
Oxygen is a colorless, tasteless, odorless gas and it is a member of the chalcogen group on the modern periodic table. Oxygen is the diatomic molecule and hence two atoms of the elements combine together to form dioxygen. The chemical formula of oxygen is O2. Oxygen is the second most electronegative non-metal after fluorine that’s why oxygen is the best oxidizing agent in the group. The oxygen has six electrons in its valance shell. They try to gain or share two electrons to complete their octet and to get stability. Accordingly, they tend to have a −2 oxidation number. The binding capacity of oxygen is limited to two oxygen forms ionic compounds with all metals.
| Atomic Number | 08 |
| Atomic mass | 16 |
| Density | 0.001429 |
| Melting point | −218℃, −368.77℉, 54.8K |
| Boiling point | −183℃, −297.31℉, 90.188K |
| Oxidation state | 0, −1, −2, +1, +2 |
| Electronegativity | 3.44 |
| Electron Affinity | 141 KJ/mol |
| Year discovered | 1774 |
| Discovered by | Joseph Priestley in Wiltshire, England, and independently by Carl Wilhelm Scheele in Uppsala, Sweden |
History
In 1772, Oxygen was discovered by a Swedish Chemist, Carl Wilhelm Scheele who obtained it by heating the potassium nitrate, mercuric oxide, and many other substances. The discovery of oxygen is attributed to Joseph Priestley. In 1774, an English chemist, Joseph Priestley obtained the element by heating a compound of Mercury and oxygen known as red oxide of mercury. The name oxygen, which means acid-producer. Oxygen is the first member of group 6A and the most active element of the group.
The name oxygen comes from the Greek word ”oxys” for sharp acid because oxygen was believed to be the constituent in the formation of acids. The Greek word ”gen or genes” meaning forming. The Greek words were combined by French chemist Lavoisier to create the name oxygen.
Oxygen-The Most Abundant Element of Earth
Oxygen is the most abundant element on the earth. It occurs in nature in both free and combined states. Free oxygen constitutes about 21% by volume of atmospheric air and about 33% by volume of dissolved air. In compounds, oxygen accounts for about 50% of the mass of the earth’s crust, oceans, and air. Oxygen is present as oxides of both metals and non-metal, which makes up the rocks and clays. Water, open of the most abundant compound on the earth, contains 88.9% of oxygen by mass.

Oxygen is present in water molecules and water is essential to all life. Oxygen is present in many organic compounds. Most living organisms use oxygen for respiration and oxygen makes up 1/4 of the atoms in living organisms. Oxygen resides in the atmosphere and is also involved in the combustion process i.e. oxidation. A lot of things can burn down in oxygen such as wood, paper, and plastic.
If we increase the oxygen concentration then things start to burn quite more intensely. Also, like any gas, oxygen can be condensed i.e. liquified. For the condensation of liquid oxygen, we have used liquid nitrogen. The boiling point of nitrogen is 13℃ lower than that of oxygen so, bypassing pure oxygen from a cylinder through a copper coil that is cooled by liquid nitrogen we can obtain liquid oxygen.
Importance of Liquid Oxygen
The oxygen liquification itself looks very fascinating. In large hospitals, oxygen is normally stored in liquid form in large tanks to save space and for easy transportation. The most interesting fact is that liquid state oxygen is not a transparent liquid, it’s in fact blue. It is because of the light absorption spectrum of oxygen atoms. Liquid oxygen is paramagnetic and is attracted to magnets. In fact, in the rocket engines of shuttle liquid oxygen served as an oxidant and liquid hydrogen was the fuel.
Also, the molecules that make up our body contain a lot of oxygen. Oxygen is produced from photosynthesis in plant cells.

Preparation of Oxygen
Preparation of oxygen in the laboratory and in the industry are mentioned below:
In the Laboratory
Generally, the thermal decomposition method is used to produce oxygen gas in laboratories.
- Heating of metal oxides with low activity such as Ag2O and Hg.
2HgO(s) → 2Hg(l) + O2(g)
Ag2O(s) → 2Ag(s) + 1/2 O(g)
- Heating of peroxides, such as Na2O2 and H2O2. Hydrogen peroxide in the presence of finely divided metals and manganese dioxide decomposes to give water and dioxygen.
