
Fluorine is the most electronegative element in the periodic table. Fluorine does not occur free in nature due to its reactivity. Fluorspar CaF2, Cryolite Na3Al F6, and, Fluorapatite [3Ca3(PO4)2.CaF2] are some important ores of fluorine. Due to the small atomic size, low bond energy, high electronegativity and, absence of 2d orbitals, fluorine shows variation in properties as compared to other halogens like Cl, Br.
Fluorine is the most reactive of all other elements. The ionic character of fluorine is also maximum and forms the most stable salts like HF. It is a strong oxidizing agent and does not form polyhalides but form monovalent compounds (F3- does not exist).
The discovery of fluorine was much later than another halogen family because of its high reactivity (even react with glass container) and low oxidation potential. Hydrofluoric (HF) is a very stable, poisonous, corrosive, and bad conductor of electricity.
History of its Isolation:
In 1771, Scheele obtained an acid solution by heated fluorspar CaF2 with conc. H2SO4 in a tin retort. He concluded that fluorspar is the calcium salt of this acid solution which he named fittoric acid. In 1809, Gay Lasac and Thenard prepared this acid and concluded that the acid shows resemblance with hydrochloric acid HCl in many properties which he named hydrofluoric acid. In 1813, Day showed that an unknown element combines with hydrogen and form hydrofluoric acid and an unknown element which he named fluorine.
By the electrolysis of an aqueous solution of hydrofluoric acid, Davy tried to prepare fluorine just as chlorine is prepared by the electrolysis of an aqueous solution of hydrochloric acid HCl. But instead of fluorine, he obtained H2 and ozone mixed with O2 at the anode.
After that, Gore attempted to electrolyze the anhydrous hydrofluoric acid to isolate the fluorine from it, but he realizes that this acid is a nonconductor and hence failed to isolate fluorine from it. In 1860, he showed that the anhydrous hydrofluoric acid dissolves in KHF2 and gives a conducting solution. But the problem is here that the great chemical reactivity of fluorine. It reacts with apparatus whether made of glass, platinum, and carbon. When the platinum vessel was used, fluorine reacts with it and form chocolate powder of PtF4 while with carbon it gave the gaseous CF4.
In 1886, the problem was finally solved by Moission. He prepared fluorine by electrolyzing a solution of KHF2 (1 part) in anhydrous hydrofluoric acid (5 parts) in a U-tube made of Pt-Ir alloy using electrodes of the same alloy. By keeping the bath, he decreased the great chemical reactivity of fluorine and collecting the gas at a very low temperature.
Difficulties Encountered in the Discovery and Isolation of Fluorine
For a long time, a number of difficulties were encountered in the discovery and isolation of fluorine. The two main causes for the late discovery and isolation of fluorine is:
- Extreme reactivity of fluorine
- Greater stability and non-conducting behavior of hydrofluoric acid
Extreme Reactivity of Fluorine
Fluorine is so reactive due to its high electronegativity that it reacts with the material of all vessels like F2 react with carbon and silicon present in the vessel and form CF4 and SiF4 respectively. Similarly, F2 also react with electrodes and vessel made up of Pt and form a chocolate-colored powder, PtF4. Lead (Pb), iron (Fe), and, Tin (Sn) vessels were also used but found to be unsuitable for the preparation of F2. Thus, non-metal could be used as a vessel or as an electrode for the preparation of F2.
Greater Stability and non-conducting nature of Hydrofluoric acid
Hydrofluoric acid is very stable, poisonous, and corrosive in nature. It is so stable that all attempts to isolate fluorine from hydrofluoric acid by oxidizing agents failed. Moreover, electrolysis of an aqueous solution of HF gives H2 and ozone mixed with oxygen while anhydrous hydrofluoric acid HF is a non-conductor.
Occurrence
As we discuss above, fluorine is very stable and hence does not occur free in nature. Fluorine is widely found in combined states like fluorides. The chief minerals of fluorine are Fluorspar CaF2, Cryolite Na3Al F6, and, Fluorapatite [3Ca3(PO4)2.CaF2]. It is also present as fluoride in soil, river water plants, plant-ash, seawater, bones, and teeth of animals. One liter of seawater contains 2 mg of fluorine. Fluorine is also present in phosphate rocks. Fluorapatite CaF2 is the principal or major source of fluorine and its compounds.
