
This blog will provide you with detailed information about diastereomers (sometimes called diastereoisomers), their properties, and a detailed explanation of them with some examples. After this, you will be able to distinguish diastereomers. So, let’s start with their introduction.
Introduction of Diastereomers
Stereoisomers have the same chemical formula, structural formula, and same sequence of connectivity but differ in the arrangement of atoms in space. They occur when two or more stereoisomers of a compound have different configurations. We can categorize stereoisomers into three types. Enantiomers, Diastereomers and conformers. Diastereomers are a type of stereoisomers.
Definition
All the optical isomers that do not mirror images are called diastereomers. They are non-identical stereoisomers. They are also called Diastereoisomers. They are neither enantiomers nor conformers.
Explanation with Example
3-Bromo-2-butanol

Explanation of diastereomers in 3-Bromo-2-butanol:
As we can see, the first two structures are enantiomers of each other and the last two structures are enantiomers of each other. If we place a mirror between the first and second structure, they will be the mirror image of each other and superimposable. And the same for the third and fourth structures.
If we look at the direction of 1st structure, it is clockwise (+ve). If we look at the direction of the 2nd structure, it is anticlockwise (-ve). And if we look at the direction of the 3rd structure, it is anticlockwise (-ve). Have a look at the direction of the 4th structure, it is clockwise (+ve). (This rotation is decided by D and L conventions)
The four possible combinations of these diastereomers are:
- (SS) 2S,3S-3-Bromo-2-butanol
- (RR) 2R,3R-3-Bromo-2-butanol
- (SR) 2S,3R-3-Bromo-2-butanol
- (RS) 2R,3S-3-Bromo-2-butanol These all four structures are diastereomers of 3-Bromo-2-butanol.
If we compare:
1st with 4th, 1st with third, 2nd with fourth, second with third;
- They are not a mirror image of each other.
- They are non-super-imposable to each other.
- Hence, they are known as diastereomers.
NOTE:
If we have more than two chiral centers for example three chiral centers with configuration RRR, then the configuration of any two of these three chiral centers will be changed (eg: RSS or SSR or SRS) not the configuration of all three have to be changed. Configuration of any one chiral carbon have to be same with parent compound.
Let's have a look at another example to understand this.

Four diastereomers are possible for Aldopentose. All four diastereomers have a different arrangement of molecules in space. They all are non-mirror images of each other and they do not superimpose each other. The first diastereomer of aldopentose contains 2R, 3R, and 4R configurations. The second diastereomer of aldopentose contains 2S, 3R, and 4R configurations. The third diastereomer of aldopentose contains 2R, 3S, and 4R configurations. The fourth diastereomer of aldopentose contains 2, 3S, and 4R configurations.
Diastereomers take place when two or more stereoisomers of a compound have different configurations at one or more of the equivalent stereocenters and are not mirror images of each other. They do not take place when all the stereoisomers have a different configuration. When two diastereomers differ from each other at only one stereocenter called epimers. Each stereocenter gives rise to two different configurations.
Some other Examples of diastereomers
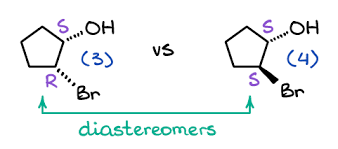
If we have a look at these compounds, they both are diastereomers of each other. Only one position is changed (from R to S). In the first diastereomer, Bromine is attached with a dashed bond that is going away from the observer while in the second diastereomer, Bromine is attached with a solid wedge bond that is going towards the observer. The position of the hydroxyl group is the same in both diastereomers (S, S).
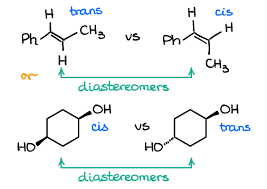
Have a look at the first example, now you can easily understand how the position of the Ch3 group is interchanged and the position of the first hydrogen is the same.
In the next example shown above, you can see the attachment of one hydroxyl group in the first diastereomer with a wedge bond while in the second it is connected with a dash bond.
Now you have a task; Why and how these above compounds are diastereomers? Show some potential in comments section.
Properties of diastereomers
- They are non-mirror images.
- Diastereomers have different physical properties.
- They have different chemical reactivity.
- They have the same structural and chemical formula.
- Diastereomers show similar chemical properties.
- They have different free energy.
- Diastereomers have different solubility.
- They have different arrangements of atoms in space.
- Diastereomerism can also take place at a double bond.
- Many conformational isomers are diastereomers as well.
- They can be separated from one another through techniques like fractional crystallization, fractional distillation, chromatography, etc.
- They have different melting points, boiling points, densities, refractive indices, dielectric constants, and specific rotations.
Conclusion
So, Diastereomers are not mirrored images, non-superimposable, and optically active compounds with different configurations (R and S) at two or more chiral centers but not all. They have the same structural, chemical, and molecular formula. The base on which we distinguish them is the arrangement of their molecules in space. They have different melting points, boiling points, and densities. They can be separated by fractional distillation and chromatographic techniques.



Leave a Reply