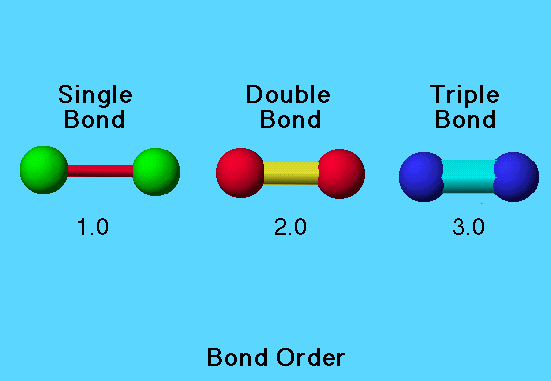
Bond order is the numerical value that explains the kind of bond formed between two bonding atoms. Understand with an example, how many bonds are present in hydrogen, oxygen, and nitrogen molecule?
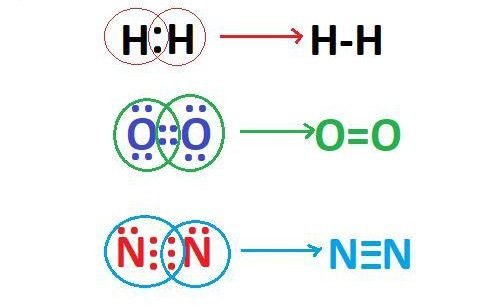
Hydrogen molecule has only one bond, which means the bond order is one. Similarly, oxygen and nitrogen molecule has two or three bonds, so their bond order is two and three respectively. So bond order is just a way of telling us how many bonds are there in between any particular atoms. The above definition of bond order is not 100% correct, this is the traditional definition. Now if we talk about the exact definition of bond order.
“It tells us about the strength of a bond“
If two atoms are connected by a single bond, what is its strength? The NO bond order is 2.5. How is possible the bond order is 2.5? The bond order of 2.5 means the bond between nitrogen and oxygen is 2.5 times stronger compared to if N and O are connected by a single bond. So this is the entire concept of bond order.
The triple bond is stronger than the double bond is stronger than a single bond.
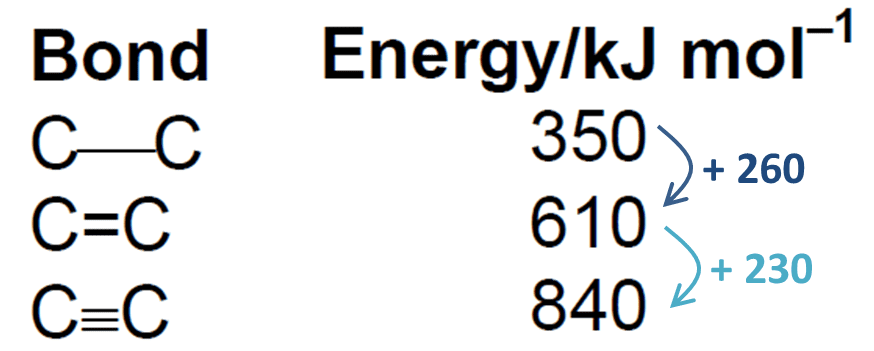
Why does Bond Length Increase with Bond Order?
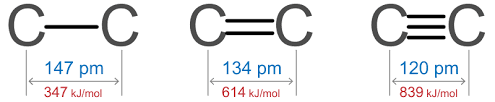
There is an inverse relationship between bond length and bond order. If the bond order is more, means two atoms are very close to each other so the bond length is short.
- The bond order of the carbon-carbon triple bond is greater than the carbon-carbon single bond.
- The bond length of the carbon-carbon single is greater than the carbon-carbon triple bond.
- If the bond is single, the energy required to break the bond is small so it means the bond enthalpy for a single bond is minimum.
- The bond enthalpy for the triple bond is maximum.
What is the relationship between Bond Enthalpy, Bond Order, and Bond Enthalpy?
Bond Enthalpy ∝ Bond Order ∝ 1/ Bond Length
| Bond length ∝ 1/ΔE.N | Bond energy ∝ ΔE.N |
| Bond length ∝ size | Bond energy ∝ 1/size |
| Bond length ∝ 1/% of S | Bond energy ∝ % of S |
| Bond length ∝ 1/bond order | Bond energy ∝ bond order |
| Bond length ∝ bond length | Bond energy ∝ 1/bond length |
What is the Formula for Bond Order?
Bond order = 1/2 (Number of electrons in BMO – Number of electrons in ABMO)
What does Bond Order tell you?