Physical Properties
The physical properties of fluorine can usually be measured or observed using our senses or involve numerical measurements.
- It’s a pale yellow gas
- Atomic number: 9
- Atomic mass: 18.998
- Oxidation states: -1
- Electronegativity: 3.98
- Electron affinity: 328 KJ/mol
- Fluorescence: It can sometimes be fluorescent
- Phase changes: Fluorine can be changes to F(g) ⇌ F(l)
- Solubility: In liquid form it is freely soluble in liquid oxygen and ozone.
- Density: 1.8×10-3 g/cm3 at 20℃
- Melting point: -219.6℃
- Boiling point: -188℃
- Atomic radius: 64 pm
- Isotopes: 17 known isotopes, but only F18.998 [F19] is stable and naturally occurring.
- First ionization potential: 1680.6 KJ/mol
- Second ionization potential: 3134 KJ/mol
- Third ionization potential: 6050 KJ/mol
- Standard reduction potential: +2.87 v
Anomalous Properties of Fluorine
Fluorine differs from the rest of the members of the halogens. This is due to:
- Smaller size: The fluorine atom has a small covalent radius of around 64 picometers.
- High electronegativity: Fluorine is the most electronegative element in the periodic table (E.N value = 4).
- High ionization enthalpy: The first ionization enthalpy of fluorine is 1681 KJ/mol.
- Non-availability of valance shell d-orbitals for bonding purposes. Hence, the covalency of fluorine rarely exceeds one.
- Fluorine is the most reactive of all the halogens due to the lower value of F-F bond dissociation energy. (F2=158KJ/mol)
- Fluorine is most electronegative and it shows a -1 oxidation state and does not show any positive oxidation state due to the absence of d-orbital in its valance shell. The other halogens show a positive oxidation state of +1, +3, +5, +7, etc.
- Due to the small atomic size and high electronegativity of fluorine, HF undergoes strong hydrogen bonding as compared to other halogen acids.
- Due to the high strength of H-bonding in HF, HF is a liquid at room temperature while other halogen acids are gases at room temperature.
- HF is the weakest of all halogen acids due to the high strength of the H-F bond.
- The salt of HF is different in several aspects. AgF is soluble in water while all other silver halides (AgCl, AgBr, AgI) are insoluble in water. But CaF2 is insoluble in water while other halides (CaCl2, CaBr2, CaI2) are soluble in water.
- HF can not be stored in glass bottles since it reacts with silicates to form fluorosilicates. While other halogens do not react with silicates and hence can be stored in glass bottles.
- Fluorides have the maximum ionic character, for example, AlF3 is ionic while other halides of Al are covalent in nature.
- Of all the halogens, fluorine has the highest positive electrodes potential (2.87 v) and it is most easily reduced and hence acts as the strongest oxidizing agent.
- It brings about the highest oxidation of other elements with which it combines. For example, with S it gives SF6, with I2 it gives IF7. Other halogens do not always bring about a higher oxidation state. For example, with S chlorine gives SCl4, Bromine gives SBr2, I2 does not react at all. F2 is so powerful oxidizing agent that it can even oxidize inert gases.
- Due to the absence of d-orbitals, fluorine does not form polyhalide ions while other halogens form polyhalides of the type I3–, Br3–, I5– etc.
Chemical Properties
- Toxicity: Highly poisonous in elemental form
- Oxidation: Fluorine does not combine with oxygen at room temperature.
- Compounds: The importance of fluorine lies largely in its compounds.
- Corrosion: Highly corrosive gas
- Reactivity with water: When mixed with water, react explosively.
- Reactivity with elements: Reacts with all elements except helium, neon and argon.
- Reactivity with heat: Heat resistant
- Fluorine is flammable
1. Reactivity
Fluorine is the most reactive element among halogens. The valance shell electronic configuration of fluorine is 2s2, 2p5 (7 valance electrons). Fluorine gains one electron and achieves the noble gas configuration and gets stability.
2. Action on Halides
The standard reduction potential value of fluorine is maximum among all the halogens corresponding to the reaction:
X2 + 2e– → 2X–
Hence, F2 can displace all the halogens from their halides i.e. F2 liberates Cl2 from chlorides, Br2 from bromides, and I2 from iodides.
