
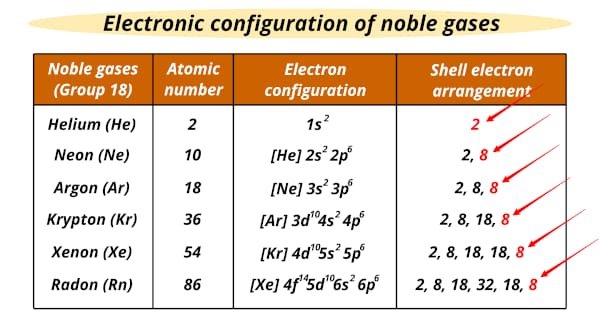
In the periodic table, the noble gases are group 18 elements including Helium, Neon, Argon, Krypton, Xenon, and Radon. Noble gases are not reactive because noble gases have two or eight electrons in their valance shell. However, if the elements do not have two or eight electrons in their valance shell they became reactive due to gaining or losing electrons. Noble gases do not react with other elements via a chemical reaction. Therefore, the inertness of noble gases occurs because all have full valance shells which makes them very stable.
Are all noble gases are inert gases?
In the past, chemists thought that noble gases could not react with other elements that’s why they were called inert or unreactive gases. He, Ne, Ar are inert gases due to the less shielding effect and have a strong force of attraction of nucleus on outermost shell electrons. However, we know today that some noble gases can indeed react to form some stable compounds. Kr, Xe, Rn reacts with oxygen and fluorine under extreme conditions. so, all inert gases are noble gases but all noble gases are not inert. let us see some compounds of Xenon:
| Compounds | Oxidation state | Physical state |
| XeF2 | +2 | Colorless crystals |
| XeF4 | +4 | Colorless crystals |
| XeOF2 | +4 | Colorless crystals |
| XeF6 | +6 | Colorless crystals |
| XeOF4 | +6 | Colorless liquids |
| XeO3 | +6 | Colorless crystals |
| XeO4 | +8 | Colorless gas |
Properties of Noble gases:
- Noble gases or rare gases are placed in the zero groups.
- All elements are non-metallic, odorless, and colorless gases.
- These have eight electrons in the valance shell of each element except helium.
- Noble gases are isolated from air by fractional distillation.
- Helium is present on earth as radioactive decay, the second most abundant element in the universe.
- Neon is 1/65000 the part of the earth.
- Neon gives a reddish glow in the gas discharge tube.
- Neon has 40 times more refrigeration capacity than helium.
- Xenon is present in the air at 0.08 ppm.
- Radon is a product of α-decay of radium.
- Boiling points increase down the group due to an increase in London forces.



Leave a Reply