Before going to discuss racemic mixture first of all we have to know important term enantiomers.
Enantiomers:
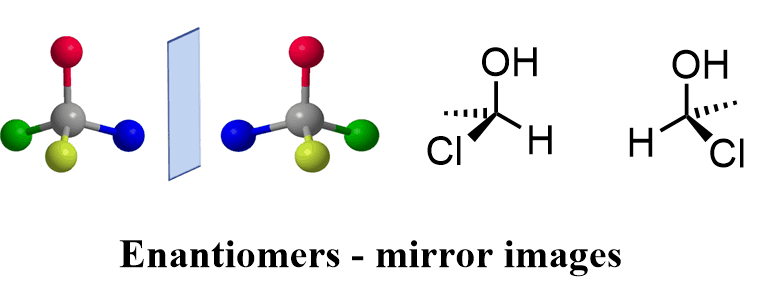
- The isomers that are non superimposable mirror images to each other are called enantiomers.
- Enantiomers have identical physical or chemical properties.
- Enantiomers rotate plane-polarized light in opposite directions.
- These are represented by the ‘+’ or ‘_’ . And by ‘D’ or ‘L’.
- Enantiomers have no center of symmetry and no plane of symmetry.
Racemic Mixture:
A racemic mixture or a racemate is a 50/50 or equivalent ratio mixture of ‘+’ or ‘_’ enantiomers. So the overall optical rotation is zero. A racemate is denoted by d/l or +_.
For example:
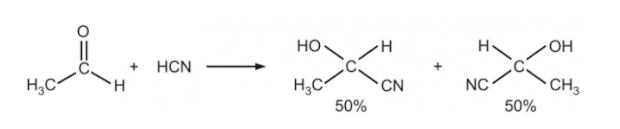
The above example is a racemic mixture of sodium ammonium tartrate.
Resolution of Racemic Mixture:
A racemic mixture resolves to separate it into original or pure enantiomers. This is called resolution of racemic mixture. As enantiomers have similar physical properties like boiling point, solubility factor, chromatographic retention time, that’s why these cannot be resolved by common physical methods.
Methods Of Resolution Of Racemic Mixture:
There are following some methods to resolve racemic mixture into enantiomers.
- Mechanical separation method
- Biochemical separation method
- Crystallization Method
- By diastereomers
- Precipitation method
- chromatography separation
- kinetic method
1. Mechanical Separation method:
Based on different shapes crystals of enantiomers are separated from the racemic mixture. Crystals of two forms have different shapes these are separated by magnifying lenses or forceps. This method was first used by Pasteur who used the method to resolute sodium ammonium tartrate that crystallized in the form of a racemic mixture below 27 degree.
Disadvantages:
This is a time-consuming method and all of the compounds cannot be crystallized out at room temperature there is a need for temperature to crystallize the different compounds.
2. Biochemical separation method:
This method was introduced by Pasteur. In this method, small membrane-bounded organisms like bacteria, fungi, molds, and yeast are grown into dilute racemic solution as a result of which these organisms eat one of the enantiomers immediately rather than the other.
For example, mold penicillium glaucum immediately destroys the + enantiomer of racemic ammonium tartrate and left the _ enantiomer in the solution.
3. Crystallization Method:
In this method seedling of a saturated solution of a racemic mixture occur with a pure crystal of one of two enantiomers. After adding an enantiomer the solution becomes supersaturated and crystallization starts.
For example, by using the crystals of – asparagine + sodium ammonium tartrate crystallize out from the racemic mixture.
4. BY Diastereomers:
It is the most commonly used method in which conversion of enantiomers occurs into the diastereomers that are physically different from each other having a different boiling point, melting points, solubility, etc.

Resolution of Racemic Mixture of Acids:
For example, if a mixture of D/L enantiomers of acid after converting it to a salt of D-configuration has a chiral base the salt will be a mixture of two diastereomers (D-acid, D- base or L-acid, D- base). These diastereomers are different in their physical properties and these may be separated by the crystallization method. If diastereomers salt will separate completely the salt will be D or L enantiomers.
- Resolution of the racemic mixture by diastereomers requires chiral bases. Naturally occurring chiral bases that are mostly used are Brucine, strychnine, and quinine.
- Synthetic amines like 2-amino-1-butanol, amphetamine, and 1-phenylethanolamine are also used as chiral bases.
Resolution of Racemic Mixture of Bases:
For the resolution of racemic base chiral acids are used. The principle is same as used for the resolution of the racemic acids.
Chiral acids like +tartaric acid, _ malic acid, _ mandalic acid and +- camphor-10 sulphonic acids are used for the separation of racemic bases.
Resolution of Racemic Mixture of Alcohols:
For the resolution of racemic alcohols, chiral acids are used that converts the alcohol to the diastereomer’s esters. This is not useful because esters being liquid high molecular weight compounds. And if diastereomers esters are not separated by the crystallization method these can be separated by some other methods.
Two chiral acids that are useful for the resolution of alcohols are menth-oxy-ethanoic acid and 3β-acetoxy-∆5 -ethanoic acid.
The most common method used for the resolution of alcohols is the conversion of racemic alcohols to half ester dicarboxylic acids e.g 1,2-benzene dicarboxylic acid with anhydrides.

5. Precipitation Method:
The precipitation method involves the precipitation of a racemic mixture by the addition of a precipitating reagent. When (+,_) Narcotine dissolved in HCl solution precipitates the +narcotine.
6. Chromatography separation:
Certain absorbents having the ability to adsorbs d or l form of the racemic mixture are used in the chromatography method to resolute the racemate.
For example, a racemic mixture of mandalic acid can be resolute by the column chromatography method. Selective chiral reagents are used as stationary. The mobile phase has more affinity toward one of the two enantiomers. This is the latest method used for the resolution of a racemic mixture.
7. Kinetic Method:
This method is based on the ability of a enantiomers to react with the optical active compound.
For example, methanol reacts faster with + mandalic acid than -mandalic acid. Thus it depends on the kinetics of reactions.



Leave a Reply