
In order to discuss the Nernst equation, it is necessary to the terms electrode potential, Standard electrode potential, and electromotive force (Emf). In an electrochemical cell, two electrodes are used which conduct electrolyte that is the salt of the metal (anode or cathode). Also, a salt bridge is used which is used to prevent the exchange of ions between anode and cathode. The oxidation and reduction potentials are developed in these. But situation occurs when all the conditions are fixed of concentration, pressure, and temperature then standard electrode potential is developed.
Electrode Potential (E):
The electrical potential difference set up between a metal rod and its ions in solution is called the potential of the electrode simply the electrode potential. The electrode potential is defined as the potential difference of the electrical double layer formed at the contact of electrode metal and electrolyte. It is also called single electrode potential. The electrode potential is the measure of the tendency of an electrode to lose or gain the electron when it is in contact with its own ions.
Thus, we have oxidation potential and reduction potential. Electrode potential can also be defined as the tendency of an element to lose or gain the electron when it is in contact with its own ions in the solution. Electrode potential is developed in a half cell so electrode potential is also called the potential of the half cell because the half cell contains a single electrode in it. a single electrode represents the single electrode potential it is represented by denoted by E. Under the state of equilibrium, the potential difference developed between metal electrodes and their ions in the electrolyte is called electrode potential. Electrode potential may be of two types oxidation potential and reduction potential.
Oxidation Potential & Reduction Potential:
The cell reaction consists of two half-cell reactions of which one is an oxidation reaction and the other is a reduction reaction. Electromotive force Emf of a cell may be regarded as being made up of two single electrode potentials. The algebraic sum is equal to the Emf of a cell. So, Emf is defined as the sum of electrode potential of anode and cathode. Electromotive force is also denoted by Emf or E.
ECell = EAnode + ECathode
- EAnode is the tendency of an anode to lose the electron. It also tells how many electrons are lost from the surface of the anode. Basically, oxidation takes place on the anode. When oxidation takes place on an anode the electrode potential of the electrode is called oxidation electrode potential. In other words, we can say that when an electrode loses electrons its electrode potential is called oxidation electrode potential. In a reaction, an increase in oxidation state causes oxidation.
M → Mn++ ne–
- ECathode is the tendency of the cathode to accept or gain the electron. It also gives great information about electrons and how many electrons an electrode may accept. The reduction process takes place on the cathode. When reduction takes place on the cathode then the potential that develops on the cathode is called reduction electrode potential. In other words, we can say that when an electrode gain an electron its electrode potential is called reduction electrode potential. In a reaction, a decrease in the oxidation state to zero may cause a reduction.
Mn++ ne– → M
It is concluded that the oxidation and reduction potentials of an electrode are just the reverse of each other. Thus, if the oxidation potential of an electrode is +1.6 volts and the reduction potential of an electrode is -1.6 volts. The oxidation potential is the reverse of the reduction potential.
EOxidation = -EReduction
Standard Electrode Potential (E°)
In a half cell, the metal rod is suspended in a solution of its ions having one molar concentration and the temperature is 298 K the electrode potential of the half cell or electrode is for standard electrode potential. It is the kind of electrode potential in which many of the conditions are fully fixed. The standard electrode potential is denoted by E°. The conditions that are fixed should be:
- 1 molar concentration
- 298 Kelvin temperature (at room temperature)
- 1 atm pressure (in case of gas)
It is maybe of further two types Standard oxidation potential and standard reduction potential. The oxidation potential or reduction potential under the standard condition of concentration, temperature, and partial pressure (in terms of gas) is called Standard oxidation or reduction potential. The standard oxidation potential is denoted by E°oxi and the standard reduction potential is denoted by E°red. The standard oxidation potential is negative of the standard reduction potential or vice versa.
According to IUPAC (International Units of Pure & Applied Chemistry), the standard reduction potential is the standard electrode potential.
Sign of convention of E°:
Sign of convention refers to the positive, and negative values of the standard electrode potential. Basically, signs of the convention are the declaration of the IUPAC system. According to the IUPAC system:
- The electrode potential is given a positive sign if the electrode reaction involves reduction when connected to a standard hydrogen electrode. The sign of convention depends upon the combination of the electrode with the standard hydrogen electrode in a Galvanic cell. For example: in the SHE, the following reaction occurs:
At SHE: H2 ⇌ 2H+ + 2e– (OXIDATION)
At copper electrode: Cu2+ + 2e– ⇌ Cu (REDUCTION)
Hence, E° (Cu2+/Cu) = 0.337 volts
- Standard electrode potential is given a negative value when the electrode is combined with a standard hydrogen electrode (SHE) to give a Galvanic cell. The electrode attached to SHE acts as an anode and oxidation take place on the anode. With respect to SHE oxidation occurs and it will give a negative sign. For example: when the zinc electrode is connected with SHE oxidation occurs.
