
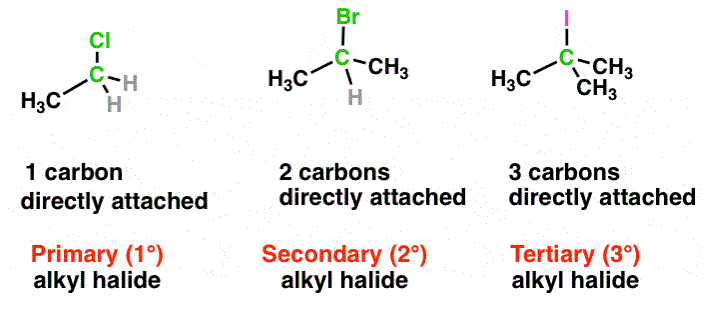
Monohaloderivatives of alkanes are called alkyl halides. The general formula of alkyl halides is R X, where R is an alkyl group or substituted alkyl group and X is any halogen atom. They contain the halogen atom attached to a saturated carbon atom (- C – X) as their functional group.
Alkyl halides are classified as primary (1°), secondary (2°), or tertiary (3°) depending upon whether the halogen atom is attached to a primary, secondary, or tertiary carbon.
Prepration of Alkyl Halides
1. Halogenation of Alkanes
A reaction in which an atom or a group in a molecule is replaced by another atom or a group is called a substitution reaction, if the substituent is a halogen atom, the reaction is called halogenation.
Alkanes react with Cl2 or Br2 in the presence of UV light or at 250 – 400℃ to give alkyl halides along with polyhalogen derivatives. Therefore, halogenation of alkanes is not a useful method of preparation because of the difficulty of separating the products. Halogenation of alkanes proceeds by a free radical chain mechanism.
CH4 + Cl2 → CHCl3 + CH2Cl2 + CHCl3 + CCl4
The reaction with fluorine F2 is too violent to control and I2 does not react at all.
2. Addition of Halogen Acids to Alkenes
Alkyl halides are formed when halogen acids (HCl, HBr, Hl) are added to alkenes. The addition follows Markovinikov’s rule, except for the addition of HBr in the presence of organic peroxides (R-O-O-R).
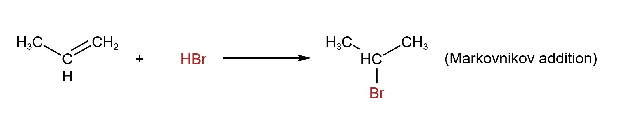
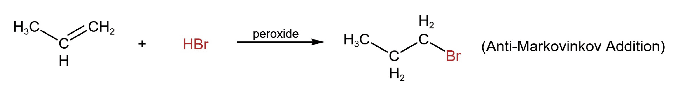
The order of reactivity of halogen acid is HI > HBr> HCl.
3. From Alcohol
When alcohols react with halogen acids or with inorganic acid halides such as thionyl chloride SOCl2, PCl5, PBr3, etc. to produce alkyl halides.
(1) Reaction of Alcohols with Halogen acids gives Alkyl halides
As we discuss above the reactivity of halogen acids decreases in the order HI > HBr > HCl and the order of reactivity of alcohols is tertiary > secondary > primary.
ROH + HX → RX + H2O
Mechanism
The alcohol accepts a proton from the halogen acid and then the protonated alcohol dissociated to form a carbocation and water. Primary alcohols react with hydrogen halides by the SN2 mechanism.

In the next step, the carbocation reacts with a nucleophile (as a halide ion) to form an alkyl halide.
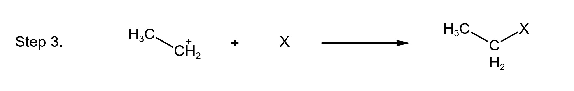
Tertiary alcohols react with hydrogen halides by the SN1 mechanism.