2H2O2(l)→ 2H2O + O2 (g)
- Heating of some nitrates (NO3–) and chlorate (ClO3–) compounds.
2KClO3 → 2KCl + 3O2 (g)

- Heating of permanganate (MnO4–) compounds.
2KMnO4 (S) → K2MnO4 (s) + MnO2(s) + O2(g)
- Electrolysis of water, if electricity is used for decomposing a substance is called electrolysis. When electricity is passed through acidic water it decomposes into oxygen and hydrogen gases. Water is a bad conductor of electricity. Therefore, a little salt or alkali is added to water for easy conduction of electricity. When the direct current passes through the water oxygen appears at the positive anode while hydrogen is released from the negative cathodes. In terms of volume, exactly two times more hydrogen is produced than oxygen. This method is more expensive than others.
2H2O(l) → 2H2(l) + O2(g)
In Industry
The preparation of oxygen in the industry involves two steps:
- The liquefaction of air
- The fractional distillation of the liquid air
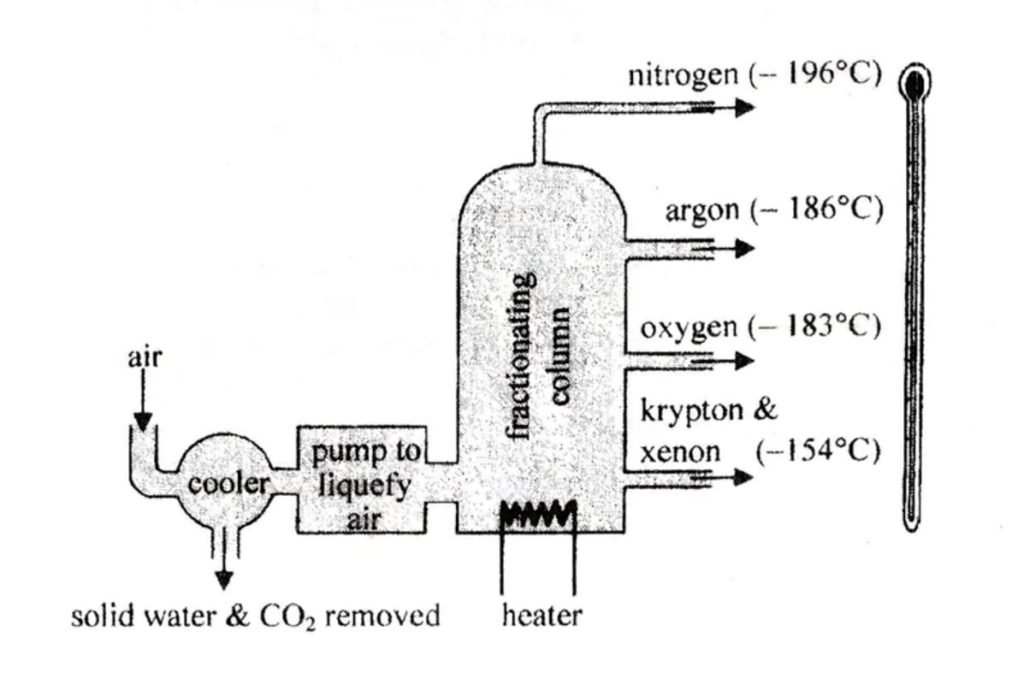
Liquification of Air
Air, in the gaseous atom, is first passed through caustic soda to remove any CO2 present. It is then compressed to a pressure of about 200 atm then cooled and allowed to escape rapidly through a very small hole. The sudden expansion of the air into a region of lower pressure causes it to cool. It is cooled until it becomes a liquid at -200℃.
Fractional Distillation of Liquid Air
The liquid air is then led to a fractionating column. During distillation nitrogen gas, with a lower boiling point of -196℃, is evolved first, leaving behind the liquid very rich in oxygen. Further heating turns the liquid oxygen into a gas -183℃. The oxygen is dried, compressed, and stored in a steel cylinder under a pressure of 100 atm.
Physical Properties
- It is colorless, odorless diatomic gas, and paramagnetic in nature. Paramagnetic nature means it is attracted to magnets.
- Oxygen is highly reactive non-metal.
- This diatomic gas is a strong oxidizing gas.
- It is the second most electronegative element after fluorine.
- Dioxygen reacts with metals and non-metals to give oxides of the respective element.