At zinc electrode: Zn ⇌ Zn2+ + 2e– (OXIDATION)
Hence, E° (Zn/Zn2+) = -0.763 volts
Electromotive force (Emf)
In an electrochemical cell, there are two electrodes that are corrected by the battery, and two containers are linked by a Salt Bridge. One of the electrodes has a higher potential than the other electrode means that one electrode is involved in the reduction and the other is involving oxidation. The difference between the electrode potential of two electrodes of an electrochemical cell when no current is flowing in the circuit is called electromotive force. It is denoted by the symbol Ecell.
Chemical Representation of electrochemical cell: M|M+ || M+|M
Emf of cell = Ecell = Eoxidation potential (Anode) + Ereduction potential (Cathode)
According to the IUPAC system, the oxidation potential of an anode is negative of the reduction potential of the anode.
Hence, Eoxidation potential (Anode) = -Ereduction potential (Anode)
Ecell = -Ereduction potential (Anode) + Ereduction potential (Cathode)
Ecell = -Eleft + Eright OR//
Ecell = Eright – Eleft
AT THE STANDARD CONDITION OF CONCENTRATION, TEMPERATURE, etc. The equation becomes:
E°cell = E°right – E°left
E°cell = E°cathode – E°anode
Numerical Problems:
1. What is the cell reaction and the emf of the cell at 298K of the cell?
Zn|Zn+2||Pb2+|Pb
E°(Zn|Zn+2) = +0.76V; E°(Pb+2|Pb) = +0.13V
E°cell = E°cathode – E°anode
E°cell = -0.13 -(-0.76)
E°cell = -0.13 + 0.76
E°cell = 0.63 V
We have taken the negative value because of the IUPAC system declaration. And also that reduction potential is preferable with negative signs.
1. What is the cell reaction and the emf of the cell at 298K of the cell?
Zn|Zn+2||Cu2+|Cu
E°(Zn|Zn+2) = -0.76V; E°(Cu+2|Cu) = +0.39V
E°cell = E°cathode – E°anode
E°cell = 0.39 -(-0.76)
E°cell = 0.39 + 0.76
E°cell = 1.15 V
2. What is the cell reaction and the emf of the cell at 298K of the cell?
Zn|Zn+2||Pb2+|Pb
E°(Zn|Zn+2) = +0.76V; E°(Pb+2|Pb) = +0.13V
E°cell = E°cathode – E°anode
E°cell = -0.13 -(-0.76)
E°cell = -0.13 + 0.76
E°cell = 0.63 V
3. What is the cell reaction and the emf of the cell at 298K of the cell?
Ni|Ni+2(1M)||Ag+|Ag(1M)
E°(Ni|Ni+2) = -0.25V; E°(Ag+|Ag) = +0.80V
E°cell = E°cathode – E°anode
E°cell = 0.80 -(-0.25)
E°cell = 0.80 + 0.25
E°cell = 1.05 V
Nernst Equation:
In 1888, a scientist named Walther Hermann Nernst discover the solubility product and the Nernst equation. He also relates the solubility product with the Nernst equation. Nernst equation is the equation that is used to find out the electrode potential of a half-cell and emf of a complete cell. It is also used to calculate the standard electrode potential.
If the concentration of electrolyte solution is 1 mole per liter or 1M and the temperature is 298 K (room temperature) the electrode potential of the given electrode is called standard electrode potential. However, if the concentration of the electrolyte solution is different from one molar the electrode potential of the electrode has a different value from that of standard electrode potential this value can be calculated with the help of the Nernst equation. For this purpose, the electrode reaction is always written as a reduction reaction. Suppose we have a reaction to determine the electrode potential of Mn+|Mn.
For a single electrode potential for half-cell
E = E° – [RT/nF]log10Q
E = E° – [RT/nF]log10[P]/[R]
The following terms are described under:
E = Electromotive force of the half-cell may be oxidation or reduction.
E° = Standard electrode potential of a half cell.
R = General Gas Constant (8.314 J/mol K).
n = No. of electrons lost or gain.
Q = Reaction quotient. It is basically the ratio of the concentration of products to reactants.
F = Faraday’s constant (96,500 C)
T = Temperature.