3. Reacts with Elements
Fluorine reacts with most of the metals (Na, Ca, Al, Mg, Ni, etc.), non-metals (H, C, P, Cl, Br, I, etc.), and metalloids (As, Sb) to form the fluorides.
4. Reacts with Xenon
Fluorine reacts with xenon (noble gas) to form fluorides like XeF2, XeF4, XeF6. The final product depends upon the mixing ratio of the constituents, pressure, and temperature.
5. Action of H2O
When fluorine reacts with water it gives O2 and O3.
2F2 + 2H2O → 2HF + O2
3F2 + 3H2O → 6HF + O3
Simultaneously, oxygen fluoride OF2 is also formed.
6. Oxidizing action
Fluorine acts as an oxidizing agent when it reacts with water and giving oxygen and ozone. It oxidizes chromic salts (Cr=+4) to dichromates (Cr=+6), bisulfates to persulphates, chlorates to perchlorates, iodates to periodates and, halides to halogens.
KClO3 + F2 + H2O → KClO4 + H2F2
2KHSO4 + F2 → 2HF + K2S2O8
7. Action on Glass and Quartz
Fluorine readily reacts with glass and quartz forming SiF4. That’s why hydrofluoric acid can not be stored in glass bottles.
8. Action of Alkalies
Fluorine does not form oxy salts with alkalies and no oxyacids of fluorine are known. Oxygen difluoride OF2 is formed when fluorine reacts with dilute alkalies. But when F2 reacts with conc. alkalies, O2 is formed.
2F2 + 2NaOH (2% solution) → OF2 + 2NaF + H2O
2F2 + 4NaOH (conc.) → O2 + 4NaF + 2H2O
9. Action of NH3 and H2S
2NH3 + 3F2 → N2 + 6HF
NH3 + 3F2 → NF3 + 3HF
H2S + 4F2 → SF6 + 2HF
Xenon fluorine compounds
Xenon forms three binary fluorides XeF2, XeF4, and XeF6 by using the direct reaction of elements under appropriate experimental conditions.
Xenon Difluorine XeF2
When xenon is in excess, XeF2 is prepared by heating xenon and fluorine in a nikal tube at 673K and 1bar pressure.
Xe(g) + F2 → XeF2 (S)
It can be obtained by irradiating a mixture of Xe and Fluorine with light from a high-pressure mercury arc. It can be made by the action of dioxygen difluoride on xenon at -118℃.
Xe + O2F2 → XeF2 +O2
Xenon tetrafluoride XeF4
XeF4 is prepared by heating a mixture of xenon and fluorine (1∶5) in a nikal vessel at 873K and 7bar pressure.
Xe(g) + 2F2 → XeF4 (s)
when XeF4 react with xenon, XeF2 produces.
XeF4 + Xe → 2XeF2
Xenon hexafluoride XeF6
XeF6 is prepared by heating a mixture of xenon and fluorine (1∶20) in a nikal vessel at 573K and 70bar pressure.
Xe(g) + 3F3 → XeF6 (S)
XeF6 can also be prepared by the interaction of XeF4 and O2F2 at 143k.
XeF4 + O2F2 → XeF6 + O2
XeF2, XeF4, and XeF6 are colorless crystalline solids and sublime readily at 298K. They are powerful fluorinating agents. They are hydrolyzed even by traces of water. For example, XeF2 is hydrolyzed to give Xe, HF, and O2.
2XeF2 (S) + 2H2 O → 2Xe(g) + 4HF(aq) + O2 (g)
The structure of three xenon fluorides can be deduced from the VSEPR theory. XeF2 and XeF4 have linear and square planner structures respectively. XeF6 has six bond pairs and one lone pair thus, have a distorted octahedral structure.
Xenon fluorides react with fluoride ion acceptors to form cationic ions and fluoride ion donors to form fluoroanions.