- It is highly soluble in water. Marine organisms utilize this dissolved for their survival.
- It can be liquified at normal pressure and at a temperature of -183℃. Liquid oxygen is pale blue in color. At -218.4℃ liquid oxygen freezes into a snow-like mass.
- It has three stable isotopes O816, O817, and O818.
- Oxygen exhibit allotropic forms.
- Oxygen is usually divalent.
Chemical Properties
Chemical properties are the characteristics that determine how a substance will react with other substances when it changed chemically. Even though oxygen is the second most active non-metals, the reaction of oxygen is slow because of the strong double bond between oxygen atoms. Generally, oxygen takes a -2 charge in its compounds except for OF2. Oxygen has only a +2 charge in OF2. Oxygen combines with all elements (except the noble gases, some halogens, and some unreactive metals) to form oxides. Some chemicals properties of oxygen are given below:
- Flammability-It does not burn.
- It supports combustion
- Occur in many compounds including water, CO2, and, Iron ores.
- Oxides of some metals form peroxides by the addition of oxygen.
- It doe not react with water.
- It does not react with alkalies.
Reactions
Reactions with Metals
4Na + O2 → 2Na2O
2Ca + O2 → 2CaO
Mg + O2 → 2MgO
4Al + O3 → 2Al2O3 (In the presence of ∆)
4Fe + 2O3 → 2Fe2O3 (In the presence of ∆)
Reactions with Non-metals
H2 + O2 → 2H2O
N2 + O2 → 2NO (In the presence of ∆ and an electric spark)
S + O2 → SO2
C + O2 → CO2
Reactions with Compounds
2NO + O2 → 2NO2
2SO2 + O2 → 2SO3
4NH3 + 5O2 → 4NO + 6H2O
Reactions with Hydrocarbons
CH4 + 2O2 → CO2 + 2H2O + ∆H
The more the number of carbon in hydrocarbons, the more the heat is evolved.
C2H6 + 7O2 → 2CO2 + 3H2O + ∆H
Classification of Oxides
Oxides are classified into simple oxides and mixed oxides (Fe2O3, Pb3O4).
Simple Oxides
Simple oxides are classified into three types:
1. Acidic Oxides
Acidic oxides are non-metallic oxides e.g. CO2, SO2, Cl2O7, NO2. When we react these acids with water they give respective oxides.
CO2 + H2O → H2CO3
SO2 + H2O → H2SO4
2. Basic Oxides
Basic oxides are metallic oxides e.g. Na2O, CaO, MgO. When they react with water they give respective bases.
CaO + H2O → Ca(OH)2
Na2O + H2O → 2NaOH
3. Amphoteric Oxides
Amphoteric oxides are metalloids e.g. Al2O3.
Note:
CO, N2O, NO are neutral oxides and have no effect on litmus paper.
Allotropic Form of O2-The Ozone
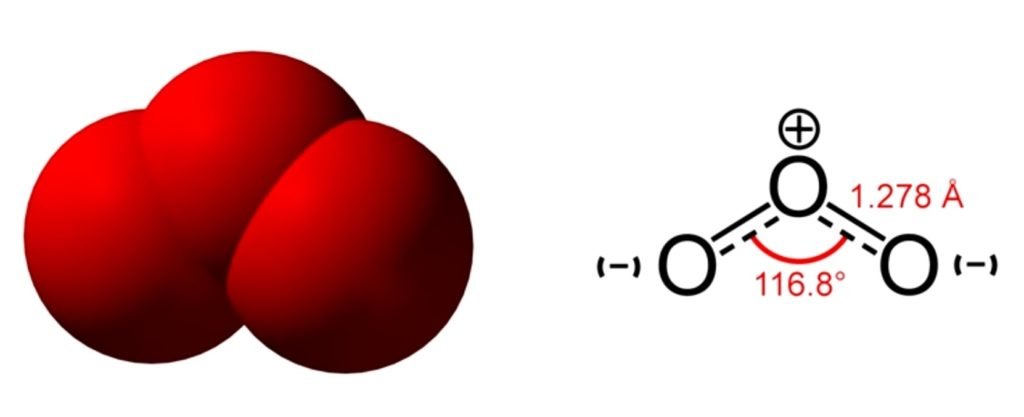
Oxygen has two-atom molecules, but there is a molecule that consists of 3 oxygen which is ozone. Such a phenomenon is called an allotropic modification. Ozone is a light blue-colored gas with a sharp pleasant odor. It may be liquified at -112℃ and may be solidified at -193℃. Ozone is slightly soluble in water. Ozone can be prepared by passing pure oxygen gas through an electrical discharge.
