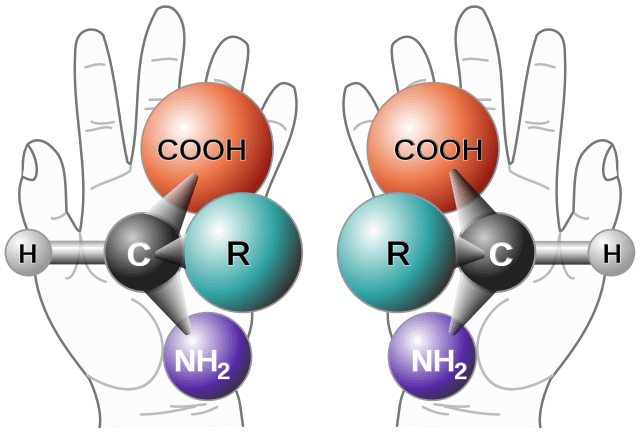
Definition
Enantiomers are non-superimposable mirror images. They are characterized by having an atom attached to four different groups called asymmetric or chiral (A molecule or ion that is not superimposable on its mirror image is said to be chiral). They are also called enantiomorphs.
Brief Introduction
Enantio is a Greek word that means opposite mirror images.
In chemistry, two stereoisomers are said to be enantiomers if the mirror image of one cannot be superimposed on that of the other. In other words, an enantiomer is a non-superimposable mirror image of itself. They have the same physical and chemical properties, except for interaction with plane-polarized light or with other chiral molecules. They are chiral molecules. The equimolar mixture of each enantiomer in a solution is called a racemic mixture and is optically inactive. Enantiomers are always in pairs. To understand enantiomers, the most straightforward example is our hands. Both hands are enantiomers of each other.
Properties of enantiomers
Here are some properties of enantiomers.
- They are Stable Compounds.
- They are isolable (Can be separated but not with ordinary means and by chemical reaction).
- Can not be interconvertible under ordinary conditions.
- Both mirror images have identical properties.
- They differ in interaction with plane-polarized light (Positive clockwise and negative anticlockwise).
- Their equal amount of mixture is a racemic mixture that is optically inactive.
- Individual compounds are optically active.
Representation
For convenience, enantiomers are usually represented by the Fisher projection. This consists of imagining that the molecule is observed so that two of the bonds are forward and the other two are backward. The bonds above the plane are represented by a solid bond or by a Wedge bond or can be represented by a bold line, and the bonds below the plane are represented by a dashed bond (dotted lines). In this way, a kind of cross is formed, with the asymmetrical carbon in its center, showing itself as the intersection of the lines. The groups in the horizontal line are towards the front, and the vertical ones are in the back.

Here in the above Structure, The Solid bond represents the attachment of a molecule or compound above the plane, and the Dash bond represents below the plane.
In enantiomers of any two compounds having a wedge and a dashed bond, the wedge or solid bond is replaced by a dashed bond and vice versa.
Examples of Enantiomers
(D) and (L) Tartaric Acid
Two optically active structures of tartaric acid are the easiest examples of enantiomers. The configuration of these molecules is shown below:
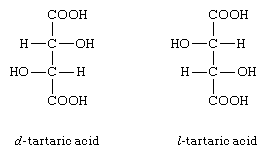
In d-tartaric acid, OH is on the left side and H is on the right side while in l-tartaric acid, OH is on the right side and H is on the left side and they both are enantiomers of each other. These two acids have pretty same melting points, solubility, and density in optically inactive solvents and have the same rate of reactions with optically inactive reagents.
(R) and (S) Lactic acid
Here is another example which is of Lactic acid.

