This blog will provide you with a great piece of information about the supersaturated solution. But discussing different types of solutions is necessary. We know that two things a solute or solvent recombine to form a solution. In other words, we can say that a solution must be single-phase having a uniform composition.
We can also define a solution as:
A homogeneous mixture (having uniform composition) of two or more substances without any fixed ratio. The composition of solute or solvent added to each other can vary within certain limits. This is called a solution.
The best example of the solution is:
- Salt in water.
- Presence of carbon dioxide in carbonated drinks.
Types of Solutions
On the basis of the presence of the concentration of solute, solutions are divided into three types: Saturated solution, Unsaturated solution, and Supersaturated solution.
1. Saturated Solution
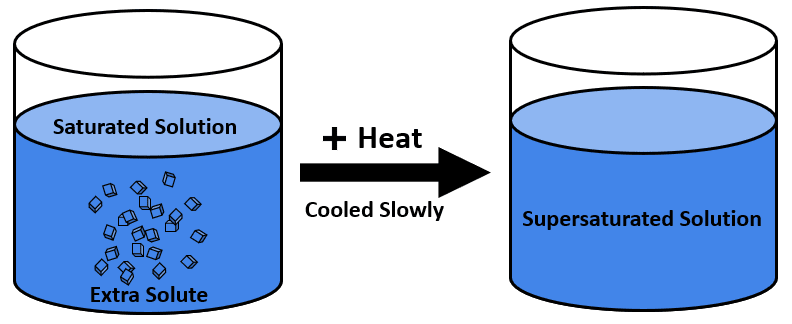
If the maximum amount of solute is added to the solvent, then the solution is said to be saturated. In other words, if a condition is given to the solution, and all the solute is added to the solvent under this condition, the solution is said to be saturated.
Under the given condition of heat, the solute is mixed with the solvent. Still, a condition occurs when adding more solute causes the precipitation of the solute molecule and even gas being released. This condition is referred to as a “SATURATED SOLUTION“.
We can prepare a saturated solution by adding more and more solute resulting in the precipitation of solid solute. These crystals form a highly saturated solution. An excellent example of a saturated solution is carbonated water having carbon dioxide in it.
2. Unsaturated Solution
The unsaturated solution must have less solute concentration than its equilibrium solubility. When a solute is added to the solvent, two reactions occur. The first is dissolution and the second is crystallization. When the rate of dissolution is much greater than the rate of crystallization then a solution is said to be unsaturated.
Equilibrium solubility refers to the saturation point. A point where the maximum solute is present. If the solute is less than that of saturation point then the solution is said to be unsaturated. For example:
- Less concentration of NaCl or sugar in water than the required concentration.
- Air, smoke & mist are good examples of unsaturation solutions.
What is Supersaturated Solution???
A supersaturated solution must refer to more solute concentration than a saturated point. This is the solution that contains a relatively more amount of solute than to saturate a solution. In this, a condition of dynamic equilibrium is established. Concentration is basically regarded as the dissolution of solute dissolved per volume. So, if the concentration of solute is present more than the maximum theoretical concentration is called a super saturated solution. But understanding the concept of dynamic equilibrium is necessary.
How will you discuss Dynamic equilibrium??
The supersaturated solution gives a deep concept of Dynamic equilibrium. The state at which the rate of two reactions takes place at the same time but in opposite direction. In supersaturated solution, the rate of dissolution and rate of crystallization occurs at the same time but in opposite direction.
Explanation:
Solubility may occur if no condensation or crystallization takes place. This means to say, at that position, the solute can be present at its maximum level above the point of saturation. But there is a phenomenon representing that blocked process of crystallization & condensation which is called the Kinetic barrier to Nucleation.
Nucleation is basically a kinetic barrier to the process of crystallization because nucleation refers to the process by which solute when separated from solution either forms a crystal or water droplet. It is a kinetic barrier to crystallization because when the atomic structure of a crystal is formed it requires energy great amount to form the crystal.
How does supersaturation occur in Phase change??
- The formation of 3-D nuclei occurs when physical & chemical processes take place & also occurs when the medium is supersaturated.
- The formation of nuclei is also associated with Gibb’s free energy. During thermodynamics, when a homogeneous mixture is formed no nuclei are formed soon in the system, and the solution becomes supersaturated.
- So, the system is in a state of dynamic equilibrium.
- The nucleation of a new phase depends upon many factors. Temperature and pressure.
- There is always a supersaturation level when new phase nucleates instantaneously. So, a new phase precipitates are formed.
- Dynamic equilibrium is a metastable equilibrium that defines metastable & dynamic width. The upper limit of metastable equilibrium corresponds to the supersaturation level.
Characteristics of Supersaturated Solution
- The concentration of solute exceeds its maximum saturation level.
- The solution must be in a metastable state.
- Two processes are kinetically inhibited i.e. crystallization or condensation didn’t cross the kinetic barrier to nucleation.



Leave a Reply