3O2 → 2O3
The electrical energy breaks the bonds in some O2 molecules to give oxygen atoms that react with other oxygen molecules to form ozone.
O2 → 2O
O2 + O → O3
Ozone exists naturally in the upper atmosphere of the earth. In the troposphere, we do not have much ozone and it is mainly found in the upper altitudes. The ozone layer is especially important because it absorbs UV light, acting as a screen to block this radiation, which can cause skin cancer. Without it, our eyes would have ceased to see in a couple of hours. The retina would have simply been burned and life on earth would be pretty much impossible. When an ozone molecule absorbs this energy, it splits into an oxygen molecule and an oxygen atom.
O3 → O2 + O
The characteristic smell of ozone can often be detected near high voltage electrical apparatus, such as some early model photocopiers.
Properties
- It is pale blue gas having a pungent smell.
- It is heavier than air.
- It is slightly soluble in water but more soluble in organic solvents like CCl4, ether, etc.
- Ozone is diamagnetic in nature.
Chemical Properties
- Neutral towards litmus. (It either converts blue litmus to red and red litmus to blue)
- It is thermodynamically unstable (with respect to oxygen) because ∆H is negative and ∆S is positive.
- At high concentrations, ozone can be explosive.
- It is a very good oxidizing agent.
O3 → O2 + [O]
[O] is the nascent oxygen and it is responsible for oxidizing the nature of ozone. For example;
PbS + O3 → PbSO4 + 4O2
In the reactant, Sulpher has a -2 oxidation state.
In the product, Sulpher has a +6 oxidation state.
Structure of Ozone
What is Medical Oxygen
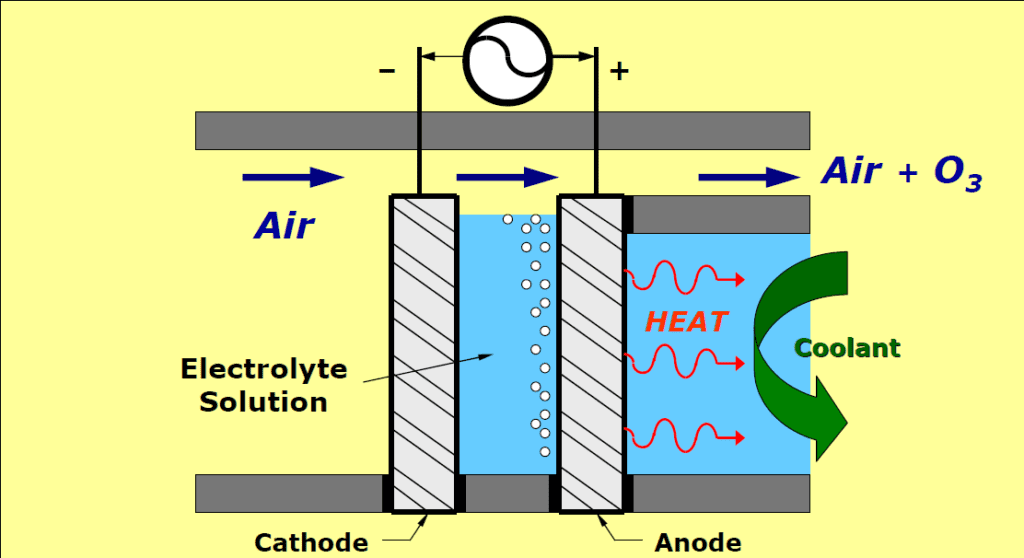
65% of the human body is oxygen. It is the basic need of human life on earth to survive covid-19 hampers the supply of oxygen in various parts of the body in severe patients and hence they require medical oxygen. But what is medical oxygen? How is it made? How is it supplied?
Oxygen is the lifeline of humans and animals but this life-sustaining gas is also used in many industrial and scientific applications medical oxygen is different. It is high-purity oxygen that is used for medical treatments. There are several methods to produce medical oxygen. The most common commercial method is the separation of air in an air separation unit or ASU. Air separation units are the plants that treat large volumes of gas from a few hundred tons to twenty thousand tons per day. The Air separation units used a method called the fractional distillation method to produce pure oxygen.
