
Nickel is a metal that is the d-block element as well as a transition metal. It is the d-block element because its electronic configuration contains a d-orbital. It is a unique element that is silvery-white lustrous metal. It has the symbol Ni and atomic mass 28. It is a hard, brittle, and ductile metal that is converted into hard wires, pipes as well as coins. It is also present in the powdered form because it reduces the surface area but large pieces of nickel are slow to react because an oxide layer covers it so that it will be reduced or prevented corrosion and rusting. It has a standard atomic weight of 58.69 amu. Nickel is basically a ferromagnetic material that is hard, and tough than iron.
Earth’s outer and inner core is composed of an iron-nickel mixture. Nickel is slowly oxidized means it has the ability to lose electrons. It is an electropositive metal by air at room temperature. In the early ages, nickel is basically used in plating, coating chemistry equipment & manufacturing alloys.
Nickel metal is valuable in modern times in alloys: about 68% of world production is used in stainless steel. Nickel contains a different number of niches such as a catalyst for hydrogenation, and a cathode for batteries. Nickel is an essential nutrient for some microorganisms & plants. It is also used to convert Ghee into oil.
Discovery & History of Nickel:
Intentional uses of nickel are commonly ancient that can be traced to 3500 BCE. it is observed that 2% of nickel is part of the bronze medal. In ancient Chinese manuscripts, white copper known as cupronickel is discovered in 1700 and 1400 BCE. Some coins of nickel alloy were mined by Agathocles and Pantaleon in the 2nd century BCE. Different meteorites contain a mixture of iron and nickel. It was named as native of Pew as a kind of silver-Zinc alloy is called Pai-T’aing used in China as long as 200 BCE.
In 1751, Axal Fredrick was working in Stockholm where he mined a metal. He called it nickelins (NiAs). Nickelins came when mining was done in Los Angeles, Sweden. He thought that it should contain copper but when he extracted he saw that a new metal arose. So, he called it nickel in 1754.
Different chemists of different countries thought that it was an alloy of cobalt, arsenic, copper, and iron but until 1775, pure nickel was produced by Torbern Bergman confirmed its elemental nature. Earlier an ore of the same type was called Kaperfernickel after “Old nickel” and his mischievous gnomes because it resembles copper ore.
Periodicity & Physical properties:
Each element in the periodic table may possess different trends in periodic properties as well as physical properties. Changes in the periodic am physical properties are due to:
- Strong intermolecular forces.
- Increase nuclear attraction.
- Change in the size of an atom.
| PROPERTIES | NICKEL ELEMENT |
| 1. State at STP | Solid |
| 2. Electronic configuration | [Ar]3d8, 4s2 |
| 3. Melting point | 1455°C. 2615 F |
| 4. Boiling point | 2913°C, 5275 F |
| 5. Density | 8.90g/cm3 |
| 6. Heat of fusion | 17.48 KJ/mol |
| 7. Heat of Vaporization | 379 KJ/mol |
| 8. Molar heat capacity | 26.07 KJ/mol |
| 9. Youngs modulus | 180 GPa |
| 10. Shear modulus | 200 GPa |
| 11. Poisson ration | 0.31 |
| 12. Crystal structure | Face centered cubic |
| 13. Electrical resistivity | 69.3nΩ |
| 14. Thermal conductivity | 90.9 W/mK |
Changed electronic configuration:
The electronic configuration of Ni with atomic number, 28 is somehow different because in this the d-orbital is involved which is a higher energy orbit. The electron should fill first in the lower energy orbit and then in the higher energy orbit. It is due to stability. If we discuss the stability of the orbital then it is noticeable that a partially filled orbital is more stable than that of a half-filled orbital. So, to attain stability and symmetry electrons should fill first in the s-orbital and then in the d-orbital. Hence, the configuration of Ni becomes:
Nickel = Ni = 28 = 1s2, 2s2, 2p6, 3s2, 3p6, 3d8, 4s2
Now, we will discuss the periodic properties of Nickel. Nickel belongs to group number 10 and 4th period. So, its periodic properties vary in groups as well as periods:
- Its oxidation states are -2, -1, 0, +1, +2, +3, +4
- 1st Ionization energy of nickel is 737.1 KJ/mol & 2nd the ionization energy is 1753.0 KJ/mol. The increase in ionization energy is due to a decrease in the size of an atom and an increasing nuclear charge.
- The electronegativity of nickel is 1.91 (According to Pauling scale)
- Its atomic radius is 124 pm.
- Van der waals radius of nickel atom is 163 pm.
