The kinetic molecular theory of gases tells us about the kinetic equation for gases. This theory also explains the kinetic energy of gases and on what factor the kinetic energy of ideal gas depends. The kinetic molecular theory of gases is completely valid for ideal gases but, partially valid for non-ideal gases or real gases. Here are some scientists and their contributions are given below:
- Bernoulli (1738): Founder of the kinetic molecular theory of gases.
- Clausius (1857): Derive kinetic equation and deduced all the gas laws from KMT.
- Maxwell: Presented the law of distribution of velocities.
- Van der Waal: Corrected the pressure and volume factor in the ideal gas equation to make it applicable to real gases.
Before we derive the kinetic equation, it’s very important to understand the postulates of the kinetic molecular theory of gases.
Postulates of Kinetic Molecular Theory
- Gas is made up of small particles called atoms or molecules. Gases like helium, neon, and argon have mono-atomic molecules.
- Molecules of small gas are identical in all respects (having the same mass, shape, and size).
- Molecules are constantly in random motion along a straight line. (assumption)
Let a molecule of gas is moving with speed u. The components of u are in three directions ux, uy, and uz.

It assumes that the probability of movement of a gas molecule in the X-direction, Y-direction, and Z-direction is the same so,
ux = uy = uz
The magnitude of u:
u = ux i + uy j + uz k
u = √ux2 + uy2 + uz2
u2 = ux2 + uy2 + uz2
If; ux = uy = uz
u2 = 3ux2
4. All the collisions of gas molecules among themselves and with the wall of the container are elastic in nature. Elastic in nature means there is no loss in momentum (momentum remains conserved) and kinetic energy (kinetic energy remains the same before and after collision).
5. The pressure of a gas is due to the collision of molecules with the wall of the container.
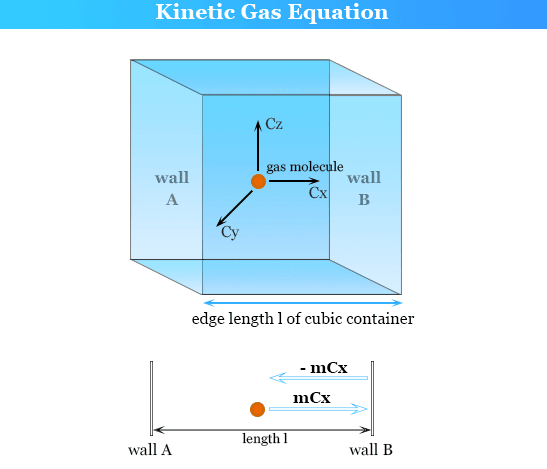
We have a container containing a gas molecule, a molecule of gas is moving with momentum mv:

When a gas molecule collides with the wall of the container, its momentum is changed (-mv). The momentum of a gas molecule is the same but opposite in direction. The gas molecule exerts a force on the wall of the container and the change in momentum is due to force so, the force exerted by a gas molecule per unit area is called pressure. The more the number of molecules collides, the more will be the pressure.
6. The kinetic energy of gas (ideal) depends only and only upon absolute temperature. The kinetic energy of gas does not depend upon the nature of the gas. It also does not depend upon the pressure.
The nature of gas means if we take one mole of hydrogen gas (ideal) and 1 mole of Oxygen gas (ideal) at 27 degrees Celsius, the kinetic energy of the gas is the same.
7. The volume occupied by gas molecules is negligible when compared to the volume of gas. (assumption)

8. There is no intermolecular force of attraction among molecules. (assumption)
9. There is no effect of gravity on the motion of the molecules of a gas.
Kinetic Equation for Gases
On the basis of kinetic theory, it is possible to derive a relation between the pressure and volume of gas in the form of an equation.
PV = 1/3 mn urms2
Where,
- P = Pressure
- V = Volume
- m = mass of one molecule of gas
- n = number of molecules
- urms2 = Root mean square velocity of molecules
What is urms2?
Let us have an nth number of gas particles having different velocities u1, u2, u3,…, un.
u = u1 + u2 + u3 + …… + un
Squaring both sides
u2 = u12 + u22 + u32 + …… + un2
Taking average (mean)
u2 = u12 + u22 + u32 + …… + un2 / N
where, N = total number of molecules
Taking under root both sides
√u2 = √u12 + u22 + u32 + …… + un2 / N
urms = √u12 + u22 + u32 + …… + un2 / N
This is called root mean square velocity.
Derivation of Kinetic Equation For Gases
Let we have a gas container having n number of gas molecules of direction x, y, and z, and length L.

Consider a small molecule of a gas with mass m and velocity u1. As we know that the velocity of a molecule has three components but we assume only ux component and we are also considering only one side of the container which is also along the x-direction.