XeF2 + PF5 → [XeF]+ + [PF6]–
XeF4 + SbF5 → [XeF3]+ + [SbF6]–
XeF6 + MF → M+ + [XeF7 ]– (M= Na, K, Rb, Cs)
Isolation of Fluorine
Different methods are used for the isolation of fluorine are given below:
1. Moissan’s Method
In 1886, Moissan has used a method first time to isolate fluorine known as Moissan’s method. The apparatus used by Moissan are:
a) Electrolytic cell
The electrolytic cell consists of a U-shaped tube made up of Pt-Ir alloy cooled at -23℃ by placing it in a bath of CH3Cl (boiling pt. -23℃).
b) Electrodes
Electrodes are made up of Pt-Ir alloy and these are insulated with fluorspar stoppers covered with shellac.
c) Electrolyte
The solution of potassium hydrogen fluoride, KHF2 in anhydrous hydrofluoric acid, H2F2 by the ratio of 1∶5 is used for electrolyte.

Process
In U-shaped electrolytic cell, Moissan took the solution consisting of KHF2 and anhydrous H2F2 (1∶5 ratios) at a temperature of -23℃ by placed the cell in a bath of CH3Cl and electrolyzed the solution. On electrolysis, hydrogen H2 was produced at the cathode and F2 at the anode which is then passed through a platinum spiral tube placed in the bath of CH3Cl.
The vapors of hydrofluoric acid got condensed in the platinum spiral tube. Fluorine gas then passed through a tube containing fused sodium fluoride, NaF which absorbed even the last traces of H2F2 forming NaHF2. The fluorine is obtained by this method is pure.
2KHF2 (In anhydrous H2F2) → 2KF + H2F2
H2F2 → 2H+ + 2F–
At Anode
F– → F + e–
F + F → 2F
At Cathode
H++ e– → H
H + H → 2H
2NaF + H2F2 → 2NaHF2
In spite of the low temperature of -23℃, about 5 grams of platinum was a waste for the preparation of 1 gram of fluorine. In 1899 he observed that copper tube could be used instead of a Pt-Ir tube since the copper tube gets coated with a protective layer of CuF2 which prevents further action of fluorine on the cupper, but he continued the use of Pt-Ir electrodes.
2. Deniss Method
In 1932, this method of isolation of fluorine was devised by Deniss, Veeder, and Rockow. The apparatus used in this method are:
a) Electrolytic cell
The electrolytic cell consists of a V-shaped tube made up of copper about 2″ wide and fitted with copper caps into which graphite electrodes are fixed with bakelite cement. The V-shaped Cu-tube is thickly coated with an insulating layer of asbestos cement. For heating the V-shaped tube electrically, this layer is also wound by a resistance wire. The overall apparatus is then thickly lagged to prevent heat loss.
b) Electrolyte
The electrolyte used in this method is pure and dry KHF2. The melting point of KHF2 is 227℃. KHF2 should be pure and perfectly dry, since the presence of traces of moisture results in the formation of O2 and OF2. KHF2 is mostly dried in a current of air at 140 to 150℃.
c) Electrodes
Graphite electrodes placed into the V-shaped Cu-tube by bakelite cement are used.

Process
H2 is liberated at the cathode and F2 at the anode when a current of 5 amperes and 12 volts is passed through a perfectly dry and pure KHF2 electrolyte. F2 so produced attacks the copper walls forming the layer of CuF2 which protects the deeper layers of copper from further attack. The reaction takes place are:
2KHF2 → 2KF + H2F2
H2F2 → 2H+ + 2F–
At Cathode
2H+ + 2e–→ H2
At Anode
2F– → 2F + 2e–
F2 further passes through copper tubes containing dry NaF which absorb HF and forming NaHF2.
2NaF + H2F2 → 2NaHF2
Disadvantages
The following are the disadvantages of this method:
- H2 and F2 mixed together and may cause explosion.
- F2 is evolved slowly due to narrow exit around the anode which may cause frothing of the electrolyte and thus may prevent the escape of F2.
3. Whytlaw-Gray’s Method
A copper cell wound with resistance wire for electrical heat is used as apparatus in this method. It also acts as cathode resistance and has an outlet for H2 near the top.

A rod of graphite acts as the anode enclosed in a copper cylinder. This cylinder is perforated near the bottom and has an outlet near the top for the removal of F2 gas. The electrolyte is fused with Potassium hydrogen fluoride (KHF2). After electrolysis, it gives hydrogen and fluorine.