The reaction depends upon the nature of alcohols and halogen acid.
- Hydrochloric acid is the least reactive of the halogen acid and it does not react with primary alcohols unless ZnCl2 or similar lewis acid is added to the reaction mixture as well. ZnCl2 is a good lewis acid that forms a complex with the alcohols, which provides a better leaving group than – +OH2. A concentrated solution of ZnCl2 in hydrochloric acid is often used for the preparation of primary or secondary alkyl halides from alcohols.
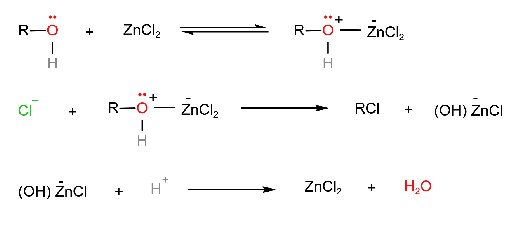
On the other hand, the tertiary alcohols react with hydrochloric acid readily even in the absence of ZnCl2 because of the relative stability of the tertiary carbonium ion.
- Alkyl bromides are produced by heating alcohols with constant boiling HBr in the presence of little conc. H2SO4. Sulphuric acid, however, is not used in the preparation of secondary and tertiary bromides because in its presence they are dehydrated to form alkenes. It is convenient to prepare the HBr in situ by the reaction of KBr with conc. H2SO4.
KBr + H2SO4 → KHSO4 + HBr
ROH + HBr → RBr + H2O
- Alkyl iodides are prepared by heating the alcohol with concentrated HCl, But the yield of alkyl iodide is very low due to its reduction to alkane by HI.
CH3CH2OH + HI → CH3CH2I + H2O
CH3CH2I + HI → CH3CH3 + I2
This difficulty is overcome by heating alcohol with sodium or potassium iodide in 95% phosphoric acid.
3ROH + 3KI + H3PO4 → 3RI + K3PO4 + 3H2O
(2) Reactions of Alcohols with Phosphorus halides or Thionyl chloride gives Alkyl halides
- Alkyl halides are prepared when primary or secondary alcohols react with PBr3 or PI3. PBr3 and PI3 are produced in situ by the addition of Br2 and I2 to red phosphorus.
3CH3CH2OH + PBr3 → 3CH3CH2Br + H3PO3
3CH3CH2CH2OH + PI3 → 3CH3CH2CH2I + H3PO3
The reaction of alcohol with PX3 or SOCl2 does not involve the formation of carbocation, hence occurs without the rearrangement of the carbon skeleton and elimination. That’s why PX3 or SOCl2 is preferred as a reagent for the conversion of an alcohol to the corresponding alkyl halide.
Mechanism
The reaction involves the initial formation of a protonated alkyl dibromophosphite by a nucleophilic displacement on phosphorus. The alcohol acts as the nucleophile.
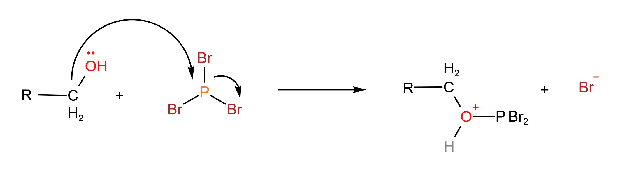
Then a bromide ion acts as a nucleophile and displaces HOPBr2.
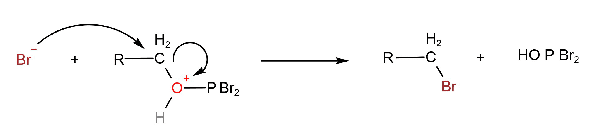
The HOPBr2 can react with more alcohol so that the net result is the conversion of three moles of alcohol to alkyl bromide by 1 mole of PBr2.
- Alkyl halides are prepared when primary and secondary alcohols react with thionyl chloride in the presence of pryidine.
CH3CH2OH + SOCl2 → CH3CH2Cl + SO2 + HCl
Mechanism
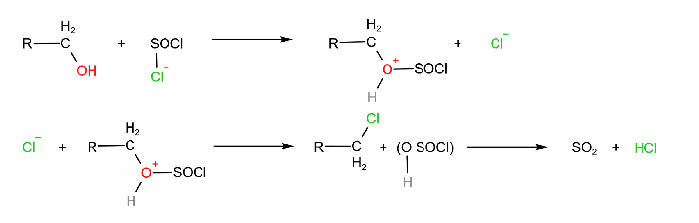
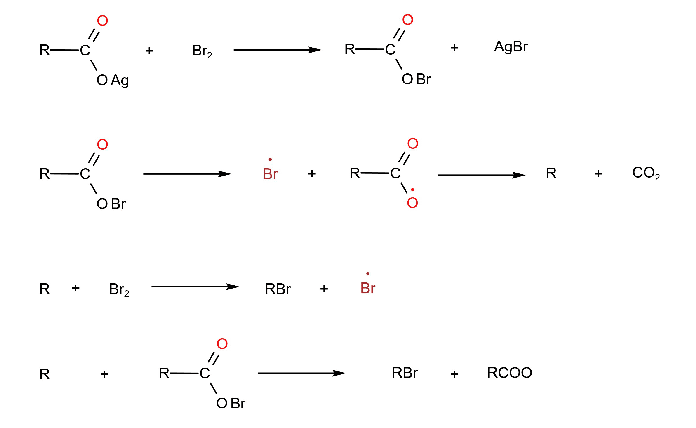
4. From Carboxylic Acid (Hunsdiecker Reaction)
When dry silver salt of carboxylic acid upon refluxing with bromine in the presence of CCl4 gives the corresponding alkyl bromide with the lubrication of carbon dioxide.
RCOOAg + Br2 → RBr + CO2 + AgBr
Mechanism
The reaction involves the initial formation of an acyl hypohalide that decomposes into free radicals which then lead to the products as shown below:
Note:
R and RCOO are free radicals in the above figure.
Bromine gives a good yield and often used halogen, chlorine and iodine have also been used. But this method is used limited because only the primary alkyl halides are obtained in good yields whereas the yield of secondary and tertiary alkyl halides are quite low.
5. By Halide exchange
The alkyl bromide or chloride is react with the solution of NaI in acetone to produce an alkyl iodide. This method is particularly suitable for preparing alkyl iodides.

