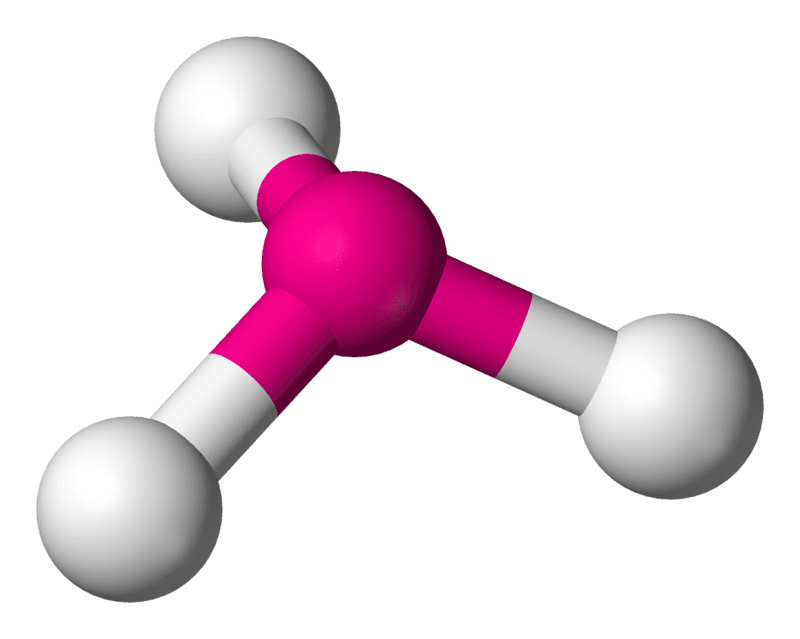
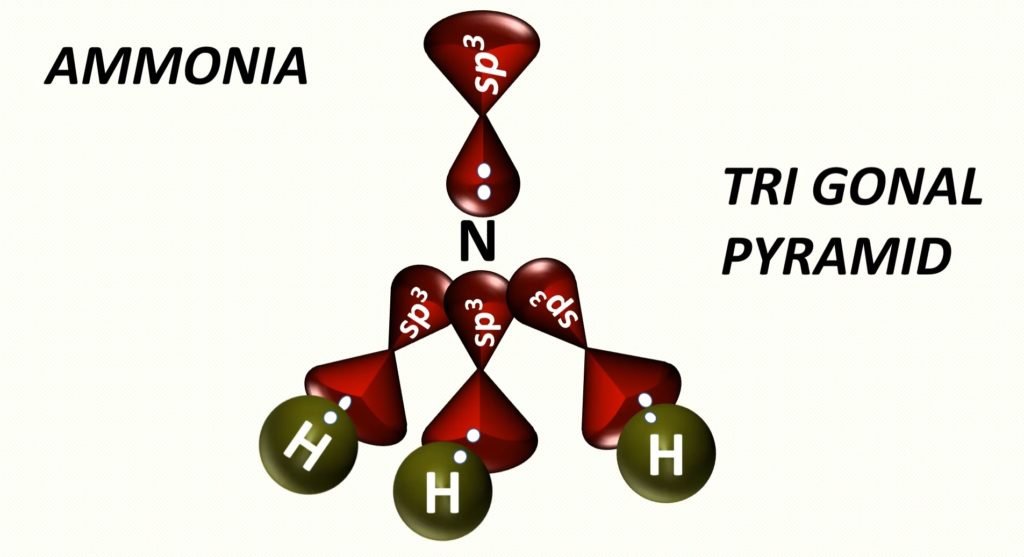
Trigonal pyramidal is a geometry of some molecules like ammonia and phosphine. Let us an example of ammonia to understand the trigonal pyramidal molecular geometry. Ammonia is an inorganic compound with three hydrogen atoms surrounding one nitrogen atom. All three hydrogen atoms shared their valence electrons with the nitrogen atom forming three strong covalent bonds. The nitrogen atom in ammonia contains a lone pair of nonbonded electrons.
This pair of nonbonding electrons on nitrogen repeal the electron shared by the hydrogen atoms. Due to this repulsion, the hydrogen atom spreads out to form a triangular base with the nitrogen atoms sitting at the apex of the molecule. When the shape is drawn out it resembles a triangular-based pyramid with the hydrogen atoms forming the bottom base and the nitrogen atom forming the apex of the pyramid.