2KHF2 → 2KF + F2 + H2
The anode and the cathode are separated by a cylindrical diaphragm. Fluorine and hydrogen gas escapes out of the cell without any possibility of mixing with each other.
Precautions
- The electrolyte must be absolutely dry, because if it is not, the fluorine may react with it to produce oxygen and ozone. The presence of moisture may hinders electrolysis.
- All the part of apparatus must also be free from oil and grease.
- The gas must be freed from HF by using NaF. On the other hand, if HF is present, it will attack the glass vessels in which the fluorine is kept.
- The vessels must also be absolutely dry in which the fluorine is collected.
- As fluorine attacks organic matter of all types, it should be very carefully handled.
Advantages of this method
- The anode and cathode are separated by copper cylindrical diaphragm with the result that the liberated H2 and F2 are avoided to mixing with each other and thus the possibility of explosions is avoided.
- There is no chance of explosion due to the frothing of the electrolyte by the accumulation of gases.
4. Modern Method
In this method, the preparation of F2 consists of electrolyzing fused KHF2 mixed with 2 to 3 moles of H2F2 at 70-80℃ in a rectangular steel vessel using a current o 1000-2000 amperes at 8.5 to 11 volts. The steel vessel act as an anode and the cathode was made up of graphite form of carbon. Now anode of petroleum coke impregnated with copper is preferred.
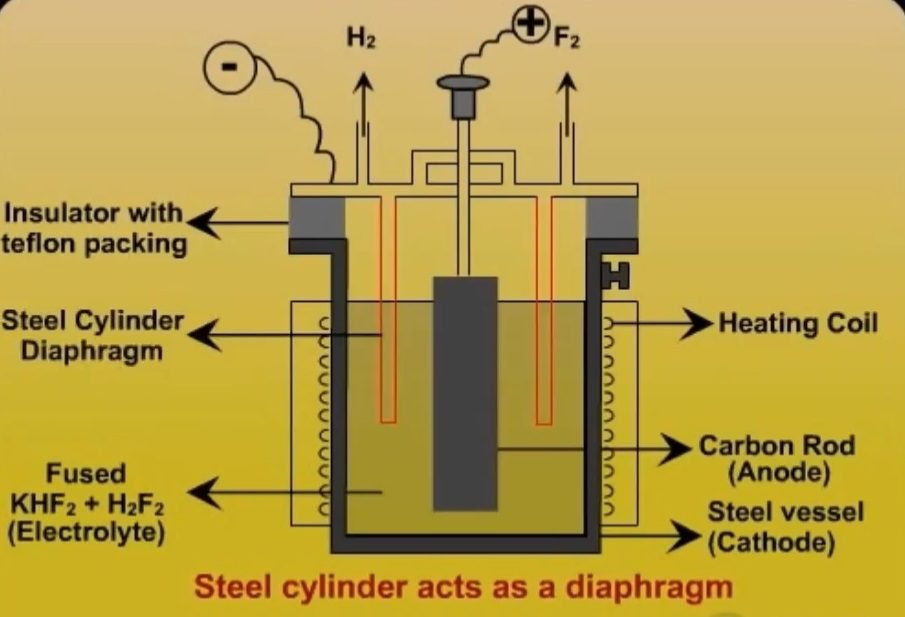
A steel cylinder attached to the lid act as a diaphragm which does not allow fluorine and hydrogen to mix with each other and thus the two gases H2 and F2 are led out through separate exits. And the possibility of explosion is avoided. The fluorine obtained at the anode is freed from H2F2 by passing it through dry NaF and is stored in steel cylinders.
Fluorocarbons
When fluorine F2 reacts with hydrocarbons explosively, fluorinated hydrocarbons like CHF3, CF4, CH2F2, C2F8, C3F3 are formed which are called fluorocarbons. The formation of fluorocarbons has been successfully carried out by diluting fluorine with nitrogen N2.