The less soluble NaBr or NaCl precipitates can be removed from the solution by filtration.
6. Allylic Halogenation
The halides in which the halogen atom is attached to a carbon atom that is adjacent to a double bond are called allylic halides. The carbon atom which is adjacent to a double bond is called allylic carbon. The substituent attached to the allylic carbon is called allylic substituent.
When alkenes react with chlorine or bromine at high temperature gives alkyl halides (chlorides or bromides).
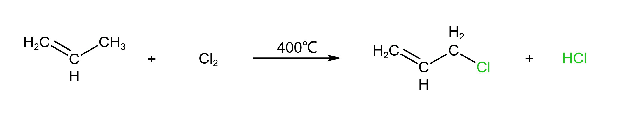
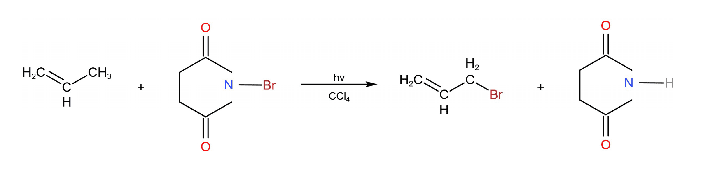
When alkenes react with N-bromosuccinimide (NBS) in CCl4 gives alkyl bromide.
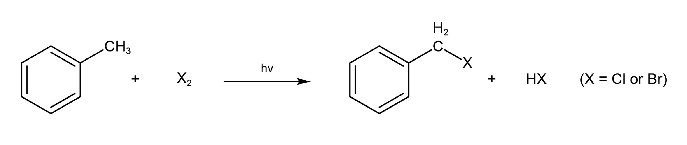
Benzylic halides can be prepared similarly.
7. Chloromethylation
A hydrogen atom of a certain aromatic ring can be replaced by the chloromethyl group – CH2Cl. The reaction is called chloromethylation. Benzyl chloride is formed when benzene is treated with formaldehyde and HCl in the presence of a catalyst such as SnCl2, H2SO4, or H3PO4.

