


while discussing the chemical defenses in plants, usually, plants have two main chemical groups: primary metabolites and secondary metabolites. Primary metabolites are chemicals synthesized by all plant cells and are engaged directly in growth, development, and reproduction. Sugars, proteins, amino acids, and nucleic acids are all examples. Although secondary metabolites are not engaged directly in growth or reproduction, they are often implicated in plant defense. These chemicals are often classified as Terpenoids, Phenolics, or alkaloids some of which are nitrogen containing compounds.
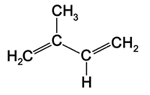
Terpenoids (terpenes) exist in all plants with more than 22,000 identified chemicals representing the larger class of secondary metabolites. The simplest terpenoid is hydrocarbon isoprene (C5H8), a volatile gas produced by huge amounts of leaves during photosynthesis which may protect the membranes of cells from high temperatures or light damages. The numbers of isoprene units utilised for the construction of terpenoids are categorised. Monoterpenoids include, for example, two isoprene units: sesquiterpenoids (three units), diterpenoids (four units), and triterpenoids (six units).

Essential oils are composed mostly of monoterpenoids and sesquiterpenoids, which are highly volatile chemicals that contribute to the fragrance (essence) of the plants that generate them. Numerous essential oils serve as insect repellents, and many protect against fungal or bacterial assault. Mint plants (Mentha spp.) generate and store a significant amount of the monoterpenoids menthol and menthone in glandular trichomes on the epidermis.
Pyrethrins are chrysanthemum-derived monoterpenoid esters that act as neurotoxins on insects. Numerous widely available pesticides are really synthetic pyrethroid analogues of pyrethrins, including permethrin and cypermethrin. Pine tree resin includes high concentrations of the monoterpenoids alpha- and beta-pinene, which are effective insect repellents; these chemicals also contribute to the strong odour of the organic solvent turpentine.
We don’t simply utilise them as pesticides. Plants use essential oils to produce insect poisons that are safe to humans. Examples include peppermint (Mentha spp. ), spearmint (Mentha spp. ), basil (Ocimum spp. ), oregano (Origanum spp. ), rosemary (Rosmarinus spp. ), sage (Salvia spp.) and savoury (Satureja spp). (Laurus spp.).
Diterpenoids like gossypol (Gossypium hirsutum) have antifungal and antibacterial effects. Triterpenoids are structurally related to sterols and steroid hormones. Phytoectysones are moulting hormone mimics. Soil-borne phytohormones impair larval development and increase insect mortality.
Lemon and orange peels smell fresh because of limonoids, a kind of triterpene. Azadirachtin is a strong limonoid found in neem trees (Azadirachta indica) that repels insects at low quantities. Because of its minimal toxicity to humans and biodegradability, citronella is a popular insect repellant in the United States.



| Figure 13. Gossypol | Figure 14. Azadirachtin | Figure 15. Digoxin |
Not all herbivores are insects. Toxic triterpenoids like cardiac glycosides may induce heart attacks in humans and other vertebrate herbivores. The cardiac glycosides digitoxin and digoxin are found in foxglove (Digitalis purpurea) and are used to treat heart problems in humans in modest doses.
Some herbivores have overcome the dangers of cardiac glycosides and utilise them to their advantage. Monarch caterpillars eat milkweed (Asclepias spp. ), which has high levels of these poisons in its milky latex. The poisons are securely stored inside the caterpillars, and when they mature into butterflies, they are extremely deadly to most predatory birds.

Saponins are glycosylated triterpenoids (triterpenoids with sugar groups attached) found in many plant cell membranes. These detergent-like chemicals damage the cell membranes of invading fungal infections. Gaeumannomyces graminis cannot infect oats that contain avenacins, a triterpenoid saponin. Botrytis cinerea, Fusarium oxysporum, and Septoria lycopersici are all capable of degrading saponins and causing illness in saponin-producing plants.
Plants generate phenolics to protect themselves against diseases. They contain defense-related chemicals such as flavonoids, anthocyanins, phytoalexins, tannins, lignin, and furanocoumarins. Flavonoids are a big phenolic class. These brightly coloured water-soluble flavonoids pigments help plants shield their leaves from harmful UV rays.
Anthocyanins are pigments found in flowers, fruits, and leaves of deciduous plants in the autumn. Antibiotic and antifungal isoflavonoids, phytoalexins are generated in response to pathogen assault. These pathogen-specific toxic chemicals impair pathogen metabolism or cellular structure. Some examples include medicarpin (Medicago sativa), rishitin (Solanaceae family), and camalexin (Arabidopsis thaliana).



Tannins are plant-produced, water-soluble flavonoid polymers that are stored in vacuoles. Tannins are poisonous to insects because they bind to salivary proteins and digestive enzymes such as trypsin and chymotrypsin, thus inactivating the proteins. Insect herbivores who consume a high concentration of tannins lose weight and may ultimately perish. Red wine’s strong flavor is produced by grape tannins attaching to salivary proteins in the mouth, causing protein coagulation.
Lignin is a very ramified heterogeneous polymer found mostly in the secondary cell walls of plants, but primary walls may sometimes lignify. It is a major wood component consisting of hundreds or thousands of phenolic monomers. Lignin offers a good physical barrier against pathogens since it is insoluble, stiff and practically indigestible.
Phenolic substances generated by a number of plants in response to pathogen or herbivore assault are furanocoumarins. They are triggered by UV light and may, owing to their incorporation in DNA, be extremely poisonous for some herbivores of the vertebrates and invertebrates, which lead to quick cell death. Grapefruit juice really includes little amount of furanocoumarins, which significantly increases the absorption of certain medications from the intestines in the circulation. Some medications have warning labels that advise patients not to beat grapefruit juice while taking the pills to prevent an accidental overdose.
Leave a Reply