
To understand the concepts of ligands, first, you need to know about the introduction of coordination compounds. In this context, we discussed what are ligands? Types of ligands on the basis of denticity and on the basis of the behavior of ligands.
Coordination Compounds
An addition or molecular compound that results from the combination of two or more simple salts or molecules and retains their identity in solid as well as the aqueous state. Those compounds which contain complex molecules or complexions capable of independent existence are called coordination compounds or Complex compounds. The formation of coordination compounds is one of the properties of these d block elements.
- When we add a solution of ammonia to a solution of copper sulphate, a dark blue colour due to the formation of [Cu(NH3)4]2+ appears.
- When we add Ammonia solution to a precipitate of silver chloride, the precipitate dissolves due to the formation of [Ag(NH3)2]Cl.
- When we add Ammonia to a solution of nickel chloride, a violet colour due to the formation of is [Ni(NH3)6]2+ obtained.
These are all examples of coordination compounds. Thus, [Cu(NH3)4]2+, [Ag(NH3)2]Cl, and [Ni(NH3)6]2+ are examples of coordination compounds. These are also known as Complex compounds. Transition elements have a tendency to form coordination compounds. The species in the square brackets remain as a single entity. They do not ionize into its constituent when it is dissolved in water.
Importance of Coordination or Complex Compounds
Complex compounds are widely present in minerals, plants, and animals. They play important functions. Hemoglobin and chlorophyll are coordination compounds of iron and magnesium respectively as Central metal atoms. Coordination compounds find wide applications in electroplating, textile Dyeing, and analysis of trace elements.
Difference Between Double Salts and Coordination Compounds
Those compounds which lose their identity in solution are called double salts. when FeSO4. (NH4)SO4. 6H2O dissolved in water the parts of molecules are separated from each other. The compounds are dissociated in water to give simple ions.
FeSO4. (NH4)SO4. 6H2O → Fe2+ + 2NH4+ + 2SO42- + 6H2O
KCl. MgCl2. 6H2O (Carnalite), K2SO4. Al2(SO4)3. 24H2O (Alum) are examples of double salts.
Those compounds that do not lose their identity in solution are called coordination or Complex compounds.
K4[Fe(CN)6] → 4K+ + [Fe(CN)6]4-
[Fe(CN)6]4- is a complexion that does not give the test of Fe2+ ion. [Cu(NH3)4]SO4, K[Ag(CN)2] are examples of complex compounds.
Types of Coordination Compounds
- On the basis of ions
- On the basis of ligands
On the basis of Ions
As we know that any molecule or salt or compound contains both the cationic part and anionic part. For example, NaCl contains Na+ cation and Cl– anion. H2O contains H+ cation and OH– anion etc. So, coordination compounds are also mixed salt and contain cation and anion.
- Simple Cation Complex anion
- Complex cation Simple anion
- Complex cation Complex anion
- Neutral molecule
Simple Cation Complex anion
K4[Fe(CN)6] → 4K+ + [Fe(CN)6]4-
Complex cation Simple anion
[Cu(NH3)4]SO4 → [Cu(NH3)4]2+ + SO42-
Complex cation Complex anion
[Pt(NH3)4][PtCl4] → [Pt(NH3)4]2+ + [PtCl4]2-
Neutral Molecule
[Ni(Co)4], [Ni(DMG)2]
Important Terms in Coordination Compound
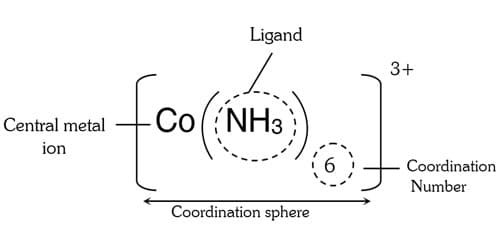
In order to understand the structure and nature of complex compounds, we should learn the basic terms:
Central Metal Atom
It is a metal that accepts lone pairs from ligands to form a co-ordinate bond. Usually, the center metal atom is a d-block element due to the high nuclear charge and availability of vacant d-orbital. Nuclear charge means the number of protons present in the nucleus.