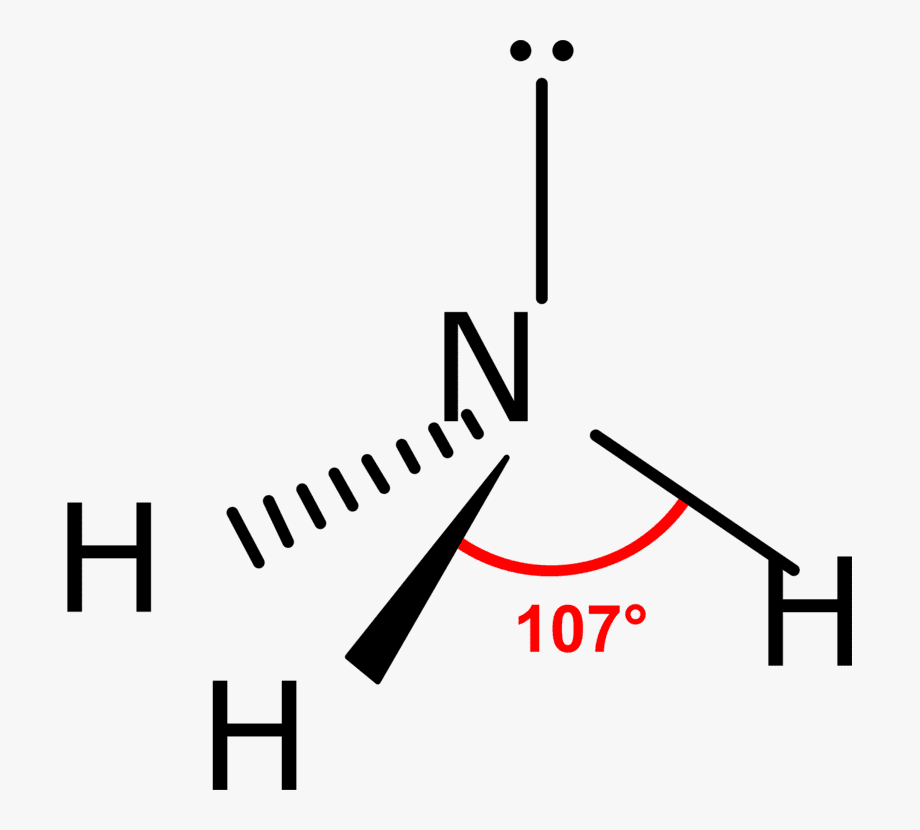
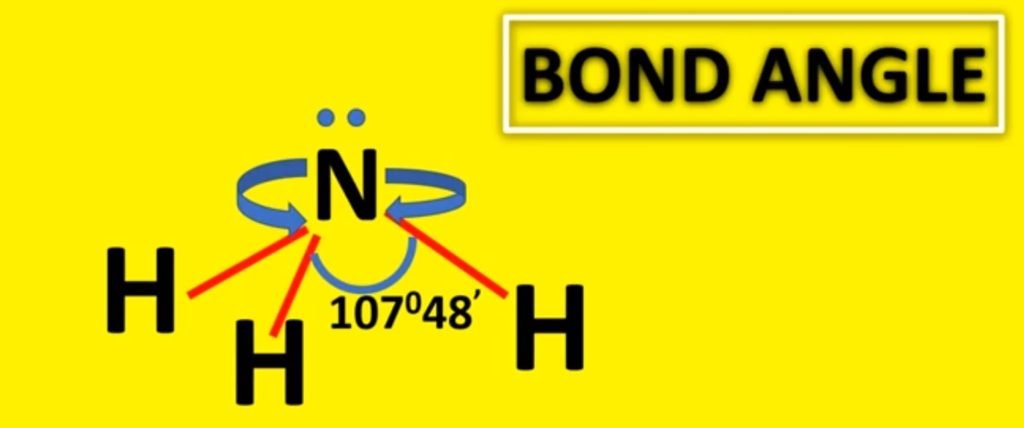
The bond angle in ammonia is approximately 107 degrees and this shape is similar to the tetrahedral shape of methane. In the sense that they can both be drawn out at a tetrahedron. However, the difference is that the central atom in ammonia is at the apex of the tetrahedron while the central atom in methane is embedded in the tetrahedron.
Trigonal Pyramidal Geometry of Ammonia
The molecular formula of ammonia is NH3. The central atom is nitrogen having atomic number 7. The electronic configuration of nitrogen is 1s2, 2s2, 2px1 2py1 2pz1. The hydrogen atomic number is 1. Its electronic configuration is 1s1. If 1s orbitals of hydrogen atoms overlap with three p orbitals of one nitrogen atom. There must be three p-s sigma bonds with a bond angle of 90 degrees between HNH in ammonia. But actually, the bond angle is approximately 107 degrees. How does it happen?
If we observe the valency electronic configuration of nitrogen, there is a minor energy difference between 2s and 2p orbitals.
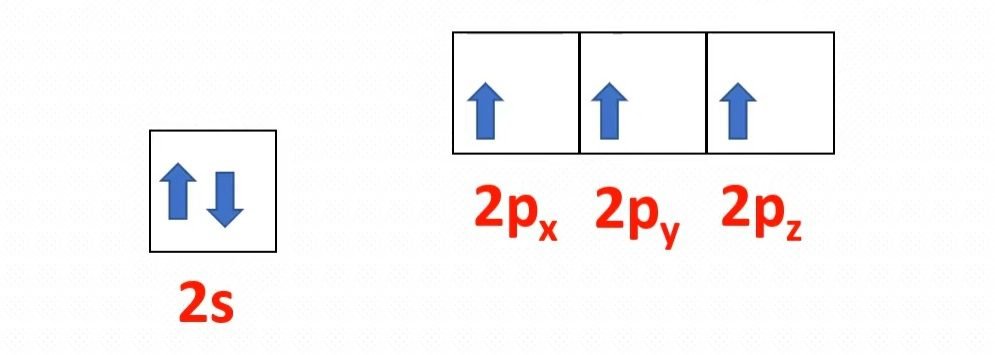
In nitrogen, between 2s and 2p orbitals, the process of hybridization is occur. Hybridization is a phenomenon of intermixing of outermost atomic orbital and their re-shuffling into the same number of orbitals but with equal properties like energy and shape.