For a redox reaction emf formula for Nernst equation is:
Ecell = E° – [2.303RT/nF]log10Q
Ecell = E° – [2.303RT/nF]log10[P]/[Q]
For a redox reaction, Nernst equation becomes (At standard condition and constant values)
Ecell = E° – [0.059/n]log10Q
Ecell = E° – [0.059/n]log10[P]/[Q]
Consider a reaction of oxidation and reduction. Fe2+ + 2e– → Fe. It is a reduction reaction in which ferric ion accepts the electron. It is noticed that, concentration of pure liquid and pure solid is equal to unity or 1. The Nernst equation becomes:
Ecell = E° – [0.059/n]log10[Fe]/[Fe2+]
Ecell = E° – [0.059/n]log10[1]/[Fe2+]
Relation between Free energy and EMF of a reaction:
If in a cell n equivalents of reactants are converted into products, then the quantity of electricity that flows through the cell is nF, where F is the Faraday constant whose value is equal to 96500 coulombs per equivalent. If this amount of charge is transported through the cell of emf E volts, the amount of electrical work done by the cell is nFE. Since electrical work does not involve mechanical work resulting from the change in volume. So, this electrical done is equal to a change is Gibbs free energy.
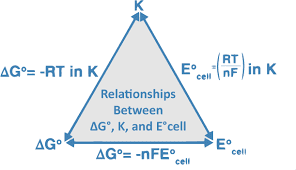
-△G = nFE
△G = -nFE
Gibbs’s free energy is also used to show the criteria of spontaneity that represents a reaction that takes place itself or not without any external force or push. The following table will represent the relation between the magnitude of △G, the magnitude of EMF, and reaction.
| REACTION | MAGNITUDE OF △G | MAGNITUDE OF EMF |
| Spontaneous | △G < 0 | E > 0 |
| Non-spontaneous | △G > 0 | E < 0 |
| Equilibrium | △G = 0 | E = 0 |
Derivation of Nernst Equation:
By the relation of Gibbs free energy and EMF
△G = -nFE
△Go = -nFEo
By the equation of Gibbs free energy for thermodynamics
△G = △Go + 2.303RT log10Q ……………..(1.1)
By putting the value of △G and △Go in equation 1.1…
-nFE = -nFEo + 2.303RT log10Q ……………(1.2)
Dividing equation 1.2… by -nF
-nFE/-nF = -nFEo/-nF + 2.303RT/-nF log10Q ………….(1.3)
Ecell = Eocell – 2.303RT/nF log10[P]/[R] ………………..(1.4)
At the standard condition of concentration of 1M, temperature (room temperature) 298K, R, F, and 1atm partial pressure the Nernst equation becomes:
Ecell = Eocell – 0.059/n log10[P]/[R] …………………..(1.5)
The terms used in the equation are described earlier after Nernest equation introduction.
Determination of Equilibrium Constant with Nernst Equation:
The Nernst equation is also related to the equilibrium constant. If we consider a reaction that is reversible. In that condition when the reaction goes in the forwarding direction products are formed the concentration of a reactant decreases but the concentration of the product increases.
So, to equilibrize the concentration of reactants and products reaction will go in backward direction towards the reactants. In this condition when equilibrium is established ΔG = 0, E = 0, -nFE = 0, and Q = kc. So, the Nernst equation becomes:
logKeq = nEo/0.059V
This equation represents a unique result that equilibrium constant Keq is directly proportional to the standard electrode potential Eo. The following results show that:
- K>1 means Eo>1 shows the forward reaction and reactants are converted to products.
- K<1 means Eo<1 shows the reverse reaction and products are further converted to reactants again.
The above points show that it will fit exactly to Le-Chatlier’s Principle. Le-Chatlier’s principle states that “reaction will go in that direction where there is low concentration and temperature. Rate of reaction will depend upon the temperature”.
Numerical Problems
1. Find the reduction potential and oxidation potential of the following reaction.
Fe2+ (0.01M)/Fe at 25oC
E°(Fe2+/Fe) = -0.45V
Solution:
This question can be solved by using the Nernst equation at standard conditions:
Ecell = Eocell – 0.059/n log10[P]/[R]
According to the given condition, there is a reduction reaction taking place in an electrochemical cell. So, the reaction occurs at that time is:
(0.01M) Fe2+ + 2e– → Fe (1M)
In this reaction, we take the concentration of Fe 1 or unity because the concentration of pure solid or liquid is 1 or unity. And the value of “n” is taken 2 because Fe2+ takes 2 electrons to reduce itself.