Chemical Properties :
It is important to explain the chemical properties with the explanation of physical properties. In the chemical properties of nickel, we will discuss in detail the chemical reactivity of the Nickel. Nickel may react with oxygen, halogens, dilute sulphuric acid and also act as a catalyst.
- Nickel does not react with water under normal circumstances.
- Nickel reacts with air in ambient conditions. The reaction between air and nickel appears to proceed to the product nickel (II) oxides:
2NiO + O2 → 2NiO
- Nickel reacts with halogens except for fluorine because fluorine does not react with nickel due to its small size and electronegative atom. Nickel reacts with Cl2, I2, and Br2 to form nickel (II) dihalides:
Ni + Cl2 → NiCl2
Ni + Br2 → NiBr2
Ni + I2 → NiI2
- It also reacts with a dilute acid such as dilute sulphuric acid, to form a solution containing aquated Nickel (II) ions together with hydrogen gas:
Ni + H2SO4(aq) → Ni2+ + SO42- + H2
- Nickel does not react with bases i.e. Strong or weak.
- It acts as an active catalyst in the conversion of vegetable ghee into vegetable oil. Hydrogenation of alkene is done by using nickel catalyst in presence of 250°C to 300°C temperature.
CH2 = CH2 + H2 → CH3 – CH3
Occrance:
Nickel constitutes about 0.02% of the earth’s crust. Metallic nickel is formed only in some meteorites. Cheif ores of nickel are: Pentlandite(Ni, Cu, Fe)S, nickel glance (NiAsS), Garnierite (Ni, Mg)SiO3.xH2O, Smaltite (Ni, Co, Fe)As2, Niccolite, and White nickel ore. It is also found in the form of arsenide ores that are found in Norway, Canada, Spain, and the USA.
Nickel is considered to be the fifth most abundant element on the earth. In the earth’s inner core it is the second (2nd) abundant metal. Norway used nickel in smelting sites. So, in Norway, it is abundant and acts as a catalyst called Pyrrohotite. Large deposits of nickel occur due to ancient meteor impacts. It was also discovered in Russia & South Africa in the early 1900s. In Canada, about 30% of Nickel is obtained. Indonesia and Australia have 43.6% of the world’s today. Nickel basically occurred in two types of deposits;
- Laterite Deposits: Results of intensive weathering of surface nickel-rich rocks.
- Magnetic Sulfide Deposits:
Nickel also occurs in the environment. Windblown dust, volcanic eruptions, vegetation forests fires & meteoric dust contain very harmful Nickel particles. Different anthropogenic activities such as mining, refining, roasting, smelting, bassmerization, combustion, and incineration rush out Nickel as harmful material in the atmosphere. Nickel is widely available in the soil and an essential nutrient for photosynthesis.
The emission of nickel from the estimated source is about 8.5 million per year, in the early 1990s. The general population is exposed to low levels of nickel in ambient air, water, and food through tobacco consumption/combustion.
Compounds:
Most commonly nickel exists in the oxidation state of +2 but those compounds are introduced that have oxidation states 0, +1, +3 & also these compounds are introduced that have an exotic oxidation state of -2, -1 & -4. Following are the compounds of nickel:
1. Nickel (0):
Nickel (0) is called tetracarbonyl nickel. It was discovered by Ludwing Mod. He discovered it when he reacted four (4) moles of carbon monoxide with nickel resulting in tetracarbonyl nickel (Nickel (0)). Basically, Nickel (0) is used as an active catalyst in organonickel chemistry to prepare the organic compounds in which nickel is involved.
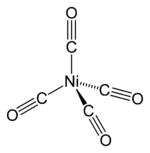
The reaction by which it is formed is:
4CO + Ni → Ni (CO)4
This compound has tetrahedral geometry. Nickel forms 4 bonds with carbon and each carbon form a triple covalent bond with carbon by which electrons are mutually shared. In this structure, the bonds between CO are 2 but according to Coulson’s treatment of CO, it possesses a 2.5 bond order that means a positive or negative charge may shift either on carbon or on oxygen.
2. Nickel (I):
Nickel (I) is basically a complex having tetrahedral geometry. It possesses Ni-Ni bonding and by reduction of K2[Ni2(CN)6] with sodium amalgam dark red diamagnetic K4[Ni2(CN)6] is obtained by the liberation of H2 gas. Nickel-containing enzymes such as [NiFe]-Hydrogenase is important to Nickel (I) which involves the catalyzes of reversible proton H2.