Physical Properties of Alkyl Halides
- Methyl chloride CH3Cl, methyl bromide CH3Br and ethyl chloride CH3CH3Cl are gases at room temperature, whereas other alkyl halides up to C18 are colorless liquids, and alkyl halides having C-atom more than 18 are colorless solids.
- Alkyl halides are soluble in organic solvents but are usually insoluble in water.
- CH2Cl2, CHCl3, and CCl4 are often used as solvents for nonpolar and moderately polar organic compounds.
- The boiling point of alkyl halides increases with an increase in C-atom.
- The melting and boiling point of alkyl halides are greater than those of alkanes with the same number of carbon atoms. This is due to higher molar masses which result in greater van der Waals attractions. Having the same alkyl group, the melting and boiling point increase with the increase in the atomic mass of halogens.
- The density of alkyl halides is greater than the hydrocarbons. For a given halogen atom (Fluoride, chloride, bromide, iodide) density of alkyl halides decreases with an increase in the size of the alkyl group.
- Many chlorohydrocarbons including CHCl3, CCl4 have a comuative toxiciy, therfore, they should be used in fume hoods and with great care.
- Organic halogen compounds are less inflammable than the corresponding hydrocarbons. The inflammability, however, decrases with incrasesing halogen content. Carbon tetrachloride is used as a fire extinguisher wisher under the name “pyrene”.
- Alkyl iodide decomposes in sunlight hence, on standing it becomes violet or brown.
- As we know that bond strength is inversely proportional to bond length. So, the order of bond strength is CH3F > CH3Cl > CH3Br > CH3I.
- The order of bond length is CH3I > CH3Br > CH3Cl > CH3F.
- Decreasing the order of density among alkyl halides is RI > RBr > RCl > RF.
- Stability order is RF > RCl > RBr > RI.
| Bond | Bond Length (pm) | Bond Enthalpy (KJ/Mol) | Boiling Point (K) |
|---|---|---|---|
| CH3F | 139 | 452 | 194.5 |
| CH3Cl | 178 | 351 | 248.8 |
| CH3Br | 193 | 293 | 276.6 |
| CH3I | 214 | 234 | 315.4 |
Halogenation of Unsaturated Hydrocarbons, Alkanes
The replacement of the hydrogen atom of alkane by a halogen group is called halogenation of alkanes. Alkanes react with chlorine or bromine in the presence of sunlight or UV light at high temperatures, it gives alkyl halides (chloride or bromide) or a mixture of products. Some important points are given below:
- The extent of halogenation depends upon the amount of halogen used.
- The reaction of alkanes with fluorine is highly violent and gives a mixture of carbon, fluorinated alkanes, and hydrofluoric acid.
- Iodine does not substitute directly because the reaction is too slow and reversible.
- The order of reactivity of halogens is F2 > Cl2 > Br2 > I2.
- This reaction occurs by a free radical mechanism. In the free radical mechanism, three steps are involved:
- Initiating step
- Chain propagation step
- Chain termination step
Chlorination of Methane
When methane (CH4) is reacted with chlorine in the presence of hv or sunlight or peroxide or high temperature, methyl chloride and HCl is formed.
CH4 + Cl2 → CH3Cl + HCl
This reaction is not stopped if the amount of Cl2 is excess because we know that the extent of reaction depends upon the amount of halogen used. So, if Cl2 is in excess, it again reacts with CH3Cl and gives the product.
Note:
- If Cl2 is in excess amount then the product form is CH2Cl2, CHCl3, and, CCl4.
- If CH4 is in excess amount then only CH3Cl is formed.
When Cl2 reacts with CH3Cl then CH2Cl2 and HCl are formed.
CH3Cl + Cl2 → CH2Cl2 + HCl
When Cl2 reacts with CH2Cl2 then CHCl3 and HCl are formed.
CH2Cl2 + Cl2 → CHCl3 + HCl
When Cl2 reacts with CHCl3 then CCl4 and HCl are formed.
CHCl3 + Cl2 → CCl4 + HCl
Reactions with Mechanisms
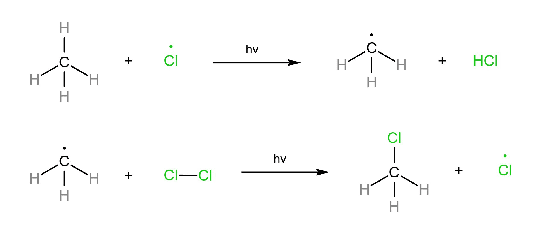
Three steps are involved in the free radical mechanism. Chain Initiation: In chain initiation, Cl2 is broken by homolytic fission in the presence of hv and produced a free radical. This reaction is endothermic. Chain propagation: In chain propagation, free radical forms more free radicals, and this reaction is exothermic except for I2. In the case of I2, this reaction is endothermic. Chain termination: In chain termination, all free radicals react and form different products (major or minor).
Reaction
CH4 + Cl2 → CH3Cl + HCl
Mechanism
Initiation step:
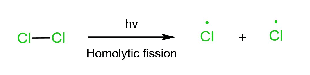
Propagation step:
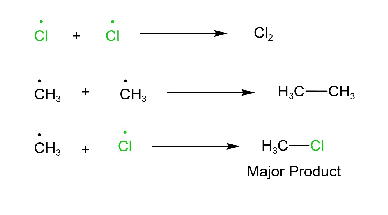
Termination step:
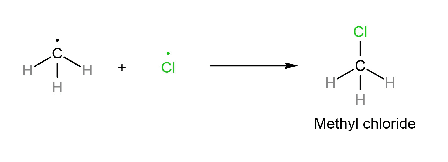
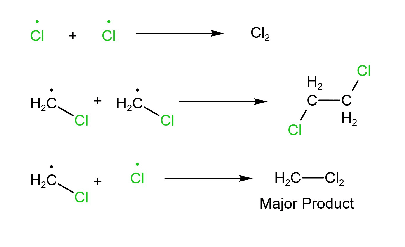
Note: In this reaction, not only methyl chloride is formed but also CH3-CH3 and Cl2 are produced. But the major product is CH3-Cl.
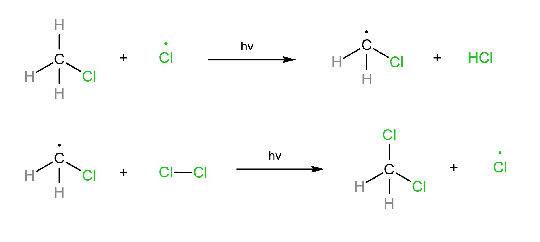
Reaction
CH3-Cl + Cl2 → CH2Cl2 + HCl
Mechanism
Initiation step:
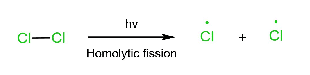
Propagation step:
Termination step:

Note: In this reaction, different products are formed (major or minor).
Reaction
CH2Cl2 + Cl2 → CHCl3 + HCl
Mechanism
Initiation step:
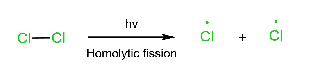
Propagation step:

Termination step:
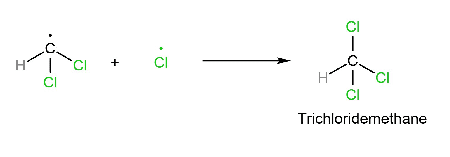
Note: In this reaction, different products are formed (major or minor).
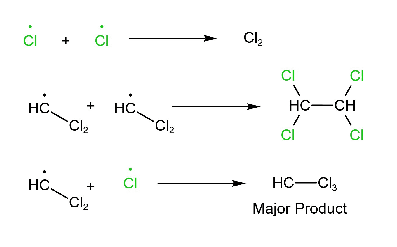
Reaction
CHCl3 + Cl2 → CCl4 + HCl
Mechanism
Initiation step:
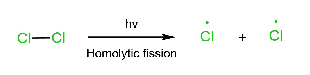
Propagation step:
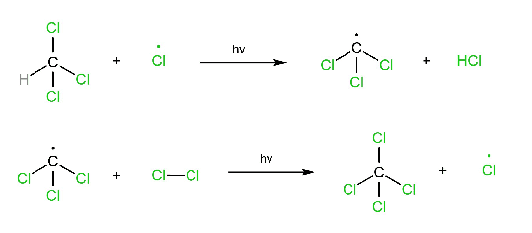
Termination step:
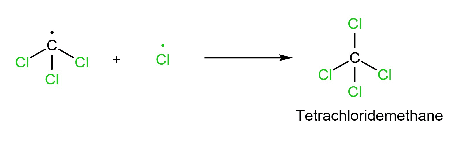
However, polychlorinated can be minimized by using a large excess of methane.
Chemical Reactions of Alkyl Halides
Alkyl halides or haloalkanes are highly reactive compounds due to the polar nature of the carbon halogen bond in their molecules. Alkyl halides undergo several important reactions such as halogenation, nucleophilic substitution reaction, elimination reaction, formation of organometallic compounds such as Grignard reagents, etc.
That’s why alkyl halides are considered one of the most important or valuable reagents in organic chemistry. Some of the most important reactions of alkyl halides are discussed below:
Nucleophilic Substitution Reaction
The reaction in which an atom replaces another atom already present in a molecule is called a substitution reaction. A negative ion or a molecule containing an unshared pair electron that can donate an electron pair to an electron-deficient center in a reaction is called a nucleophile or nucleophilic reagent. Nucleophile means nucleus loving. For example, Cl–, Br–, OH–, CN–, NH3, H2O, etc.
A species that accepts an electron pair from nucleophiles is called an electrophile. Electrophile means electron loving. For example, CH3+, NO2+, etc. Consider a reaction,
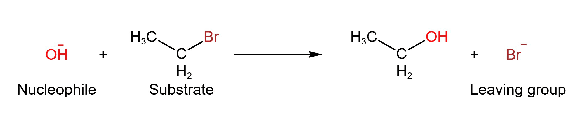
A molecule like CH3CH2Br that is attacked by a reagent is called a substrate, On attacking by a nucleophile, bromide is separate from the substrate is called the leaving group or nucleofuge.
A reaction in which one nucleophile (Nu–) displaces another nucleophile (X–) from combination with an electrophilic carbon atom is called a nucleophilic substitution reaction. This reaction is donated by SN reaction where S stands for substitution and N stands for nucleophilic.