Gases from the air are separated into various components, after cooling it into a liquid state then extracting liquid oxygen from it. The air is first cooled to -181℃ at this temperature the oxygen liquefies. The boiling point of nitrogen is -196℃ so it remains in gaseous form at this temperature but Argon has a similar boiling point to that of oxygen which is -186℃. So, a significant amount of argon liquefies with oxygen. This liquid mixture of oxygen and argon is drained, decompressed, and passed through a second low-pressure distillation column in order to separate.
How is it Supplied?
The final purified liquid oxygen is then transported using cryogenic containers. Cryogenic containers are highly insulating containers in which liquid gases are stored at a very cool temperature. Hospitals can also opt for on-site generation of oxygen by using the pressure swing adsorption technique also known as Pressure Swing Adsorption or PSA. In this method, oxygen is produced from the air by concentrating it. Producing the oxygen near the hospitals is an advantage as there is no transport involved and it can provide more oxygen than the hospital can secure with cylinders.
There are portable oxygen generators as well known as oxygen concentrators. These can be used at home. In this, the air is compressed using compressors and then sent to sieve beds. In sieve beds, there is a compound called zeolite which separates nitrogen from oxygen. This leads to the generation of pure oxygen and the resultant air is 90 to 95%.
Molecular Orbital Theory
- MOT tells us about the molecular orbitals.
- MOT tells us about the magnetic nature. According to molecular orbital theory, O2 is paramagnetic in nature.
- MOT tells the stability of molecules e.g O2, O22-, O2+ the order of stability is explained by MOT.
- MOT tells us the bond order in fractions e.g. The bond order of O2+ is 2.5.
MOT Diagram of O2
σ1S2, σ*1S2, σ2S2, σ*2S2, (π2Px2=π2Py2) σ2Pz2, (π*2Px1=π*2Py1) σ*2Pz

Bond Order
no of an electron in bonding – no of an electron in anti-bonding / 2
8-4 / 2
Bond order of O2 = 2
Magnetic Nature
O2 is paramagnetic in nature.
MOT Diagram of O2+
σ1S2, σ*1S2, σ2S2, σ*2S2, (π2Px2=π2Py2) σ2Pz2, (π*2Px1=π*2Py) σ*2Pz

Bond Order
no of an electron in bonding – no of an electron in anti-bonding / 2
8-3 / 2
Bond order of O2+ = 2.5
Magnetic Nature
O2+ is paramagnetic in nature.
MOT Diagram of O2–
σ1S2, σ*1S2, σ2S2, σ*2S2, (π2Px2=π2Py2) σ2Pz2, (π*2Px2=π*2Py1) σ*2Pz

Bond Order:
no of an electron in bonding – no of an electron in anti-bonding / 2
8-5 / 2
Bond order of O2– = 1.5
Magnetic Nature
O2– is paramagnetic in nature.