This diagram is showing the structure of nickel I. It contains two nickel atoms that are single-bonded to each other and also cyanide CN is placed on both adjacent Nickel atoms. It is a complex having an oxidation state of -4.
3. Nickel (II):
Nickel (II) compound of nickel forms many complexes with sulfates, sulfides, carbonates as well as hydroxides. The formation of Nickel (II) with other compounds is given below:
- By the addition of Nickel (Ni) in Sulphuric acid (H2SO4), the Nickel (II) sulfates are produced. This is also produced by using Ni – oxides instead of using Ni – metal.
H2SO4 + Ni → NiSO4 + H2
- Different meta aqua complexes are formed when common salt of nickel is dissolved in water (aqueous medium) to form meta aqua complexes.
- Nickel (II) chlorides are most illustrative of other halides. These are produced when Nickel-metal or its oxide are reacted chemically with HCl to give a green hexahydrate solution.
Ni + 2HCl → NiCl2 + H2
4. Nickel Hydroxides & oxides:
Ni-OH is used as a catalyst intermediate and in the manufacturing of Nickel – Cadmium (Ni-Cd) Batteries. Nickel may also react with oxides and hydroxides to form the following compounds:

Green nickel oxides are a relatively pure form of nickel monoxide. It is produced by firing a mixture of nickel powder and water in the air at 1000 °C. This is also used for the manufacturing of nickel catalysts.
Ni (mixture of nickel powder) + 2H2O → Ni(OH)2 + H2

Black nickel oxides are precursors to nickel salt. They have the same level of high chemical purity and have a similar high surface area. It contains 76.6% to 77.9% nickel content. Black nickel oxides are basically pure nickel monoxide. It is manufactured by the calcination of nickel hydroxycarbonate or nickel nitrate at 600°C. Nickel trioxide is also called Black Nickle oxides.
5. Nickel Salts:
Following are the nickel salts:
- Nickel acetate is used in electroplating and as a dye mordant.
- Nickel ammonium sulfates are used as metal finishing composition.
- Nickel chloride is used in absorbing towers of industrial gas masks where its absorbs ammonia.
Isotopes:
Basically, there are 18 isotopes of nickel. But nickel possesses only five stable isotopes:
| Isotopes | Relative Abundance | Applications & Uses |
| 1. 58Ni | 62.27% | It is used to study human absorption of Nickel |
| 2. 60Ni | 26.01% | It is used to study bone densitometry, gamma camera preference cell |
| 3. 61Ni | 1.13% | It is used in PET |
| 4. 62Ni | 3.59% | It is used in electron capture source in Gas Chromatography |
| 5. 64Ni | 0.91% | It is used in Radiomunrotherapy |
Extraction of Nickel from different Pentlandite Ore:
Extraction is basically the procedure by which a pure ore is extracted and purified by the different intermingled ores. So, in this heading, we will cover how nickel is extracted from the different ores.
1. Concentration of ore:
First of all the impure and insoluble materials are separated out on the basis of their weight. The ore is ground and crushed in a crusher and then enters the Froth Floatation Process. The palm oil is introduced into the floatation chamber and then the heavy objects which are insoluble settle downward and remain floating on the surface of the water. The useful components are driven out and then roasted and smelted to obtain the pure and concentrated nickel in the form of NiS (Nickel Sulfide).

2. Roasting:
No, the concentrated ore obtained is roasted in the excess supply of oxygen in the blast furnaces. It requires approximately 8-10 weeks for the roasting. Due to roasting some of the conclusions are drawn:
- Excess of the free sulfur is oxidized to SO2.
- Most of the FeS is oxidized to the FeO. some FeS remains unaffected.
- Moisture is removed.
During roasting, NiS and CuS remain unaffected and the roasted mass thus obtained NiS, CuS, and a little free sulfur and FeS. The roasting equipment is trying to maintain the heat evolution during the burning of sulfur.
3. Melting/Smelting:
The roasted mass containing NiS, CuS, and a little free sulfur and FeS is mixed with coke and fluxing material (silica, SiO2, limestone, and calcium carbonate) & then it is smelted at high heat in the smelting furnace so that the remaining FeS and free sulfur are oxidized to FeO and SO2 respectively. FeO is produced during roasting and smelting combined with silica to form ferrous silicate. FeSiO3.
FeO + SiO2 → FeSiO3
At the same time, CaO obtained from the decomposition of CaCO3 also combines with silica to form calcium silicate, CaSiO3.
CaO + SiO3 → CaSiO3
Both the iron silicate and calcium silicate are fusible slag which is not a required product. Due to their lighter weight, both float on the molten mass that consists of NiS, CuS, and little free sulfur.