Stability of molecules
The greater the bond order of the molecule, the greater will be the stability. If the number of electrons in the bonding molecular orbital is greater than the number of electrons in the anti-bonding molecular orbital, it means the value of bond order is high. The higher the bond order means the strength of the bond between the atoms is greater so the molecule moves toward stability. We can say that if nb > na, the molecule will be stable and vice versa.
Bond Energy
The higher the bond order, the higher will be the bond energy of the molecule.
Bond Length
The higher the bond order, the shorter will be the bond length of the molecule.
How do you find the Bond Order shortcut?
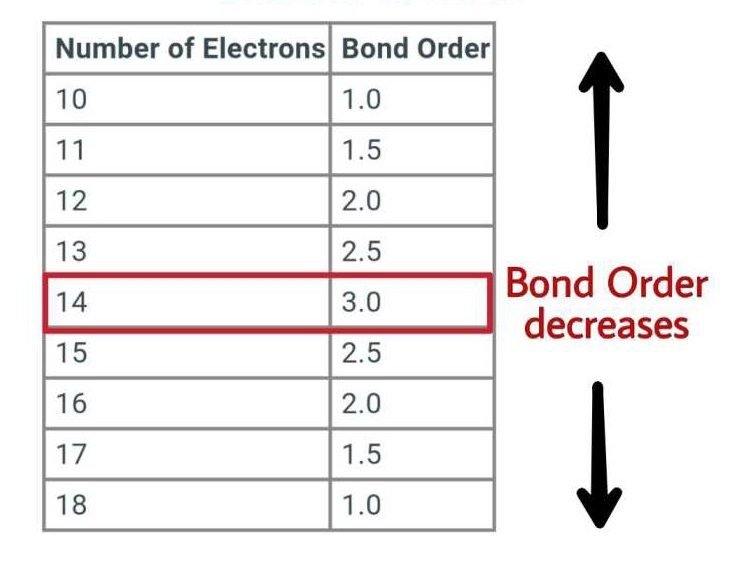
Find the Bond Order of CN, CO, O2, and NO+?
Bond Order of CN
- The number of electrons of carbon is 6.
- The electrons of nitrogen are 7.
- The total electrons of the CN molecule are 6+7=13.
- If the total electrons are 13 then the bond order will be 2.5.
Bond Order of CO
- The number of electrons of carbon is 6.
- The number of electrons of oxygen is 8.
- The total electrons of the CO molecule are 6+8=14.
- If the total electrons are 14 then the bond order will be 3.
Bond Order of O2
- The number of electrons of oxygen is 8.
- The total electrons of the O2 molecule are 8+8=16.
- If the total electrons are 16 then the bond order will be 2.
Bond Order of NO+
- The number of electrons of nitrogen is 7.
- The number of electrons of O+ is 7.
- The total electrons of the NO+ molecule are 7+7=14.
- If the total electrons are 14 then the bond order will be 3.
How do you Calculate the Bond Order of F2?
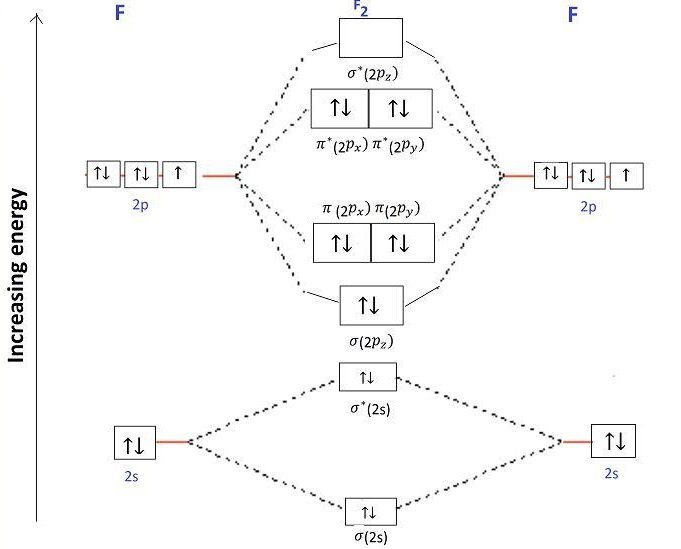
Bond order = 1/2 (Number of electrons in BMO – Number of electrons in ABMO)
Bond order = 1/2 (8-6)
Bond order = 2/2
Bond order = 1
Is F2 Paramagnetic or Diamagnetic?
All the electrons are paired so F2 is diamagnetic in nature.
How do you Calculate the Bond Order of NO?
