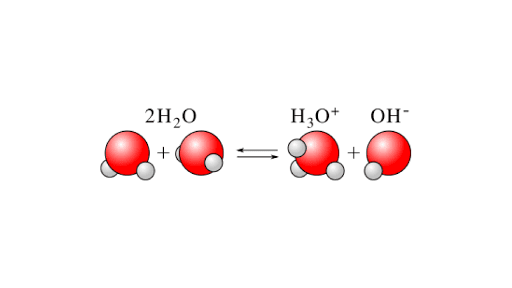
H2O + H2O ⇌ H3O+ + OH–
The animation shows how self-ionization occurs. A combination of two water molecules will form hydronium and hydroxyl ions. In this reaction, one of the water molecules will act as Lowry-Bronsted acid and another water molecule will act as Lowry-Bronsted Base.
Following is the derivation for the calculation of ionic product or self-ionization of water. Since water is a weak electrolyte its self-ionization may occur in this form:
H2O + H2O ⇌ H3O+ + OH–
k = [H3O][OH–] / [H2O]2
k[H2O]2 = [H3O][OH–]
kw = [H3O][OH–]
If a single water molecule may dissociate the equation for the ionic product of water may be written as:
kw = [H+][OH–] at 25℃
- The value of kw is independent of the amount of water.
- But the value of kw depends upon temperature.
- Simply if we talk about the value of kw it must be 1.00 × 10-14.
kw = [10-7][10-7] = 10-14 At 25℃
In this equation at 25℃, the concentration of H and OH is the same, i.e. 10-7. This value 10-14 is not universal it changes with the change in temperature. It is directly proportional to the change in temperature. An increase in the change of temperature causes an increase in the value of kw. If the temperature increases:
- At 63℃, the value of kw is 10-13.
- At 11℃, the value of kw is 0.3×10-14.
Relationship of ka, kb, & kw
Consider the following reactions for ionization constant ka:
CH3COOH ⇌ CH3COO– + H+
Consider the following reaction for ionization constant kb:
CH3COO– + H2O ⇌ CH3COOH + OH–
To calculate redox reaction:
CH3COOH ⇌ CH3COO– + H+
CH3COO– + H2O ⇌ CH3COOH + OH–
So, we cancel out the same elements or constituents of a reaction. Then only one product is obtained that’s water. This water is dissociated to form H and OH ions:
H2O ⇌ H+ + OH–
Since kw is an ionic product of water. So, it is equal to ionization constant ka & kb. Ka occurs due to the presence of proton but Kb occurs due to the presence of hydroxyl ions. So, the equation becomes:
Kw = Ka * Kb
Relation in terms of putting values of ka & kb
If we introduce the value of ka and kb we can calculate the relationship of ionic product of water with ka and kb very effectively. Since,
Ka = [CH3COO–][H+] / [CH3COOH]
Kb = [CH3COOH][OH–] / [CH3COO–]
By using the relation: Kw = Ka * Kb
Kw = ([CH3COO–][H+]/[CH3COOH])([CH3COOH][OH–]/[CH3COO–])
Kw = (H)(OH)
Kw = Ka * Kb
Relationship of Kw with pH & pOH
pH and pOH are the potentials of Hydrogen and hydroxyl ions. This term is given by Soren Sorenson. To calculate the acidity or bascity of a solution we can calculate its pH or pOH. pH and pOH are relative terms.
pH = -log [H+] & pOH = -log [OH–]
pkw = -log kw & pka = -log ka & pkb = -log kb
pKw = pKa × pKb
pKa = -log ka = -log [H+] = pH
pkb = -log kb = -log [OH–] = pOH
pKw = pH + pOH
Now we will calculate, how at 25℃ value of pKw = 1.00 × 10-14
pkw = pH + pOH
pKw = (-log [ 1.00 × 10-7]) + (-log [ 1.00 × 10-7])
pKw = 7 + 7
pkw = 14
So, a conclusion is drawn:
- pKw = 7 {Neutral solution}
- pKw > 7 {Acidic solution}
- pkw < 7 {Basic solution}
Dependence of Kw on Temperature
The ionic product of water is directly proportional to the change in temperature. As water is a weak electrolyte, so reversible reaction occurs. An increase in temperature favors the reaction to that side from where equilibrium is maintained. So, it works on the LE-CHATLIER’S PRINCIPLE.
Numerical Problems
Find the concentration of [H] and [OH] ions for the solution of a weak acid HA whose ka = 2 × 108 and 8 moles of acid are present in 1 liter of solution????
HA ⇌ H + A–
8 ⇌ 0 + 0
8-8∝ ⇌ 8∝ 8∝
Ka = [H+][A–] / [HA]
Ka = 8∝2 / 1 – ∝
Ka = 8∝2
∝ = 0.5 * 10-4
So, to calculate H+ and OH- we use formula:
[H+] = 8∝
[H+] = 8 (0.5 * 10-4)
[H+] = 4 * 10-4 mol/L
As we very well know the following relation:
kw = [H+][OH–]
10-14 = 4 * 10-4 [OH–]
[OH–] = 0.25 × 10-10 mol/L
Calculate the pH of HCl having 10-8 M in solution. Also calculate concentration of OH ions present in water at 25 degrees.
pH = -log H
pH = -log 10-8
pH = 8
Since, at 25 degree celsius value of kw becomes 10-14 so by using formula:
pKw = pH + pOH
14 = 8 + pOH
pOH = 6
TEST YOURSELF:::::::::::
If a weak base M(OH)2 has kb = 1.6 * 10-8 at 298 K. Find the concentration of OH and Hydronium ion in a solution having 2M concentration of base ???? (0.25 * 10-11)
Hence, it is concluded that the ionic product of water may depend upon ionization constant ka and kb. And also it depends upon pH and pOH. This determines the acidity and basicity of the solution.



Leave a Reply