E(Fe2+/Fe) = -0.45 – 0.059/2 log10[Fe]/[Fe2+]
E(Fe2+/Fe) = -0.45 – 0.059/2 log10[1]/[10-2]
E(Fe2+/Fe) = -0.45 – 0.059/2 * 2
E(Fe2+/Fe) = -0.509 V (REDUCTION POTENTIAL)
Oxidation potential or reduction potential are the vice versa of each other. So the oxidation potential becomes:
E(Fe/Fe2+) = +0.509 V (OXIDATION POTENTIAL)
2. Find the reduction and the oxidation potential of Cr/Cr3+ (0.1M) at 25oC
Eo (Cr/Cr3+) = -0.75 V
This question can be solved by using the Nernst equation at standard conditions:
Ecell = Eocell – 0.059/n log10[P]/[R]
According to the given condition, there is a reduction reaction taking place in an electrochemical cell. So, the reaction that occurs at that time is:
(0.1M) Cr3+ + 3e– → Cr (1M)
In this reaction, we take a concentration of Fe 1 or unity because the concentration of pure solid or liquid is 1 or unity. And the value of “n” is taken 3 because Cr3+ takes 3 electrons to reduce itself.
E(Cr3+/Cr) = -0.75 – 0.059/3 log10[Cr]/[Cr3+]
E(Cr3+/Cr) = -0.75 – 0.059/3 log10[1]/[10-1]
E(Fe2+/Fe) = -0.45 – 0.059/3 * 1
E(Fe2+/Fe) = -0.77 V (REDUCTION POTENTIAL)
Oxidation potential or reduction potential are the vice versa of each other. So the oxidation potential becomes:
E(Fe/Fe2+) = +0.77 V (OXIDATION POTENTIAL)
3. Find the Electromotive force (EMF) of a cell. The cell representation is as under:
Cr/Cr3+ (0.1M)|| Fe2+/Fe (0.01M) at 25
Eo(Cr3+/Cr) = -0.75V
Eo(Fe2+/Fe) = -0.45V
This question can be solved by using the Nernst equation at standard conditions:
Ecell = Eocell – 0.059/n log10[P]/[R]
But to find the Eo of the cell we should use the formula: E0cell = Eocathode – Eoanode
According to the given condition, there is reduction reaction as well as oxidation reaction taking place in an electrochemical cell. So, reaction occur at that time is:
(0.01M) Fe2+ + 2e– → Fe * 3 (REDUCTION)
(0.1M) Cr → Cr3+ + 3e– * 2 (OXIDATION)
2Cr + 3Fe2+ → 2Cr3+ + 3Fe (REDOX REACTION)
In this reaction, we cannot take the concentration of 1 or unity because the concentration of both metals is given in the required condition of the question. In this reaction, 3 electrons are lost and 3 electrons are gained in oxidation, and reduction so, so the value of “n” is taken 6.
E0cell = Eocathode – Eoanode
E0 = -0.45 – (-0.75)
E0 = -0.45 + 0.75
E0 = +0.30 V
Now, we will calculate the EMF of an electrochemical cell:
Eocell = +0.30 – 0.059/6 log10[Cr3+]2/[Fe2+]3
Eocell = +0.30 – 0.059/6 log10[10-1]2/[10-2]3
Eocell = +0.30 – 0.06/6 * log 104
Eocell = +0.30 – 0.01 * 4 log 10
Eocell = +0.30 – 0.01 * 4
Eocell = +0.30 – 0.04
Eocell = +0.26 V
Test Yourself????????
Find the electromotive force EMF for the following reaction.
Zn/Zn2+ (0.1M)|| Sn2+(0.001M)/Sn
Eo(Zn/Zn2+) = -0.76 V
Eo(Sn2+/Sn) = -0.14 V
Answer: 0.56 V
Applications of Nernst equation
Following are the applications of Nernst equation:
- Used to calculate the single electrode potential i.e. oxidation or reduction potential.
- Also used to calculate the standard electrode potential.
- Used to show the relationship between EMF and Gibbs free energy.
- Also used to compare the spontaneity and feasibility of the cell.
- It is also used to measure the pH and solubility of sparingly soluble compounds in water.
- Also used to calculate the unknown ionic concentration.
Limitation of Nernst equation
It is very important demerit is that it cannot measure the cell potential because the flow of current affects the activity of ions on the electrodes. The ionic activity must be close to infinity in the dilute solution. For high concentration solutions, ionic activity is not equal to the ionic concentration. In such cases, to use Nernst equation experimental value must be conducted to take the true value of ionic concentration.



Leave a Reply