Synthesis
The fluorocarbons are synthesized by:
1. By the replacement of halogen atom (other than F) in alkyl halides by P-atom in metal fluoride
RCl + AgF → AgCl + RF
CH3Cl + AgF → AgCl + CH3F
C6H5PCl3 + AsF3 → AsCl3 + C6H5PF3
2. By the replacement of halogen atom by F-atom in anhydrous HF
CCl4 + HF → CCl3F + HCl
CCl3F + HF → CCl2F2 + HCl
CCl2F2+ HF → CClF3 + HCl
CClF3 + HF → CF4 + HCl
CHCl3 + 2HF → CHCl2F + 2HCl
Chloro-fluorocarbons obtained from CCl4 are called freons. The catalyst used for the above reactions is SbCl5 at 50-150℃ and pressure of 50-500 lb. inch-2.
3. By electrolytic replacement of H2 by F2
In this method, organic compounds are subjected to electrolysis in liquid HF at 4-6 volts below that required for the lubrication of F2. Steel cells are used with nickel anode and steel cathode. Fluorination is taken place at the anode. This method has been used in laboratories as well as in industries.
(C2H5)2O → (C2F5)2O
CH3COOH → CF3COOF + H2O → CF3COOH
4. By the direct replacement of H2 by F2
The F2 and reacting compounds are subjected to electrolytic fluorination in a nitrogen atmosphere. Cu-gauze, silver-coated copper gauze, or cesium fluoride are used as catalysts.
C6H6 + 9F2 → C6F12 + 6HF
Properties and Uses of Fluorocarbons
- Chloro-fluorocarbons obtained from CCl4 is known as freons. Freons are used as non-toxic, aerosol bomb propellants and heat transfer, non-corrosive inert refrigerants and fire extinguishing agents. Freons are very inert chemically. This property makes them useful solvent, insulators and lubricants.
- Difluorodichloromethane (CF2Cl2) which are also called freon-12. Freon-12 is used in refrigeration and air conditioning in place of ammonia and Sulphur dioxide. It id non-flammable and non-toxic. It is also used in insecticides and as a solvent for D.D.T.
- Tetrafluoroethylene, C2F4 can also be polymerized to from poly tetrafluoroethylene, (C2F4)n. This polymerized compound is known as Teflon. Teflon is plastic like inert material. It is insoluble in any solvent. Teflon is not attacked by strong alkalies, acids and oxidizing agents. Teflon is also used in the construction of chemical plants. It is used as an insulating material in cables and it has very high electrical resistance.
- By the fluorination of CH3OH or CO by F2 in the presence of AgF2, CF3OF is formed. It has been used as strong oxidizing agent and It is stable compound up to 50℃.
CF3FO + SO2 → CF3OSO2F
CF3FO + SO3 → CF3OOSO2F
CF3OF + SF4 → CF3OSF5
Hydrofluoric Acid, H2F2 – The Halogen acid
All the halogens react with hydrogen and form covalent hydrides of HX type where X is the halogens (F, Cl, Br. I) which shows an oxidation state of -1.T These hydrides are known by various names like hydrogen acids, hydrohalic acids, hydrogen halides, hydracids.
Preparation
Pure anhydrous acid is obtained by distilling dry KHF2 in a copper, platinum, or steel vessel cooled in the freezing mixture (laboratory preparation) or bypassing dry H2 over dry AgF.
2KHF2 → H2F2 + 2KF
2AgF + H2 → 2Ag + H2F2
An aqueous solution of H2F2 is prepared by heating calcium fluoride, CaF2 (fluorspar) with 90% H2SO4.
CaF2 + H2SO4 → H2F2 + CaSO4
Storage of the Acid
The anhydrous acid is stored in well-cooled platinum, silver, or gold vessels or in steel containers. The dilute commercial aqueous acid is kept in gutta-percha bottles but when concentrated it is kept in ceresin wax bottles.
Physical Properties of H2F2
- Anhydrous hydrofluoric acid is a colorless, strongly fuming liquid.
- Boiling point: 194℃
- Melting point: -83℃
- It is dangerous to handle and extremely poisonous.
- It forms ulcerated sores when it is in contact with the skin.
- It is highly soluble in water.
- Anhydrous acid is a bad conductor of electricity.
- At low temperatures, the liquid acid and its vapors consist of polymeric form (HF)x whereas at 90℃ the density of the vapour has the normal value.
Chemical properties
1. Combustibility
H2F2 is neither the combustible nor the supporter of the combustible.