How to pressure exerted by a small molecule of gas when colliding with the wall of the container?
- The collision of a gas molecule is elastic so kinetic energy remains the same. (Before and after collision)
- The momentum is changed.
Change in Momentum in Each Collision
Intial momentum = Pi = mux1
Final momentum = Pf = -mux1
Change in momentum = Initial momentum – Final momentum
△P = mux1 – (- mux1)
△P = 2mux1 (△P in each collision)
The change in momentum is due to force
Time Taken For Each Collision
As the length of the side is “L” and the speed of the molecule is ux1.
- Before collision: a molecule of a gas is moved toward a wall of the container.
- After collision: the molecule of a gas is moved away from the wall of the container.
So, the total length is 2L.
time = distance / speed
time = 2L / ux1 (time taken for each collision)
Force Exerted on the Wall
Force exerted on wall = △P / △t = change in momentum / change in time
putting the values of △P and △t
Force exerted on wall = 2mux1 / 2L / ux1
Force exerted on wall = 2mux1 × ux1 / 2L
Force exerted on wall = mux12 / L ——- (1)
(Force applied on the wall by one molecule due to collision)
If we want to write in terms of u, then
u = ux i + uy j + uz k
ux = uy = uz
u2 = ux2 i + uy2 j + uz2 k
u2 = 3ux2
u12 = 3ux12
ux12 = 1/3 u12
Put in eq (1)
Force exerted on wall = m/L × 1/3 u12
Force exerted on wall = m/3L × u12 (Force due to one molecule)
Force due to two molecules
F2= m/3L × u22
Force due to three molecules
F3= m/3L × u32
Force due to nth molecules
Fn= m/3L × un2
Fnet = F1 + F2 + F3 + …… + Fn
Fnet = m/3L × u12 + m/3L × u22 + m/3L × u32 + ….. + m/3L × un2
Fnet = m/3L ( u12 + u22 + u32 + ….. + un2)
Multiply and divided bt “n”
Fnet = m/3L ( u12 + u22 + u32 + ….. + un2 / n) × n
As, urms = √u12 + u22 + u32 + …… + un2 / N
urms2= u12 + u22 + u32 + …… + un2 / N
Fnet = mn/3L × urms2
As,
Pressure = Force / Area
Pressure = mn/3L × urms2 / A
Pressure = mn urms2 / 3L A
As, A = L × W = L × L = L2
P = mn urms2 / 3L × L2
P = mn urms2 / 3L3
As, L3 = volume of cylinder = L × W × h = L × L × L = L3
P = mn urms2 / 3 V
PV = 1/3 mn urms2
This is called Kinetic equation for gases.
PV = 1/3 mn urms2 ——– (1)
As we know that the kinetic energy is equal to 1/2 mv2
K.E of one molecule = 1/2 mu12
K.E of two molecules = 1/2 mu22
K.E of three molecules = 1/2 mu32
K.E of nth molecules = 1/2 mun2
K.E of n molecules = 1/2 mu12 + 1/2 mu22 + 1/2 mu32 + …… + 1/2 mun2
K.E of n molecules = 1/2 m (u12 + u22 + u32 + …… + un2 )
K.E of n molecules = 1/2 m (u12 + u22 + u32 + …… + un2 / n) × n
K.E of n molecules = 1/2 mn (urms2) ——– (2)
Divided (2) by (1)
K.E / PV = 1/2 mn (urms2) / 1/3 mn urms2
K.E / PV = 3/2
K.E = 3/2 PV ——– (3)
As,
PV = nRT
For one mole of an ideal gas n=1
PV = RT
Put of the value of PV in (3)
K.E = 3/2 RT (KE of one mole of an ideal gas)
The most important thing is that the kinetic energy of the gas is independent of its nature of the gas. It only depends on the temperature.
As we know,
K.E of one mole of an ideal gas = 3/2 RT
For n moles of an ideal gas
K.E of n moles of an ideal gas = 3/2 nRT
As 1 mole = NA molecules = 6.02×1023 molecules
K.E of one molecule = 3RT / 2NA
Where R/NA is Bolt’zmen constant (K)
K.E of one molecule = 3/2 KT
Note:
Basically, these derivatives are only true for monoatomic ideal gas. ( Helium, Neon, and Argon are monoatomic gases)
Deduction of Gas Laws From the Kinetic Equation of Gas
1. Boyle’s Law
According to the kinetic equation
PV = 1/3 mnurms2 or
PV = (2/3) × 1/2mnurms2 ——– (1)
Where,
- 1/2 mnurms2 is the average kinetic energy of molecules.
According to the kinetic molecular theory of gases, the average kinetic energy of the molecule is directly proportional to the absolute temperature.
K.E ∝ T
1/2 mnurms2 ∝ T