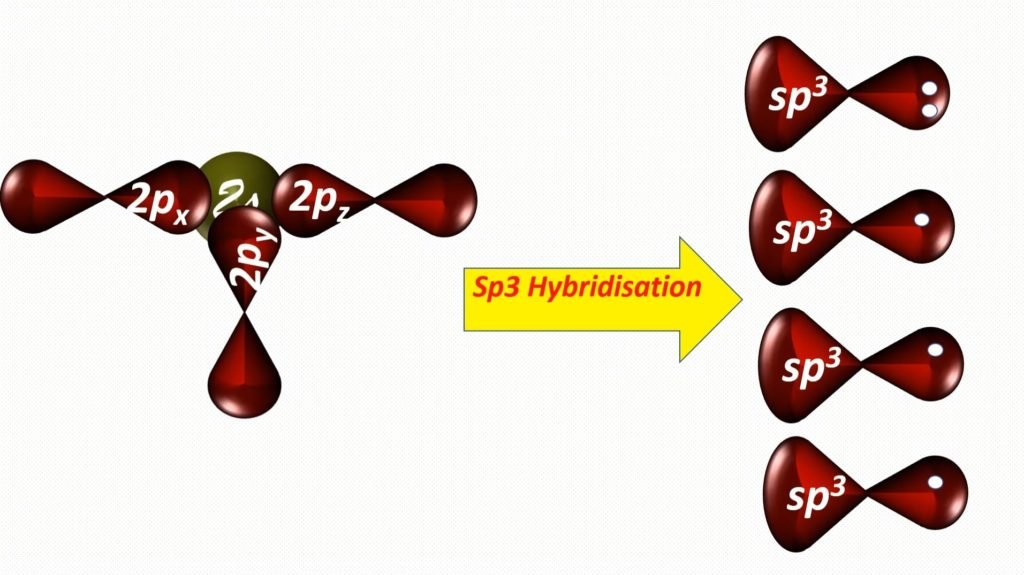
After the hybridization of the central atom, the bond formation of nitrogen and hydrogen.
Geometry of Ammonia
Trigonal Pyramidal
Bond Angle of Ammonia
Due to sp3 hybridization, its bond angle must be 109°28′ but due to the repulsion of the lone pair and bond pair, its bond angle is 107°48′.
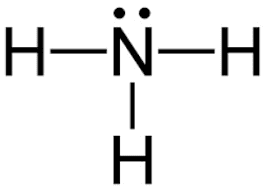
Lewis Dot Structure of Ammonia
NH3 lewis structure, first, we calculate Q.
Q = Valance electron of all atom + no of -ve charge – no of +ve charge
Q = (8 + 0 – 0)
Q = 8
B.P e– = 2 × no of bonds
B.P e– = 2 × 3
B.P e– = 6 e–
L.P e– = Q – B.P e–
L.P e– = 8 – 6
L.P e– = 2
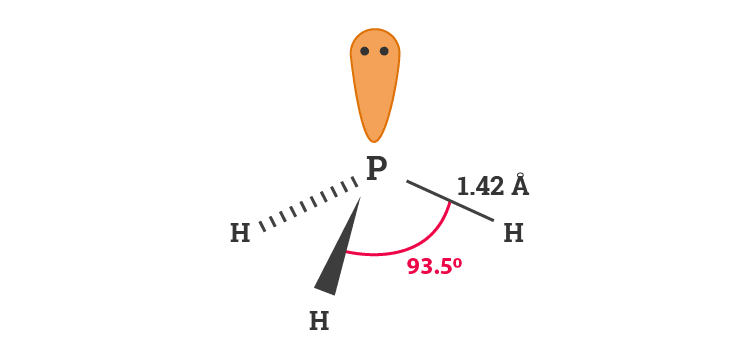
Trigonal Pyramidal Geometry of Phosphine
The molecular geometry of phosphine is similar to ammonia, trigonal pyramidal. Phosphine like ammonia has three hydrogen atoms spread out to form a triangular base just as with ammonia. This is because of lone pair of electrons present on the central atom phosphorus in phosphine.
Summary
- Some examples of trigonal pyramidal molecules are Ammonia and phosphine.
- Trigonal pyramidal molecules have three atoms arranged in a triangular base, with a central atom placed at the apex forming a pyramid.



Leave a Reply