2. Acidic Nature
It is a weak dibasic acid. H2F2 is weaker than H3PO4 but stronger than HNO2. Hence, it forms two types of salts namely normal salts (like NaF) and acidic salt (like NaHF2).
3. Action on Metals
Anhydrous acid does not attack any metals under ordinary conditions except potassium but on the other hand, an aqueous concentrated solution of the acid reacts most of the metals like Na, Ag, Ca, Zn, Mg forming their fluorides with the liberation of hydrogen gas.
4. Action on Oxides, Carbonates, Hydroxides
The concentrated aqueous solution of the acid reacts with oxides, carbonates, and hydroxides to form the fluorides.
B2O3 + 3H2F2 → 2BF3 + 3H2O
2NaOH + H2F2 → 2NaF + H2O
CaCO3 + H2F2 → CaF2 + H2O + CO2
When the concentrated aqueous solution of the acid reacts with silica (SiO2), H2SiF6 is formed.
SiO2 + 3H2F2 → H2SiF6 + 2H2O
Since glass contains SiO2, hydrofluoric acid cannot be kept in a glass container.
5. Action on Glass
Pure anhydrous acid does not react with glass but a concentrated aqueous solution of the acid attacks glass. H2F2 reacts with silica, calcium silicate, and sodium silicate which are the constituent of glass.
SiO2 + 3H2F2 → H2SiF6 + 3H2O
Na2SiO3 + 3H2F2 → Na2SiF6 + 3H2O
CaSiO3 + 3H2F2 → CaSiF6 + 3H2O
The etching of glass is based on the above reactions. For this purpose commercial H2F2 is used.
6. Action of AgNO3 and BaCl2 solution
When H2F2 reacts with AgNO3 solution, it gives no precipitate. Since AgF formed is soluble in water. With the reaction of BaCl2 solution, it gives a white precipitate of BaF2 which is insoluble in dilute HNO3 but soluble in conc. HCl.
H2F2 + 2AgNO3 → 2AgF + 2HNO3
H2F2 + BaCl2 → BaF2 + 2HCl
7. Stability
H2F2 is extremely stable and the thermal stability is due to the strength of the H-F bond in the H2F2 molecule. It is not oxidized by strong oxidizing agents or decomposed by heat.
Uses of Hydrofluoric Acid
Hydrofluoric acid is used in the formation of inorganic fluorides, fluorohydrocarbons and is also used in catalysis.

First, we see how is it used in the production of inorganic fluorides?
Inorganic Fluorides
Inorganic fluorides such as:
- Aluminum fluoride
- Uranium tetrafluoride
- Sodium fluoride
- Boron trifluoride
- Ammonium hydrogen fluoride and aqueous solutions of hydrogen fluoride
These inorganic fluorides are used in:
- the etching and polishing of glass
- metal cleaning
- electronic manufacturing
- pickling of steel
- manufacture of semi-conductors
- removing rust
- herbicides
Fluorohydrocarbons
Organofluoro-compound: Organic compounds that contain the carbon-fluorine bond. The C-F bond is found in:
- Pharmaceuticals
- agrichemicals
- fluoropolymers
- refrigerants
- surfactants
- anesthetic
- oil-repellents
- catalysis
- water repellents
Catalysis
Hydrofluoric acid is used as a catalyst in various reactions. For example:
- In alkylation reactions e.g. the dimerization of isobutene. This process is increasing in importance due to the rising demand for highly knock-resistant fuels.
Comparison of H2F2 with other Halogen Acids
1. Acid Strength
Hydrofluoric acid, H2F2 is the weaker acid of all the acids. It is because H2F2 ionizes slightly in an aqueous solution but other acids ionize completely.
2. Ionic Character of H-F bond
F atom has the highest electronegativity of all the halogen atoms. So, the H-F bond in H2F2 acid has the highest degree of polarity.
3. Physical state
At ordinary temperature, H2F2 is a liquid while other halogen acids are colorless gases.
4. Reducing property
H2F2 has no reducing property while other acids are good reducing agents.
5. Action on silica or glass
A strong solution of H2F2 reacts with silica or glass readily forming SiF4 but other halogens do not attack silica or glass.
6. Action on metals
All other halogen acids react with metals like Mg and Zn to form H2. The action of H2F2 on metals is less volatile than other halogen acids.