4. Basemerization:
The molten mass obtained at the end of the smelting operation is mixed with silica and then introduced into a Bessemer-type converter with a basic lining and fitted with tuyers to admit hot air under pressure. A blast of hot air is admitted into the converter under pressure through the layers. The hot air is oxidised almost. All the remaining sulfur and FeS are oxidized to SO2 and FeO respectively.
FeO combines with SiO2 to form a slag of FeSiO3. This slag is continuously removed. The resulting mass obtained at this stage is called besemerised NiS-CuS matte which consists almost entirely of the sulfides of Ni and Cu. The composition of matte is as: Ni = 55%, Cu = 25 – 30%, S = 14 – 17% & Fe = 0.1 – 0.5%.
5. Recovery/Refining of nickel:
The process of recovering nickel metal from the besemesrised NiS-CuS matte obtained above is termed as refining of nickel or matte. Refining of Ni is generally done by Mond’s Process in which matte is again roasted in the presence of excess O2. So, NiS and CuS are converted into NiO, and CuO is treated with dilute H2SO4 at 80 °C which converts CuO into soluble CuSO4 while NiO remains unaffected.
The residue of NiO is treated with water gas (CO + H2) when NiO is reduced to crude nickel. This crude nickel is then treated with CO so that Ni is converted into the vapors of nickel tetracarbonyl, Ni(CO)4 which on being heated to 180 °C decomposes into 99.9% pure nickel.
2NiO + H2 + CO → 2Ni + H2O + CO2
Ni(CO)4 → Ni (pure) + 4CO↑
World Production:
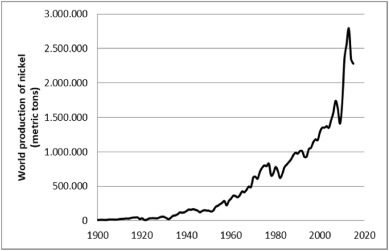
Nickel is most commonly used to manufacture high-grade steel & different Ni-Cd batteries. In 2020, it was estimated to be approximately 2.5 million metric tons. Indonesia, the Philippines, Russia & New Caledonia are the basic and major countries that mine nickel in higher quantities. Global mine production of nickel is 2.5 million tons. Global nickel reserves are 94 million tons. The largest nickel-producing company overworld is Tsingshan Group.
One of the localities in the US where nickel is ruined is Riddle, Oregon, where several square miles of nickel-bearing surface are deposits located. The eagle mine project is a new nickel mine in the Upper Peninsula. The quarter of construction is completed in 2014. But in the first full year of cooperation, the Eagle mine produces 18000 tons of nickel.
Application & Uses:
Following are the applications and uses of Nickel:
- Due to its high melting point, and great resistance to corrosion, acids, and alkalies, nickel is largely used to make crucibles, dishes, and other chemical wares and also in the electrical industry.
- It is used for nickel plating in which NiSO4. 7H2O or NiSO4. (NH4)2SO4. 6H2O solution is used as an electrolyte.
- Finely divided nickel is used as a catalyst in the hydrogenation of oil and fats.
- Nickel metal combines with many other metals and forms alloys which find a number of applications.
- Nickel is also used in many specific and recognizable industrial & consumer products. It is involved in the manufacturing of stainless steel, alnico magnets, coinage, rechargeable batteries, and electric guitar strings.
- Nickel is used in many other alloys including nickel brasses, bronzes, and alloy with copper, chromium, aluminum, lead, cobalt, and silver because it is resistant to corrosion.
- In the 20th century, nickel is considered an excellent alloying agent and a precious metal. Nickel is used to collecting six PGE elements from ores.
- PGE stands for Platinum group elements. It is also used in the recovery of platinum and palladium. These both are involved in the hydrogenation of unsaturated hydrocarbon. But these both are very expensive metals.
- Basically, nickel is the ferromagnetic means that, it has domains in it. When current passes through it these domains become straight & nickel act as a magnet. So, it undergoes a change in length with a small variation.
- The nickel acts as a binder in cemented tungsten carbide. This nickel provides great and excellent corrosiveness in cemented parts but its hardness is less than cobalt binder.
- Approximately, 27% of nickel is utilized in engineering, 10% of it is used in building and construction, and 14% of it is used in making tubular products.
- Oil is used to make margarine and leftover Ni acts as a contaminant. Type 2 diabetic patients have 0.89 ng/ml of Ni in relative blood.
- It is also used in armor plating and burglarproof vaults metals.



Leave a Reply