
The coupling constant is abbreviated with the capital letter J. In this context, we discussed what is coupling constant is? What is its importance? How coupling constant gives us an idea about doublet or singlet? Does it tell us about neighboring Protons? It also differentiates the peaks of spectra.
What is Coupling Constant J?
“It is the distance between centers of two peaks in a multiplet”
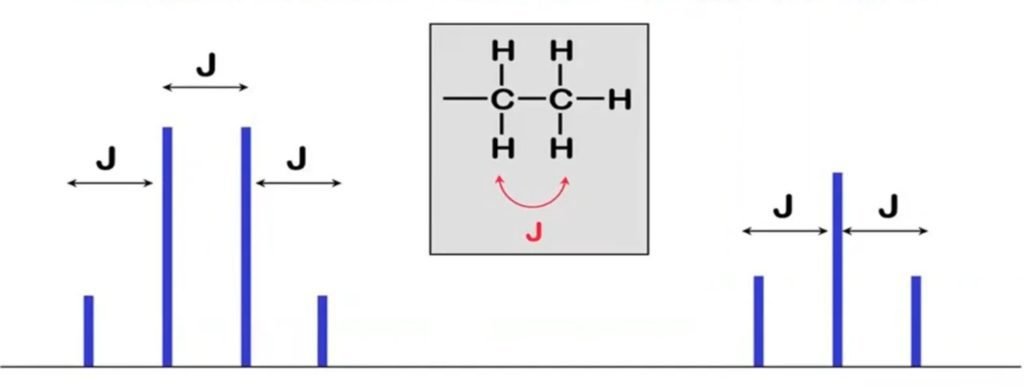
In the above diagram, we have one quartered and one triplet. Quartered means three more protons are present in the neighboring proton and triplet means two more protons are present in the neighboring proton. Quartered is a multiplet in which more than one peaks are present so the coupling constant is the distance J (measured in Hz) between the peaks in a multiplet.
If the value of J of two different peaks or multiplet is the same it means that these peaks are of that Proton that is neighboring to each other and interact with each other. J is a measure of the amount of interaction between the two sets of hydrogens creating the multiplet.
What do Coupling Constant Depends On?
The factor affecting the coupling constant are substituents, hybridization, ring stain, and dihedral angles.
The coupling constant is independent of magnetic field strength means that the value of J is not changed by changing the magnetic field strength.
How we differentiates the peaks by using Coupling Constant?

Let us have two spectrums and we observed that there are two dublets (red and blue). Now we check whether it is dublets or not? This means there are two possibilities that occur one is that it is a duplet and the second is that it is a singlet that comes close to each other and looks like a doublet. That’s why we find the value of J. Let the distance between two red and blue peaks is 15Hz.
The distance is the same and it is observed that they are neighboring protons and due to neighboring protons, they couple and that’s why the value of J is the same. To check whether it is dublets or not? We enhance the strength of the magnetic field.
After increasing the strength of the magnetic field, the spectra are changed. We observe that the distance between two red peaks is changed (20 Hz) but on the other hand there is no change in the distance between blue peaks (15 Hz).
Conclusion
- The red peaks are not doublet. (The value of J is not same after increasing the strength of the magnetic field)
- The blue peaks are doublet. (The value of J is same)
So, with the help of J, we differentiate the singlet or doublet.
Unit of Coupling Constant
The coupling constant is usually measured in frequency, so the unit is Hz or Cycle/sec.
Value of Coupling Constant
- Usually the value of J is up to 20Hz.
- The value is positive when spin is paired.
- The value is negative when spin is parallel.
What is Chemical Shift?
The Chemical shift is defined as the difference in parts per million (ppm) between the resonance frequency of the observed proton and that of the tetramethylsilane (TMS) hydrogen.
Unit of Chemical Shifts
Parts per million (ppm)

Let us have a doublet and what is the value of chemical shift? The center of the doublet is the value of chemical shift (small blue line). Let the value of chemical shift be 4 ppm. The value of the chemical shift of the red peak is less than 4 ppm and the chemical shift of the green peak is greater than 4 ppm.
- Chemical shift of red peak = 3.99 ppm
- Chemical shift of green peak = 4.01 ppm
J = 4.01 – 3.99
J = 0.2 ppm
Convert value in Hz
0.2 ppm × Hz / 200 MHz = 4 Hz
Types of Coupling
There are two types of coupling:
- Short range coupling
- Long range coupling
Short Range Coupling
2 Bond Coupling
3 Bond Coupling
Long Range Coupling
4 Bond Coupling
5 Bond Coupling
Example of Coupling Constant, Ethyl Bromide
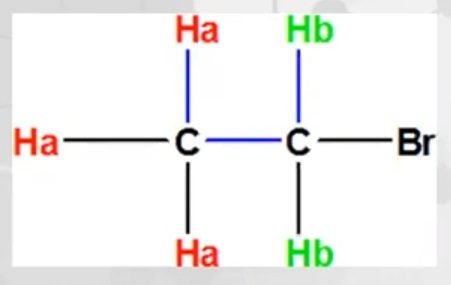
In ethyl bromide, there are two types of protons (CH3 and CH2). The coupling is 3 bond coupling.
- When b interact with a, its J3ba.
- When a interact with b, its J3ab.
- “a” protons have 2 neighbouring ‘b” protons, thus will give triplet.
- “b” protons have 3 neighbouring ‘a” protons, thus will give quartet.

“The value of J in triplet and quartet is the same showing that these are neighboring protons”
Another Example of Coupling Constant is given below:
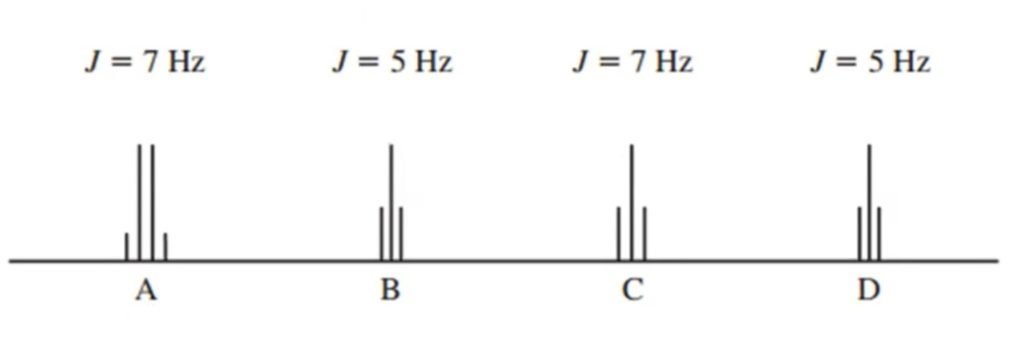
- The value of J of “A” quartet is 7 Hz
- The value of J of “B” triplet is 5 Hz
- The value of J of “C” triplet is 7 Hz
- The value of J of “D” triplet is 5 Hz
Value of J is the same means proton of neighboring carbons that interact with each other.
- Quartet in “A” is neighbouring to triplet in “C” (CH3CH2-)
- Triplet in “B” is neighbouring to triplet in “D” (-CH2CH2-)






Leave a Reply