7. Action of a mixture of MnO2 and H2SO4
When hydrofluoric acid, H2F2 is heated with a mixture of H2SO4 and MnO2, no gas is evolved. On the other hand, in the case of HCl, HBr, and HI acids, Cl2, Br2, I2 are produced having the colors greenish-yellow, reddish-brown vapors, and violet vapors respectively.
8. Action of Pb(CH3COO)2 solution
With Pb(CH3COO)2 solution, H2F2 gives no precipitate while other halogen acids give the precipitate of PbCl2 (white), PbBr2 (white), and PbI2.
9. Dissociation
H2F2 does not dissociate on heating but on the other hand, other halogen acids dissociate at definite temperatures.
Similarities between HF and H2O
- Both the HF and H2O resemble each other in fusibility, solubility, etc.
- Like water, H2F2 is a weak acid and both are ionizing solvents.
- Both form associated molecules, (HF)x and (H2O). the formation of such molecules is due to the H-bonding in HF and water molecules.
- The heat of the formation of water and H2F2 are close to each other.
MOT Diagram of Fluorine F2
σ1S2, σ*1S2, σ2S2, σ*2S2, (π2Px2=π2Py2) σ2Pz2, (π*2Px2=π*2Py2) σ*2Pz
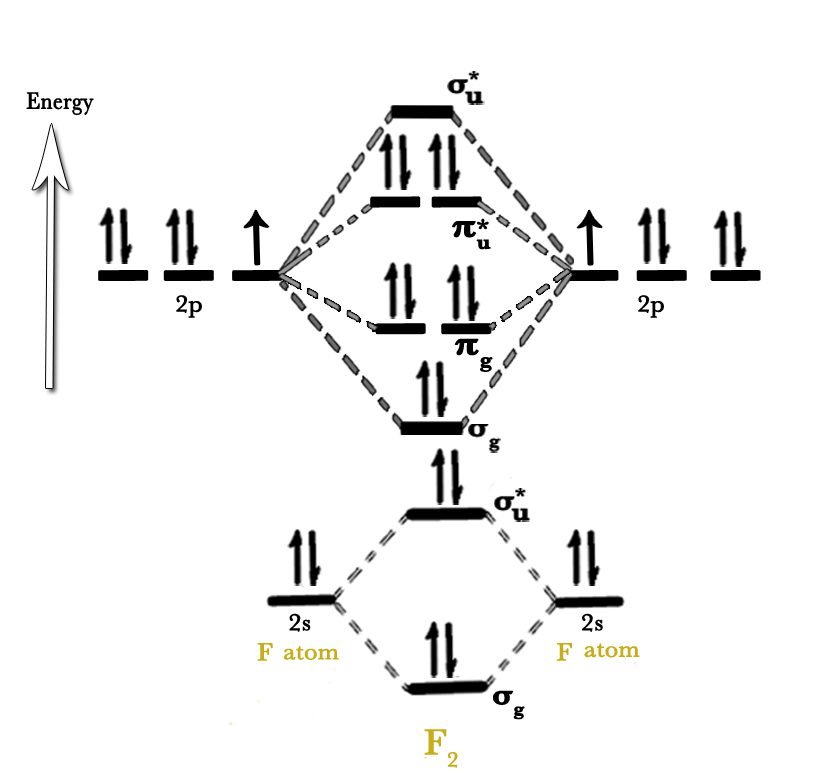
Bond Order
no of an electron in bonding – no of an electron in anti-bonding / 2
8-6 / 2
Bond order of F2 = 1
The bond order of F2 is the same as the bond order of H2.
Magnetic Nature
F2 is diamagnetic in nature.
Uses of Fluorine
- It is used as rocket propellent in combination with oxygen.
- NaF is used in fluoride toothpaste.
- It is used in prepare UF6, which is used for nuclear power generation and in the separation of uranium isotopes.
- Teflon is used in coating non-stick pans, and insulators.
- SF6 is the best electrical insulator.
Fact to Remember
- F– does not form precipitates with Ag+ as AgF is soluble in water due to high hydration energy.
- AgF has a higher value of lattice energy than other AgX.
- HF can be oxidized by anode only.
- H2F2 dissolves glass. This leads to the etching of glass.



Leave a Reply