S-lactic acid and R-lactic acid are enantiomers of each other. S- is on the right side while R- is on the left side, R-lactic acid is having H at its right and CH3 on the left. If we put S-lactic acid in front of the mirror then the same structure (R-lactic acid) could be seen.
Enantiomers of (R) and (S)-1-Bromo-1-Chloroethane
In molecular models it can be seen that the two molecules are distinct, they do not overlap.
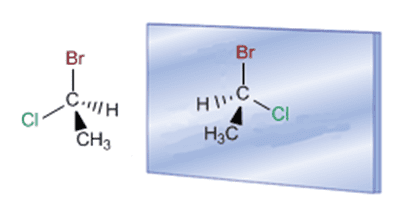
The presence of an asymmetric carbon (with different substituents: methyl, hydrogen, chlorine, and bromine) makes it possible for the molecule and its mirror image to be different.
Enantiomers of (R) and (S)-Alanine
(R) and (S)-Alanine is another example of enantiomers
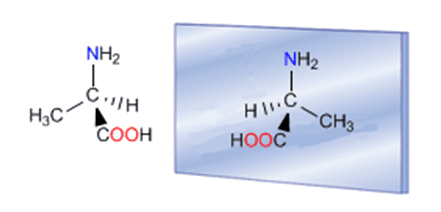
The presence of one carbon attached to four different substituents (-CH 3,-H,-NH 2, and -COOH) converts alanine into a chiral and optically active compound, with a non-superimposable mirror image (enantiomer). As can be seen in the molecular models, the molecule and its mirror image differ in the spatial arrangement of the groups and there is no twist that allows them to be superimposed (they are different).
Nomenclature of enantiomers
An enantiomer that rotates the plane of polarized light to the right (clockwise) is said to be dextrorotatory and is usually named with a lowercase letter (d), or a sign positive (+). If it does it to the left, it is levorotatory and a lowercase letter ele (l), or a negative sign (-) is usually placed on the name. A non-experimental way to name them is the one devised by CIP Rule.
Cahn-Ingold-Prelog priority rules
These rules were given by Robert Sidney Cahn, Christopher Kelk Ingold and Vladimir Prelog. In short term we also called as CIP rule.
R and S Nomenclature
R and S are derived from Latin words, R stands for Rectus meaning right (clockwise) and S stands for Sinister meaning left (anticlockwise). This system is applied only to chiral carbons.
It consists of numbering the groups of atoms bonded to the asymmetric carbon or chiral carbon. According to this nomenclature, priority number 1 is assigned to the atom or group of atoms that have the highest atomic number and priority number 2 to the atom which have less atomic number than the 1st atom, and so on. We will not count the atom or group which have the least atomic number among all the atoms or groups, we will give it the number 4 and will ignore it for some time; For example hydrogen (atomic number 1).
Hence;
If the configuration is in clockwise order, a capital r (R) is placed, while if it is anticlockwise, a lowercase s (s) is placed. We can not decide whether it is R or S on the plane of the paper, to decide we must have its model.
Example:
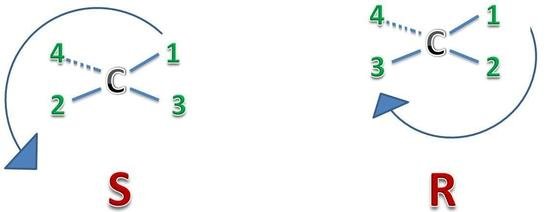
This is the simplest example of R and S enantiomers. The structure on the right is R and on the left is S. In R the rotation is clockwise because atoms with chiral carbon are designated their position according to their atomic number. While in S the rotation is anticlockwise.
Here is another example to understand this:
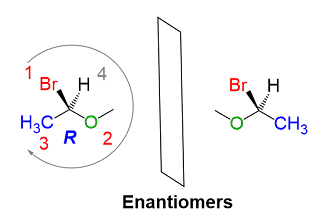
In this structure, there can be easily observed a mirror image of that compound. Priority 1 is assigned to Br because it contains the highest atomic number among all of the atoms attached to this chiral carbon and priority 2 is assigned to oxygen because after Br, O has the highest atomic number and the 3rd position is assigned to CH3, and 4th to Hydrogen. So this has a Clockwise configuration and its mirror image will be anticlockwise.
Separation of Enantiomers
The separation of enantiomers present in a racemic mixture is done by the technique called resolution. The most used resolution methods are:
Chemical Resolution
It is the separation of the enantiomers by converting the racemic mixture into a mixture of diastereomers. To do this, the mixture of enantiomers is made to react with the chiral compound that receives the name of “resolving agent”; with which the enantiomers are transformed into diastereomers, which can be separated by distillation, crystallization, or chromatography. Once separated, the resolution agent is removed to obtain each of the pure enantiomers.
Chromatographic Resolution
It is based on the use of chromatographic techniques that use a chiral compound as a stationary phase. The separation is based on the weak interactions that the enantiomers form with the chiral stationary phase. These interactions have different physical properties and thus different binding energies. Thus, the enantiomer that forms more stable complexes with the chiral stationary phase moves more slowly than the enantiomers that form fewer stable complexes; and due to this they elute at different speeds and separate along the column.
Conversion into Diastereomers
Enantiomers can be converted into diastereomers by the formation of salt with a chiral acid or base. They can be separated by their different chemical and physical properties. The acid or base is removed and the pure enantiomers are obtained. Furthermore, enantiomers can be separated by chromatographic methods; such as gas chromatography, high-pressure liquid chromatography, and thin-layer chromatography.
Other methods of Resolution are
- Biochemical resolution
- Kinetic method
- Selective adsorption method
- Mechanical separation
Conclusion:
Enantiomers are molecules that have the same molecular formula but different 3-D structures. They mirror each other, like your hands or shoes on opposite sides of your body. Opposite-handedness is called “chirality.” The most common form of enantiomerism in organic chemistry is where one hand (enantio) has a left-handed twist and the other has a right-handed twist (dextro). This article covered some basic information about this topic for those who want to learn more about it!



Leave a Reply