Ligand
The atom or group of atoms that donates lone pair to a central metal atom in order to form a co-ordinate bond is called a ligand or A neutral molecule or an ion attached to a central metal atom in a coordination compound is called ligand. The neutral molecules such as ammonia, water, ethylenediamine, and ions such as Cl–, NO2-, SCN, etc.
Coordination Number
It is the number of ligands that are directly attached to the central metal atom. The coordination number varies from 1 to 12 but more common are 4 and 6. The coordination number of silver in [Ag(NH3)2]+ is 2. The coordination number of platinum in [Pt(Cl)6]2- is 6.
On the basis of Ligands
- Anionic ligands
- Cationic ligands
- Neutral ligands
Anionic Ligands
Anionic ligands carry a negative charge. Anions whose name ends with “ide” are named by replacing “e” with “o”.
| I– | Iodido |
| CN– | Cyanido |
| S2- | Sulphido |
| O2- | Oxido |
| OH– | Hydroxido |
Cationic Ligands
Cationic ligands carry a positive charge. The name ends with “ium”
| NO+ | Nitrosylium |
| NH2NH3+ | Hydozinium |
Neutral Ligands
| H2O | Aqua |
| NH3 | Ammine |
| CO | Carbonyl |
| NO | Nitrosyl |
| CS | Thiocarbonyl |
| P4 | Tetraphosphorous |
| O2 | Dioxygen |
| S8 | Octasulfur |
Types of Liagands on the Basis of Denticity
If the ligand is attached to the central metal atom only by one coordinate bond, then the ligand is called a monodentate or unidentate ligand. For example, unidentate ligands are Cl–, NO2–, Br–, CN–, OH– H2O, NH3, CO, etc.
If ligands having two or more donor atoms are called polydentate ligands. They are further classified as bidentate (two donor atoms), tridentate (three donor atoms), tetradentate (three donor atoms) and those with five and six atoms are called Penta and hexadentate ligands respectively.
For example, In [Fe(CN)6]4-, CN is a monodentate ligand. In ethylenediamine, (NH2-CH2-CH2-NH2), a neutral ligand, is a bidentate ligand in ethylenediamine copper complexion. CO32-, SO42-, (COO)2- are bidentate ligands with a double negative charge.
Briefly, we can say that monodentate, bidentate, tridentate, tetradentate, pentadentate, hexadentate have one, two, three, five, and six donor sites respectively.
Monodentate Ligands
Those ligands have a tendency to donate only one lone pair.
| F– | Floro | Florido |
| NH3 | Ammonia | Ammine |
| H2O | Water | Aqua |
| O2- | Oxide | Oxido |
There is a difference between denticity and its classification monodentate, bidentate, etc.
Denticity
Denticity means total available sites or lone pair on ligands. On the other hand, monodentate, bidentate, etc mean total lone pair or sites donate from total available sites. For example, In water molecules, oxygen has 2 lone pairs so the denticity of H2O is two but it donates only one lone pair so, it is a monodentate ligand.
Bidentate Ligands
Those ligands have a tendency to donate two lone pairs. Oxalato (OX), ethylenediamine (en), glycinate ion (Gly), Dimethyl glyoximate (DMG) are bidentate ligands.
1. Oxalato C2O42-
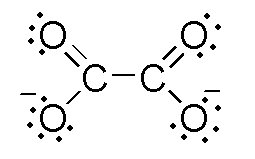
The denticity of oxalato is 10 because it has a total of ten lone pairs to donate but actually, it donates only two lone pairs so oxalato is bidentate.
2. Ethylene diamine
Ethylenediamine has two Donor sites to donate the lone pairs so, it is a bidentate ligand.