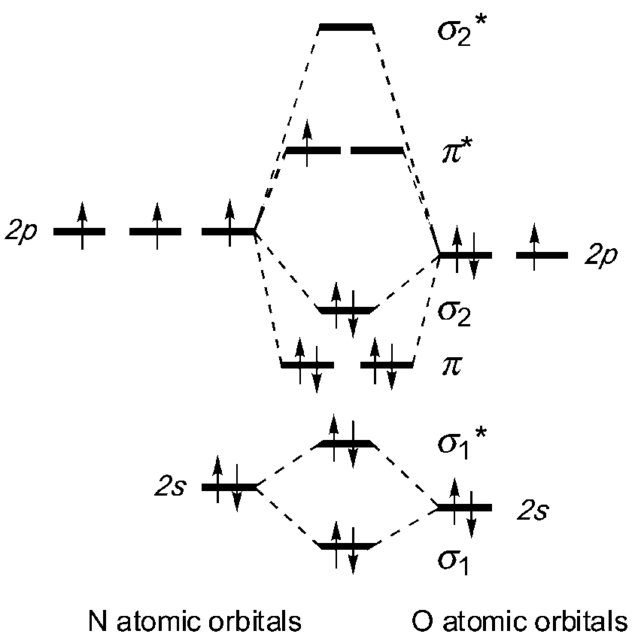
Bond order = 1/2 (Number of electrons in BMO – Number of electrons in ABMO)
Bond order = 1/2 (8-3)
Bond order = 5/2
Bond order = 2.5
Is NO Paramagnetic or Diamagnetic?
The electrons are unpaired so NO is paramagnetic in nature.
How do you Calculate the Bond Order of H2O?
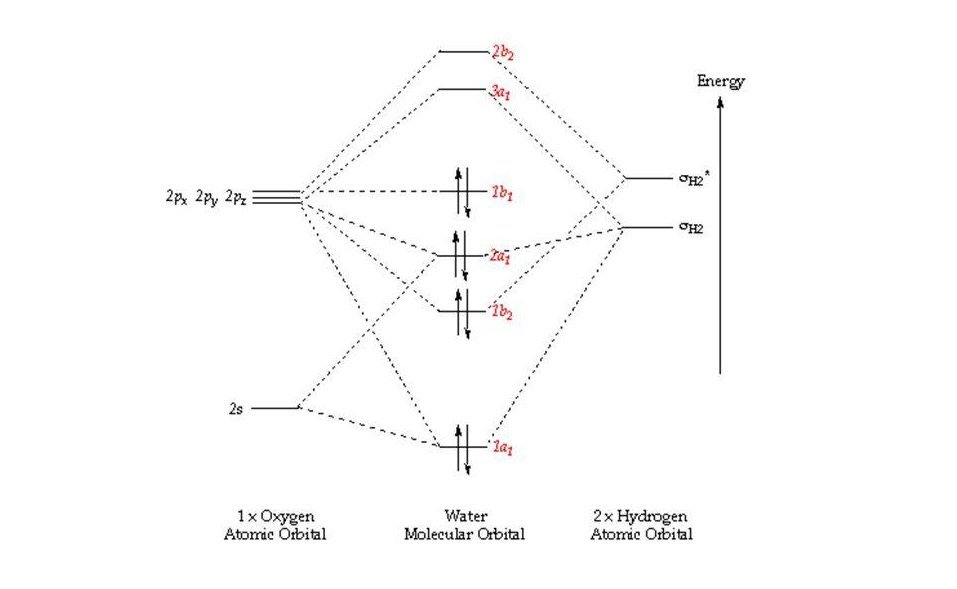
Bond order = 1/2 (Number of electrons in BMO – Number of electrons in ABMO)
Bond order = 1/2 (4-0)
Bond order = 4/2
Bond order = 2
Is H2O Paramagnetic or Diamagnetic?
The electrons are paired so H2O is diamagnetic in nature.
What is the difference between Bonding and Anti-Bonding molecular orbital?
| BMO | ABMO |
|---|---|
| BMO is obtained when two atomic orbitals combine together in such a way that their lobes with + + sign overlap. It is a + + overlapping. | ABMO is obtained when two atomic orbitals combine together in such a way that their lobes with the + – sign overlap. It is a + – overlapping. |
| The probability of finding the electron in the region between the nuclei of the combing atom is very high and hence a stable molecule is formed. | The probability of finding the electron in the region between the nuclei of the combing atom is negligible and an unstable molecule is formed. |
| The energy of BMO is less than AMBO. | The energy of AMBO is greater than BMO. |
| It is more stable than ABMO. | It is less stable than BMO. |
| The difference in energy between the molecular orbitals and the combing atomic orbitals is called stabilization energy. | The difference in energy between the molecular orbitals and the combing atomic orbitals is called destabilization energy. |



Leave a Reply