The reagent Nu– is called the attacking nucleophile, R-CH2-X contains the electrophilic carbon atom called substrate, and X– is called leaving group or nucleofuge.
The halogen atom (X) attached to a carbon atom is highly electronegative that’s why the C-X bond in alkyl halides is polar. Thus, the halogen atom carries a partial negative charge, and the carbon atom attached to the halogen carries a partial positive charge and is, therefore, electrophilic. Carbon tends to accept an electron pair from a nucleophile to form a new bond. While making a new bond between a nucleophile and carbon, the electrophilic carbon breaks its old bond with the halogen atom.
The net result of this reaction is the displacement of one nucleophile (Nu–) with another nucleophile (X–) in combination with an electrophilic carbon atom. Such a type of displacement reaction is called nucleophilic substitution reaction.
Mechanism
Alkyl halides undergo nucleophilic substitution by two mechanisms: SN1 and SN2 mechanisms. The mechanism is the path adopted by the reaction. The mechanism is the actual process that tells us about which bonds are broken, which bonds are formed, how many steps are involved in the reaction, and what is the relative rate of each reaction.
SN2 Mechanism
SN2 stands for bimolecular nucleophilic substitution. In the SN2 mechanism, the nucleophile attacks the substrate from the backside, the side directly opposite to the nucleofuge or leaving group. As the reaction proceeds, the bond between electrophilic carbon and nucleophile is formed and the bond between electrophilic carbon and the leaving group is weakened.
In the transition state, both the nucleophile and leaving group are partially bonded to the electrophilic carbon undergoing attack. The hybridization of electrophilic carbon changes from sp3 in the substrate to sp2 in the transition state, and again to sp3 in the product.
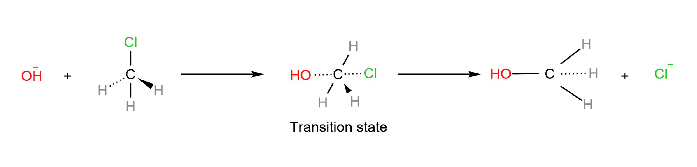
The two molecules (nucleophile or substrate) are involved in the formation of the transition state in the rate-determining step (the only step of the reaction), thus, the reaction is said to be bimolecular.
Kinetic Evidence
The SN2 mechanism involves only a single step and this is the rate-determining step. In this step, both the attacking nucleophile and substrate are involved. Therefore, the rate of reaction is directly proportional to the concentration of nucleophiles and the concentration of substrate and follows the second-order kinetics. According to the rate expression;
Rate ∝ [CH3Cl] [OH–]
Rate = K [CH3Cl] [OH–]
Where K is the specific rate constant.
However, if the solvent itself acts as a nucleophile, the mechanism may still be bimolecular but experimentally, the rate of reaction will be first order. This is because the concentration of solvent remains constant due to its huge amount and as we know that the rate of reaction will depend on the concentration of substrate. Such a reaction is called pseudo-first-order.
H2O + CH3Br → CH3-H2O+ + Br–
Rate = K [CH3Br] [H2O]
Rate = K’ [CH3Br]
Where K’ = K [H2O]
SN1 Mechanism
SN1 stands for unimolecular nucleophilic substitution. This mechanism consists of two steps. In the first step, the substrate ionizes to form a halide ion and a carbonium ion. In the second step, the intermediate carbonium ion rapidly reacts with the attacking nucleophile to form the product.

The slowest step determines the rate of reaction and is known as the rate-determining step. The first step in the SN1 mechanism is usually very slow as compared to the second step, so the first step is the rate-determining step. Only one molecule (substrate) takes part in the rate-determining step in the reaction, therefore, the two-step SN reactions are called unimolecular and are symbolized as an SN1 reaction.
Kinetic Evidence
As we discussed above, in the SN1 mechanism, only the substrate is involved in the rate-determining step, and the reaction is called a unimolecular reaction. Therefore, the rate of reaction is directly proportional to the concentration of substrate, but the rate of reaction is independent of the concentration of the attacking nucleophile. The SN1 reaction is the first-order reaction with the following rate law.
Rate ∝ [substrate]
Rate = K [(CH3)3 Br]
Solvents play an important role in SN1 reactions, since the ionization is assisted by the solvent, it does not enter the rate expression, since it is present in large excess. Mostly the solvent itself acts as a nucleophile and the reaction is generally called solvolysis.
Factor Affacting the Rates of SN1 and SN2 Reactions
Several factors affect the rates of SN1 and SN2 Reactions. The most important factors are:
- The effect of the structure of the substrate.
- The effect of attacking nucleophile (For SN2 reaction only)
- The effect of the solvent
- The effect of the leaving group
| SN1 Reaction | SN2 Reaction |
|---|---|
| SN1 stands for unimolecular nucleophilic substitution. | SN2 stands for bimolecular nucleophilic substitution. |
| SN1 is a two-step mechanism. | SN2 is a one-step mechanism. |
| In the two-step SN1 reaction, the substrate, R-X, first ionizes reversibly into R+ and X- and then the intermediate carbocation reacts with the attacking nucleophile, Nu– to form the product. | The attack of the nucleophile on carbon and the departure of halide ions take place simultaneously in a single step. |
| The rate of reaction depends only on the concentration of the substrate. | The rate of reaction depends on the concentration of the substrate as well as the concentration of attacking nucleophiles. |
| The nucleophile can attack the electrophilic carbon from either side (front or back). | The nucleophile attacks the electrophilic carbon from the backside. |
| It is expected to the first-order kinetics. | It is expected to the second-order kinetics. If however one of the reactants is the solvent, the SN2 mechanism will lead to the first-order reaction. |
| In the SN1 reaction, the product may be partially inverted concerning that of starting substrate, largely inverted or inverted to a very small extent. The product is the racemate usually. | SN2 reactions always proceed with the inversion of configuration. (backside attack) |
| The reaction is favored by polar solvent. | The reaction is favored by a non-polar solvent. |
| The reaction occurs with a weak base. | The reaction occurs with a strong base. |
| The SN1 reaction is generally given by tertiary alkyl halides. | The SN1 reaction is generally given by primary alkyl halides. |
Chemical Properties of Alkyl Halides
Alkyl halides are very important organic compounds. Some of the important nucleophilic substitution, elimination, and miscellaneous reactions of alkyl halides are given below:
Substitution Reactions of Alkyl Halides
Reaction with aqueous KOH
When alkyl halides react with aqueous KOH, form alcohols.