3. Glycinate Ion
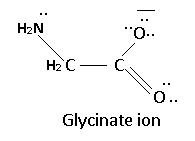
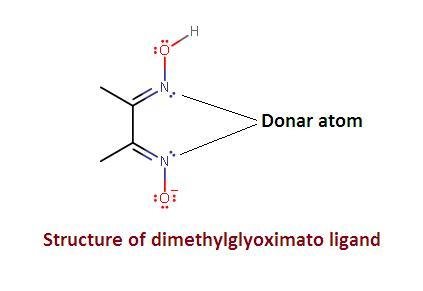
4. DMG
DMG stands for dimethyl glyoximate ion.
Note:
In a salt, are all the ligands are the same? The answer is may or may not. Basically, there are two types homoleptic and heteroleptic ligands. Those compounds in which all ligands are the same are called homoleptic ligands. Those compounds in which all ligands are not the same are called heteroleptic ligands. The example of homoleptic and heteroleptic ligands are K4[Fe(CN)6] and [Cu(H2O)2(NH3)4]SO4 respectively.
Tridentate Ligands
NH2-CH2-CH2-NH2-CH2-CH2-NH2
It is diethlene triamine (diene).
Tetradentate ligands
NH2-CH2-CH2-NH2-CH2-CH2-NH2-CH2-CH2-NH2
It is triethlene tetramine (triene).
Pentadentate Ligands
NH2-CH2-CH2-NH2-CH2-CH2-NH2-CH2-CH2-NH2-CH2-CH2-NH2
It is tetraethlene pentamine.
Ethylene diammine triaccetato ion is a pentadentate ligand.
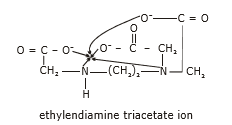
Hexadentate Ligands
EDTA stands for ethylene diamine tetraacetate is a hexadentate ligand.
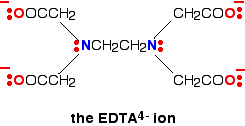
After discussing the type of ligands on the basis of denticity, now we discussed the types of ligands on the basis of the behavior of Ligands.
Types of Ligands on the Basis of Behavior of Ligands
There are three types of ligands:
- Flexidentate ligand
- Ambidentate ligand
- Chealting ligand
Flexidntate Ligands
The ligands which do not necessarily use all their donor sites to form co-ordinate bond is called flexidentate ligands. In EDTA, there are six donor sides possible but sometimes it behaves as tridentate, sometimes tetradentate, and sometimes it is hexadentate.
Example
- [Cr(OH)(EDTA)]+3
In this compound, EDTA acts as a pentadentate ligand. Cr is in a +3 oxidation state means that it forms a six-coordinate bond. In compound [Cr(OH)(EDTA)]+3 Cr form one bond with OH and five bonds with EDTA. So, EDTA acts as a pentadentate ligand.
- [Pd H2 (EDTA)]+2
In this compound, EDTA acts as a tetradentate ligand. Pd is in a +3 oxidation state means that it forms a six-coordinate bond. In compound [Pd H2 (EDTA)]+2 Pd forms two bonds with OH and four bonds with EDTA. So, EDTA acts as a tetradentate ligand.
Similarly, SO42- acts as both monodentate and bidentate ligand.
- [Co+3 (en)2 SO4]
In this compound, Cobalt is in a +3 Oxidation State. Cobalt in +3 Oxidation State forms 6 co-ordinates covalent bonds. As we know that ethylenediamine is bidentate means it donates two lone pairs and forms two coordinates bond. In the above compound, two ethylenediamine means four co-ordinate covalent bonds so, Cobalt forms four covalent bonds with ethylenediamine and the remaining two bonds with SO42- so, SO42- acts as a bidentate ligand.
- [Co+3 (NH3)5 SO4]
Cobalt forms 5 bonds with ammonia and one bond with sulfate ion so sulfate ion acts as a monodentate ligand.
As SO42- act as both monodentate and bidentate but when it acts as bidentate, a four-membered ring is formed which is unstable (remember that only 5 and 6 membered ring is stable) SO42- want to become bidentate but it is not due to instability. So, SO42- act as a monodentate ligand.