1/2 mnurms2 = KT ——– (2)
From equation (1) and (2)
PV = 2/3 KT
At constant temperature, PV = K which is Boyle’s Law
2. Charles’s Law
As
PV = 2/3 KT
V = 2/3 KT/P
At constant pressure, 2/3 K/P is constant
Hence, V ∝ T which is Charles’s Law
3. Avogadro’s Law
As the kinetic equation of gas,
PV = 1/3 mnurms2
For gas (1); P1V1 = 1/3 m1n1u1rms2
For gas (2); P2V2 = 1/3 m2n2u2rms2
If the pressure and volume of two gases are equal, then
1/3 m1n1u1rms2 = 1/3 m2n2u2rms2 ———- (3)
If two gases are at the same temperature, the average kinetic energy per molecule will be the same.
1/2 m1u12 = 1/2 m2u22 ——– (4)
Divided equation (3) by (4)
n1 = n2 which is Avodagro’s Law.
4. Graham’s Law of Diffusion
According to the kinetic gas equation,
PV = 1/3 mnurms2
For one mole of gas n=NA, we can write
PV = 1/3 mNAurms2
Where NA is Avogadro’s number (mNA = M)
PV = 1/3 Murms2
urms2 = 3PV/M = 3P/M/V = 3P/d (d=M/V)
or urms2 ∝ 1/d
urms ∝ √1/d
Now, “r” the rate of diffusion of the gas is directly proportional to the rms velocity of the molecules, thus
r ∝ √1/d which is Graham’s law of diffusion.
5. Dalton’s Law of Partial Pressure
If n1 molecules of a gas, each of mass m1 and velocity u1, occupy a volume V, then pressure P exerted by these molecules is given by,
P1 = 1/3 m1n1u1rms/V
Similarly, pressure P2 exerted by n2 molecules of another gas, each of mass m2 and velocity u2 is given by,
P2 = 1/3 m2n2u2rms/V
In the same way, pressure P3 of a third gas is given by
P3 = 1/3 m3n3u3rms/V
If all the three gases are kept together in another flask of the same volume and at the same temperature then total pressure P is given by,
P = P1 = 1/3 m1n1u1rms/V + 1/3 m2n2u2rms/V + 1/3 m3n3u3rms/V
P = P1 + P2 + P3 which is Dalton’s Law of partial pressure.
Numericals of Kinetic Equation
Find the kinetic equation of 8g of methane at 27℃.
Temp = 27℃ = 27 + 273K = 300K
K.E(1 mole) = 3/2 RT
For n moles,
K.E(n moles) = 3/2 nRT
n = m/M = 8/16 = 0.5
K.E(n moles) = 3/2 (0.5) × (8.314) × 300K
K.E = 1870.65 J/mol
Find the change in kinetic energy of one mole of an ideal gas when temperature changes by 50℃.
K.E = 3/2 RT
Intial K.E = K.Ei = 3/2 RT
Final K.E = K.Ef = 3/2 R (T+50℃)
△K.E = Final – Initial
△K.E = 3/2 R (T+50℃) – 3/2 RT
△K.E = 3/2 R (T+50 – T)
△K.E = 3/2 R × 50
△K.E = 3/2 × 8.314 ×50
△K.E = 610.05 J
At what temperature, the kinetic energy will be half of its value at 127℃?
Temp = 127℃ = 400K
K.E = 3/2 RT
K.E(127℃) = 3/2 R × 400K
At what temperature “T” the kinetic energy will half
K.E(T) = K.E(127℃) / 2
3/2 RT = 3/2 R × 400K / 2
T = 400/2
T = 200K
Another method:
K.E ∝ T
When the temperature is half, the kinetic energy will behalf
Given temp = 127℃ = 400K
K.E = 400K
Temperature becomes half 200K
K.E/2 = 200k
We have a container containing 1 mole of helium with a molar mass of 4g. One size length (L) of the container is 2cm and they make 500 collisions with the wall each second. Find the temperature of the gas.
As helium gas makes 500 collisions in one second so,
Time for one second = 1/500 s ——– (1)
Time is taken for one collision = Distance / speed
Time is taken for one collision = 2L / urms ——– (2)
Compare (1) and (2)
1/500 = 2L / urms
1/500 = 2 × 2×10-2 / uums
urms = (2 × 2×10-2) × (500)
urms = 20 m/s
K.E of one molecule = 1/2 murms2 ——– (3)
K.E of one molecule = 3/2 KT ——– (4) where K=Bolt’zmen constant
Compare (3) and (4)
1/2 murms2 = 3/2 KT
murms2 = 3 KT
T = murms2 / 3K ——– (5)
m is the number of molecules
Molar mass of one mole of He = 4g
mass of one molecule of He = 4 amu
As we know that 1 amu = 1.67×10-27kg
m = 4 × 1.67×10-27
K = 1.38×10-23 J/K (K is Bolt’zmen constant)
Putting all the values in (5)
T =4 × 1.67×10-27 × (20)2 / 3(1.38×10-23)
T = 2.672×10-24 / 4.414×10-23
T = 0.0515 K



Leave a Reply