Reaction with Sodium alkoxides
Alkyl halides react with sodium alkoxides to form ethers.

Reaction with NaCN
Alkyl halides react with sodium cyanide to form alkyl cyanides or nitriles.

Reaction with KSH
Alkyl halides react with alcoholic potassium hydrosulphide to form thiols.

Reaction with mercaptides
Alkyl halides react with mercaptides to form alkyl sulfides.
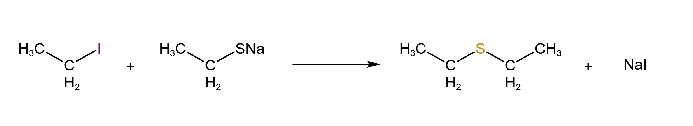
Reaction with RCOOAg
Alkyl halides react with an alcoholic solution of the silver salt of a carboxylic acid yield esters.

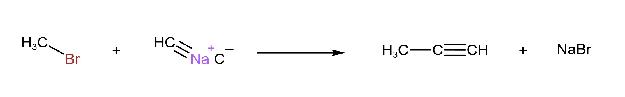
Reaction with Acetylides
Alkyl halides react with sodium acetylides to form higher alkynes.
Elimination Reactions of Alkyl Halides
Reaction with Alcoholic KOH
When alkyl halides are boiling with alcoholic KOH undergo dehydrohalogenation to form alkenes.

Miscellaneous Reactions
Reduction
Alkyl halides are reduced to alaknes by Zn + HI, LiAlH4, and H2 in the presence of Ni or Pd.

Wurtz Reaction
Alkyl halides react with metallic sodium in dry ether to form alkanes.
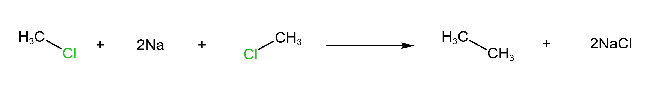
Reaction with Metals
Alkyl halides react with metals in dry ether to form organometallic compounds.

Friedel-Crafts Reaction
When Alkyl halides react with benzene in the presence of anhydrous aluminum chloride AlCl3, alkyl benzenes are formed.

Rearrangement
When alkyl halides is heated in the presence of catalyst like AlCl3, AlBr3 or at about 300℃, it undergoes rearrangement.

Organomagnesium Compounds: Grignard Reagents
Victor Grignard introduced the Grignard reagents as synthetic reagents in organic chemistry. The alkyl magnesium halides or aryl magnesium halides, R-Mg-X or Ar-Mg-X are commonly known as Grignard reagents, and Grignard was awarded the Nobel prize for his work in 1912.
Preparation of Grignard Reagents
A Grignard reagent is prepared when alkyl halides or aryl halides react with magnesium metal in the presence of dry ether. A reaction takes place at the surface of magnesium which slows down during the course of the reaction.
R-X + Mg → R-Mg-X
This reaction is take place in two steps. In the first step, alkyl halides react at the surface of magnesium metal to produce an alkyl radical and MgX radical species. After that in the second step, these two radicals react with each other to form Grignard reagents.
RX + Mg → R‘ + ‘MgX → RMgX
Grignard reagents cannot be isolated. The formation of Grignard reagents depends upon the following two important factors:
- Nature of alkyl or aryl group, and
- the halogen atom
An increase in the number of carbon atoms in the alkyl group makes the formation of the Grignard reagent rather difficult and for a particular alkyl group the ease of formation follows the order: I > Br > Cl. Alkyl bromides are most suitable for the preparation of Grignard reagents. It is because aryl or alkyl magnesium fluoride is not known. Alkyl iodides are generally expensive.
A number f other solvents like tetrahydrofuran (THF) and tertiary amines may also be used as solvents. However, the THF is the best solvent because it increases the reactivity of organic halides toward magnesium.
Reactions of Grignard Reagents
The C-Mg bond in Grignard reagents is covalent in nature but highly polar. The electronegativity of the carbon atom is greater than Mg. The electrons of the C-Mg bond move towards the carbon atom. As a result, the magnesium atom has a partial positive charge and the carbon atom has a partial negative charge.

