
What is Electronegativity?
Electronegativity is generally the ability of an atom to attract pair of bonding electrons toward itself. It is dimensionless property because it is just an ability or capability of an atom to attract pair of electrons.
The definition’s the ability of an atom to get pair of electrons toward itself is known as electronegativity.
Measuring :
So, the most common scale for the measurement of electronegativity is the Linus Pauling scale. According to this scale
- Most electronegative is Fluorine 4
- The least electronegative is Cesium 0.7
Influence of electronegativity on covalent bonding:-
Electronegativity governs the nature and strength of the covalent bond. Two atoms of the same with zero electronegativity difference from a pure covalent bond. Like H2 molecule and O2 molecule.While two atoms with different electronegativity cause covalent bond polarization. As like HF molecule and
Types on the basis of electronegativity difference:-

Taking electronegativity as a base there are three types of bonds. The list is given below.
- Polar Bond
- Non-Polar Bond
- Ionic Bond
Polar bond:-
When a covalent bond is formed they share electrons equally with each other. A polar bond is a bond in which there are two poles in an opposite direction positive and negative pole. Why these poles are formed. The only reason behind pole formation is the significant electronegative difference between bonding atoms. Due to this electronic cloud displaces near to more electronegative atoms.
As it is clear from the image the electronic cloud is closer to the more electronegative B atom.
Examples of polar bonded molecules :
Some examples of polar bonded molecules are given such as HF, water, and Methanol. These are some famous examples of polar molecules.
Non-Polar bond:-
A covalent bond is known as the non-polar if both the bonded atoms are of the same electronegativity. There is no pole formation when the electronegativity difference is close to zero. Non-polar bonds share electrons equally between bonded atoms.
As it is clear from the image that both electrons A and B attract electrons equally.
Examples of non-polar bonded molecules:-
Some common examples of non-Polar molecules are H2, Cl2, CCl4 are some examples of non-polar molecules.

Ionic Bond :-
Ionic bonds are formed as a result of the fully transferring electrons from one atom to another. In ionic bonding bounded atoms have greater electronegativity difference which results in fully transfer of an electron from one atom other.
Examples of ionic bonds:-
Some examples of ionic bonds are NaCl and KCl.
Periodicity of Electronegativity:-
The trend in the periodic table refers to periodicity. Here we are going to discuss the periodicity of the electronegativity along the groups and periods.
Electronegativity trend in periods:-
The trend in periods of electronegativity is given as the period’s electronegativity increases.
Reason: we go from left to right along period there is a decrease in the size of atoms. Know why size decreases as we move along periods. The reason is the electron and proton number increases over the period. But shell no remains the same. Due to the increase in nuclear charge more attraction is faced by the outermost shell and due to this reason shell size shrink along the period and electronegativity increases.

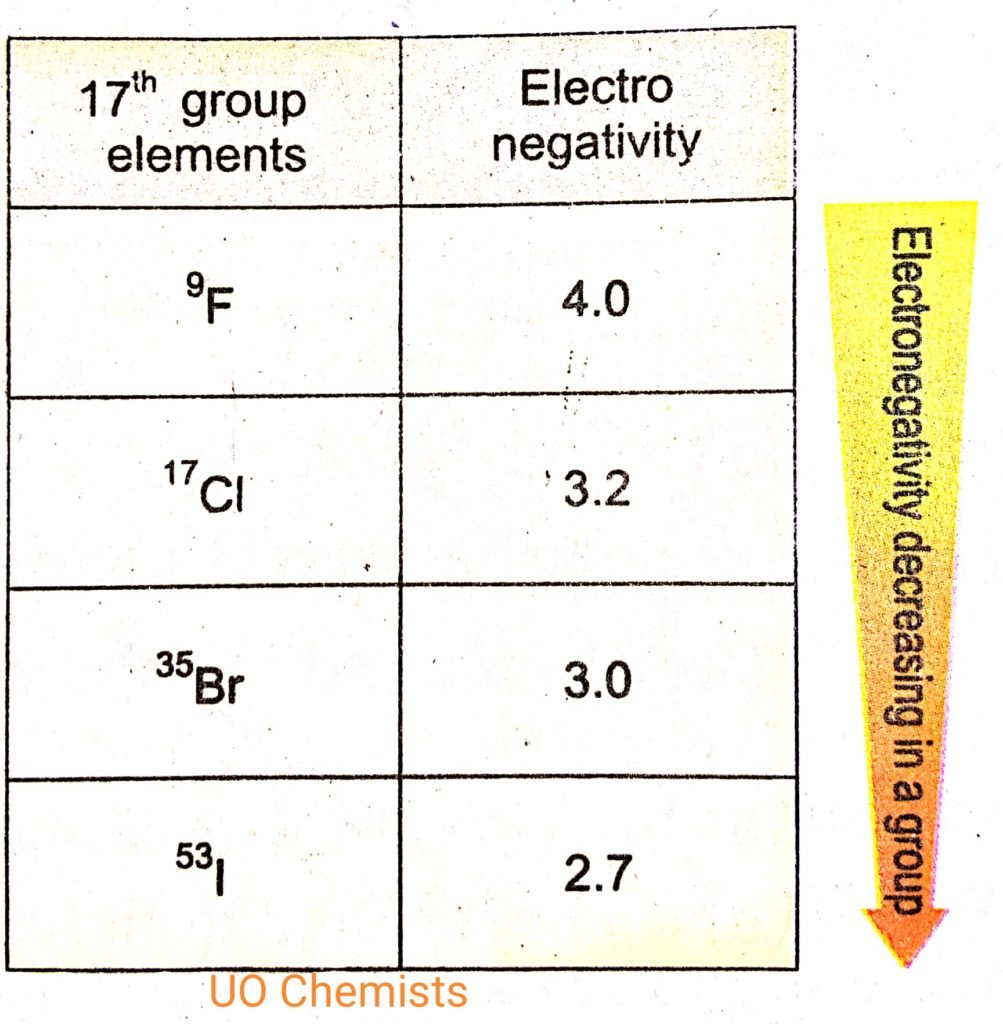
Electronegativity trend in groups :-
The trend in groups of electronegativity is given as the group’s electronegativity decreases.
Reason: we go from top to bottom along with group there is an increase in the size of an atom. Know why size increases as we move along the group. The reason is an electron and proton number increases along with groups. And also shell no increases. As shell no increase size of atoms also increases and shielding e also increases and reduce the tendency to attract shared pair of electron.
Factors that govern the electronegativity:-
Some factors govern the electronegativity, how and in which manner detail is given below.
- Atomic size
- Nuclear charge
- Substitution effect
Atomic Size and electronegativity :
Electronegativity and atomic size are inversely proportional to each other. As the size of the atom increases shielding effect also increases that reducing the ability of the atom to attract shared pair of electrons toward itself. Similarly when the size of atom decreases then more attractive forces attract a shared pair of electrons toward itself. So as a result increase in size causes a reduction in electronegativity and a decrease in size causes an increase in electronegativity.
Nuclear charge and electronegativity :
Electronegativity and nuclear charge both are directly proportional to each other. With the increase in nuclear pull when the size does not increase then electronegativity also increases. And with the decrease in nuclear pull causes a decrease in electronegativity.
Effect of substitution on electronegativity :
Substitution attached to the alpha atom also affects its electronegativity. Let’s see this because of an example. In CF3 carbon have more electronegativity as compared to carbon in CH3.



Leave a Reply