MOT Diagram of O22-
σ1S2, σ*1S2, σ2S2, σ*2S2, (π2Px2=π2Py2) σ2Pz2, (π*2Px2=π*2Py2) σ*2Pz
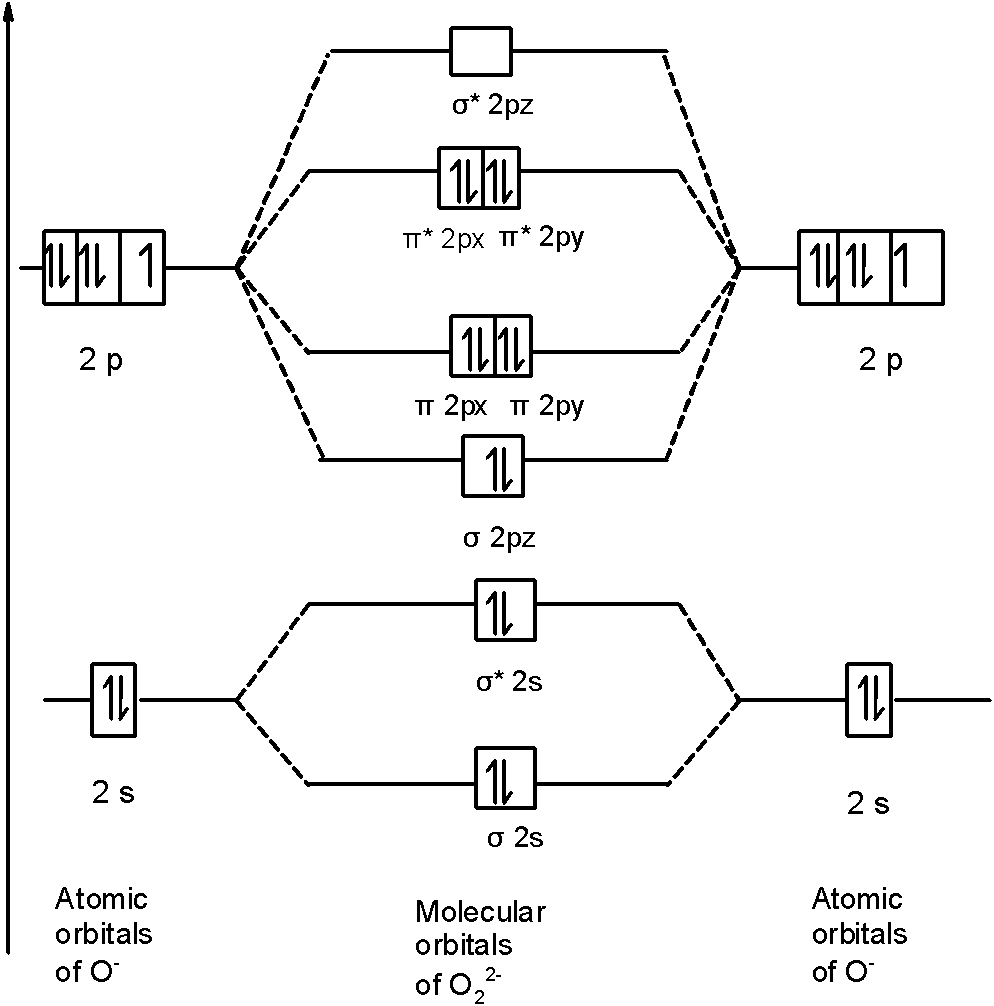
Bond order
no of an electron in bonding – no of an electron in anti-bonding / 2
8-6 / 2
Bond order of O22- = 1
Magnetic Nature
O22- is diamagnetic in nature.
Which one is more stable O2, O2+, O2–, O22-?
“More the number of the anti-bonding electrons, lesser the stability”
So, the order will be
O2+ > O2 > O2– > O22-
Uses of Oxygen
- Oxygen is used for respiration, medicine, and industrial purpose.
- Oxygen is essential for all living organisms as it is needed for resp[iration.
- Oxygen cylinders are carried by mountaineers, sea divers, and astronauts.
- Oxygen is necessary for burning fuels. It is also used in wastewater treatments.
- It is used in a large number of oxygen-containing organic compounds.
- Liquid oxygen is used as fuel.
Industrial Uses
- Oxy-hydrogen flame (2800℃) and oxy-acetylene (3300℃) are used in the cutting and welding of metals.
- Oxygen is used for the production of pig iron and the conversion of pig iron into steel.
- It is used in chemical industries as an oxidizing agent in the manufacturing of nitric acid from ammonia and sulphuric acid from sulfur dioxide.
- It is also used in metal production especially steel but also copper, zinc, and lead is used.
- It is used in metallurgical process to remove impurities.
Medical Uses
- Oxygen, stored in cylinders, is provided to persons suffering from respiratory disorders like asthma.
- Oxygen is used for reviving people in case of suffocation.
- Oxygen is a major constituent of the carbogen given to patients in order to stimulate natural breathing.
- In dentistry, a mixture of oxygen and nitrous oxide is used in anesthesia.
Explain the difference between Atoms and Molecules of Oxygen?
An atom of oxygen is the smallest unit of the oxygen element and each atom of oxygen contains 8 electrons, 8 protons, and 8 neutrons. Oxygen atoms are very unstable and reactive but they can form covalent bonds with other oxygen atoms to form pairs, called oxygen molecules. Oxygen molecules are stable and these are the particles that make up oxygen gas.



Leave a Reply