SO42- Prefers to act as Monodentate Ligand
In the above structure, sulfate is acting as the bidentate and a four-membered ring is formed which is unstable.
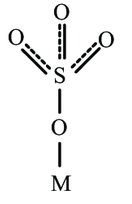
In the above structure, sulfate is acting as the unidentate which is stable. we conclude that the SO42- prefers to act as a monodentate ligand.
Ambi-dentate Ligands
Ligands that have more than one type of donor atoms are called Ambi-dentate ligands. For example, CN–, NO2–, SCN–,etc. In CN–, both the carbon and nitrogen can donate pair of electrons.
Chelating Ligands
The Legend which after donating electrons to the central metal atom forms a ring is called a chelating ligand. Remember that the ring is only 5 and 6 membered. After the formation of the chelate ring, the complex becomes more stable.
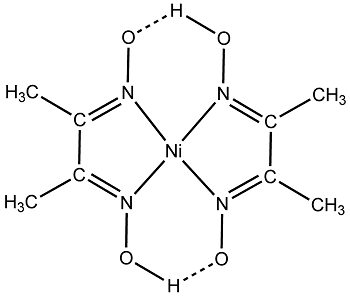
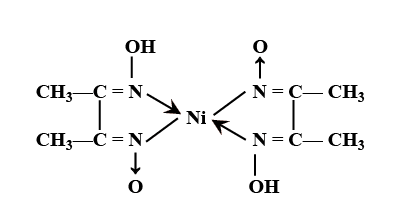
Chelates
These are special types of coordination compounds containing one or more rings in their structures. The ring structures are formed by linking metal ions with polydentate ligands such ring structure compounds are called chelates. The polydentate ligands are called chelating ligands and the process of formation is called chelation. For example, ethylenediamine, a bidentate ligand, forms a complex with copper ion, this compound contains two 5 membered rings and this compound is called a chelate. Another example of chelation is Nickel-DMG. This complex is formed when Nickel ion (Ni+2) reacts with dimethylglyoxime, DMG.
EDTA is a hexadentate ligand that is widely used for chelating metal ions.
Factor Affecting Stability of Chelates
The stability of chelates depend upon the following factors:
1. Chelate Effect
The complexes formed by polydentate ligands such as ethylenediamine (en), EDTA, etc are more stable than those formed by monodentate ligands such as water, H2O and ammine, NH3. This enhanced stability of complexes containing chelating ligands is called the chelate effect. For example, the nickel complex with chelating ligand, (en) is more stable than the corresponding complex with NH3 ligand.
Ni2+ + 6NH3 → [Ni(NH)3]2+ Less stable
Ni2+ + 3en → [Ni(en)3]2+ More stable
2. Number of Chelate Rings
The greater the number of chelate rings in a complex, the greater is the stability of the complex.
3. Size of Chelate Ring
It has been observed that the five-membered ring is more stable. Chelates having six-membered rings are slightly less stable. Chelating ligands such as ethylenediamine, oxalate ion, glycine, and DMG form a quite stable five-membered ring. It may be noticed that those five-membered chelates are more stable when atoms in the ring are joined by a single bond however six-membered chelates having a double bond in the ring are more stable than corresponding five-membered chelates.
Application of Chelate Compound
- Biological importance of chelates are:
- Vitamin B12, essential for human body, is a Cobalt complex.
- Homeoglobin in red blood cells contain porphyrin Complex and Fe2+ ion is central metal ion.
- Chlorophyll is a green plant pigment containing magnesium for porphyrin complex.
- Metal poisoning by lead copper chromium, Nickel, zinc, etc in the body can be removed by treating with EDTA. It forms a metal EDTA which is easily excreted in the urine.
2. In analytical chemistry, chelates are used in qualitative and quantitative analysis. For example, Ni2+, Mg2+, Cu2+, etc are quantitatively precipitated by the chelating agents as insoluble compounds.
3. Standard EDTA a hexadentate ligand solution is widely used in volumetric analysis to determine the hardness of the water.



Leave a Reply