The characteristics reactions of Grignard reagents are nucleophilic substitution and nucleophilic addition reactions. The alkyl group in Grignard reagents being electron rich can act as carbanions or nucleophiles. A carbon atom that has a negative charge is called carbanion. The following reactions of Grignard Reagents illustrate the synthetic importance of Grignard reagents.
1. Reaction with Compounds containing Active Hydrogen
Hydrogen that is acidic in nature is called active hydrogen. Compounds like alcohols, amines, water, carboxylic acids and terminal alkynes which contain active hydrogen react with Grignard reagents to form hydrocarbons.
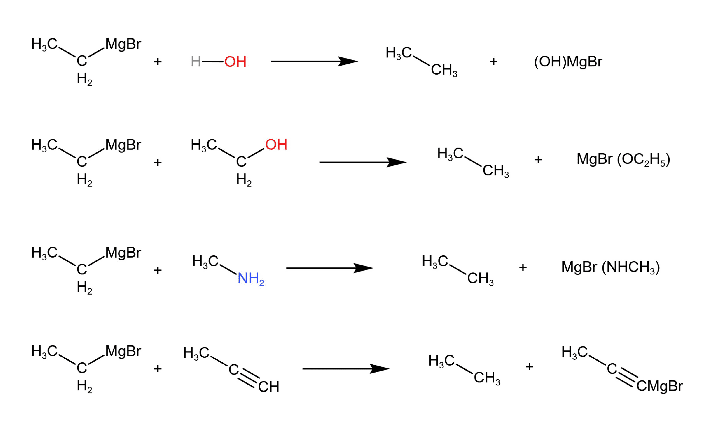
The reaction of Grignard reagents with active hydrogen compounds can be used for the determination or identification of active hydrogens in a compound. This process is known as Zerewitnoff active hydrogen determination.
2. Reaction with Acetaldehyde

3. Reaction with Ketone

4. Reaction with ethylene oxide
Grignard reagent reacts with ethylene oxide to give an additional product which on hydrolysis gives primary alcohols.

5. Reaction with Halogenated ethers

6. Reaction with Oxygen
Grignard reagents react with oxygen to give either hydroperoxides or alcohols. The reaction can be used to convert alkyl halides to alcohols without a side reaction.

7. Reaction with Sulphur
The grignard reagent reacts with sulfur to give a product which on hydrolysis gives the corresponding thialcohol.

8. Reaction with Cyanogen chloride

Uses of Alkyl Halides
Ethyl chloride
- Ethyl chloride was used to produce tetraethyl lead which was an anti-knock additive for gasoline.
- In industry, it was used to synthesize ethyl cellulose which is a thicking agent used as a binder in paints, and cosmetics.
- This is used to prevent pain caused by injections and minor surgical procedures and also used for temporary relief of minor sports injuries.
- It also helps to relieve deep muscle pain.
Chloroform
- Chloroform is used as an anesthetic but it may cause cardiac and respiratory failure so it has been replaced by another safe anesthetic.
- It is used as a cleansing agent in fire extinguishers and in the rubber industry.
- It is also used as a laboratory agent for testing 1-degree amines.
Trichloroethylene
- It is used as a solvent for a variety of organic materials.
- It is used to extract vegetable oils from plants.
- It is used for decaffeination from coffee.
- It is used as a cleaning agent to clean metal parts.
Tetrachloroethylene
- It has non-flammable properties so used as a dry-cleaning solvent.
- TCE is used for testing in the coal industry.
- TCE is used to clean prints and negatives of cinema films.
- TCE is used in typewriter correction fluid.
Dichloromethane
- It is used as a solvent due to its ability to dissolve a wide range of organic compounds.
- In the food industry, it is used to decaffeinate coffee and tea.
- DCM is used in material testing in the field of civil engineering.
Tetrachloromethane
- It is used as a solvent in many organic compounds.
- It is used to remove hool worms from the intestine of infected humans.
Iodoform
- It is used as disinfection and antiseptic.
- It was also used for sterilizing the instruments which were used for surgery.



Leave a Reply