
To understand the concept Is the salt compound or element we have to grasp a concept of what is salt??? What is the difference between compound and element????. So, let’s start understanding what is salt??
What is Salt??
A neutralization reaction’s product is referred to as salt. When an acid and base react with each other a neutralization reaction occurs and salt is formed.
HCl + NaOH → NaCl + H2O
In this reaction, HCl act as a strong acid, and NaOH acts as a strong base so, by their reaction, an acidic salt is obtained named NaCl (SODIUM CHLORIDE). Salts are made up of ions, i.e. anion or cation. It is also formed when two ions anions or cations react with each other resulting in the formation of salts. For example NaCl
Na+ + Cl–→ NaCl
Commonly salts are good conductors of electricity due to the complete dissociation of ions in water. They are good conductors in liquid form or molten form. The conductance of an electrolyte is possible due to the presence of ions.
But, the question arises that why NaCl dissociates in water as it is an ionic compound??? Generally, ionic compounds dissociate in water because it is the dielectric property of water to weak an ionic bond and dissolve ions in itself.
Salt is considered to be a neutral species. Salt is present in a large amount of seawater. In seawater, it is the main mineral constituent. It is very important for animal life and saltiness is one of the basic human tastes. The structure of salt exists in 3D in crystal lattice form.
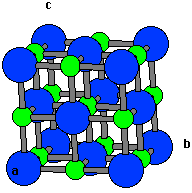
Salt may be of three types based on the presence of acid and base:
- When a diprotic acid or polyprotic acid is neutralized with a base the product obtained is an acidic salt.
H2SO4 + NaOH → NaHSO4 + H2O
- When a strong base is neutralized with the weak acid, the salt obtained is called a basic salt. When they are hydrolyzed they form a basic solution. The formation of basic salt occurs due to the formation of the conjugate base of a weak acid. For example 2PbCO3.Pb(OH)2.
- Some salts are neither acidic nor basic they are neutral salts. For example, the Zwitter ion contains anion and cation in the molecule. Zwitter ion may be any ion that has any cation and anion in the same compound.
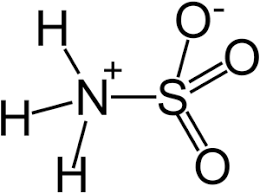
Properties of Salts:
1. Color:
Generally, salts may exist or tend to be transparent. The existence of colors in salts is possible due to the presence of cations and anions. Larger crystals of salts are transparent while polycrystalline crystals look like white powder. For example, Sodium chromate is yellow in color and cobalt nitrate is red.
2. Taste:
Salt may possess five tastes. These tastes are listed below with the salts possess these salts:
- Salty (NaCl)
- Sweet (Lead Diacetate)
- Sour (Pottasium Bitartarate)
- Bitter (Magnesium sulfate)
- Sovery (Monosodium glutamate)
3. Solubility:
Solubility refers to the interaction of ions of salts in water. Greater interaction means greater solubility. Salts may be ionic. Ionic salts dissolve in water due to the dielectric property of water. This water weakens the cohesive forces between the salt molecule and allows ions to interact with water more effectively.
4. Conductivity:
Salts in solid form are bad conductors of heat and electricity but in molten state or liquid form, they are excellent conductors of heat and electricity. Conductance may depend upon the number of ions dissociated. So, in molten or liquid form salts are dissociated into ions. So, that they can conduct electricity or heat more effectively and act as a strong electrolyte.
5. Melting point:
More commonly it is noticed that salts have a high melting point due to the presence of very strong intermolecular forces. For example, the presence of ionic bonds in the NaCl allows it to raise its melting point. NaCl → 801℃.
Is Salt compound or Element???? Let’s Discover
To discuss this variety of concepts we have to understand the basic difference between the compound and element. Both elements and compounds chemically exist in nature. Elements are substances made of a single type of unique atoms that can not be broken down into simpler substances. On the other hand, the compound is made of different elements in definite proportions.
| COMPOUND | ELEMENT |
| A compound contains the element in a fixed ratio but in different arrangments. | Elements contain atoms of unique types. Each atom has the same atomic number. |
| It can be separated by physical means, i.e. Filtration, solvent extraction, distillation, etc | It cannot be separated by physical means. |
| A compound is represented by a chemical formula. For example, NaCl, H2O. | An element is represented by a symbol. For example, C, N, O, P. |
So, according to the definition, if we observe the chemical formula of NaCl or salt, we will notice that the salt is mainly composed of an anion or cation. But these anions or cations are basically elements.
So we concluded that:
Salts are actually compounds that are made up of different elements that have definite propotion.



Leave a Reply