In a first order reaction, the sum of components of the concentration of reactants in a rate expression is equal to one. Let us take a substance (reactant) “A” which decomposes into products. Its initial concentration is “A°” mol/dm3. Let after time “t” the amount of substance left behind is “A” mol/dm3.
A → Product
Rate of reaction (ROR) = r = k [A]1
The rate of reaction depends upon the concentration of A.
Integrated Rate Law
A → Product
Rate = r = k [A]1 ———- (1)
Rate = r = – dA / dt ———-(2)
Compare eq (1) and (2)
– dA / dt = k [A]1
dA / dt = – K dt
∫dA / dt = – ∫K dt
At t=0 the initial concentration of A is A° and after time t the concentration of A is A.
A°A∫dA / dt = – 0t∫K dt
|loge A|AA° = -K |t|t0 ∵ ∫dA / dt = loge A
loge A – loge A° = -K (t-0)
loge A – loge A° = -Kt
loge A = loge A° – Kt (or) ln A = ln A° – Kt
Now convert loge to log10
2.303 log10 A = 2.303 log10 A° – Kt
Divided both sides by 2.303 and we get
log10 A = log10 A° – Kt/2.303
This is the integrated rate law of first order kinetics.
Other forms of Rate Law
log10 A = log10A° – Kt/2.303
log10 A° – log10A = Kt/2.303
log10 (A° / A) = Kt/2.303
t = 2.303/K log10 (A° / A)
As we know that
loge A = loge A° – Kt
loge A – loge A° = – Kt
loge (A / A°) = – Kt
A / A° = e-kt
A = A° e-kt
Half-Life of First Order Reaction
Half-life is a time in which the reaction completes 50%. The initial concentration is A° but when t=t1/2 the final concentration becomes half, A°/2.
Initial concentration = A°
at t=t1/2, A = A°/2
t = 2.303/K log10 (A° / A)
By putting the value of “t” and “A” we get,
t1/2 = 2.303/K log10 (A° / A°/2)
t1/2 = 2.303 log10 (2) / K
t1/2 = 2.303 (0.3) / K
t1/2 = 0.693 / K
Lifetime of Reaction (tlf)
The lifetime of the reaction is a time in which 100% reaction is complete.
At t = tlf , A = 0
tlf = 2.303/K log10 (A° / 0)
tlf = ∞
The first-order reaction can never complete 100%.
Graphical Representation of First Order Reaction
Graph: Conc. of Reactant vs time
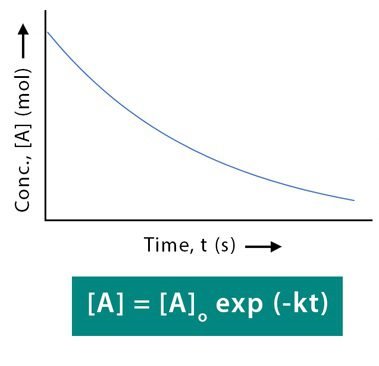
Graph: ln [A] vs time
![Graph: ln [A] vs time](https://www.uochemists.com/wp-content/uploads/2022/11/First-Order-Reaction-Equation-Graph-1.jpg)
Numericals
In a first order reaction, the concentration of the reactant decrease from 800 mol/dm3 to 50 mol/dm3 in 2×104 s. Find the rate constant.
t = 2.303/K log10 (A° / A)
K = 2.303/t log10 (A° / A)
K = 2.303/2×104 log10 (800 / 50)
K = 2.303/2×104 log10 16
K = 2.303/2×104 log10 24
K = 2.303/2×104 4 (log10 2)
K = 2.303/2×104 4 (0.3)
K = 2.303/2×104 4 (03/10)
K = 13.818 / 105
K = 13.8×10-5
K = 1.38×10-4
A first order kinetics is 20% complete in 5 min. Find the time in which it is 60% complete.
Let A° = 100
20% reaction is complete so the remaining concentration is A = 80
t = 5 min
t = 2.303/K log10 (A° / A)
5 = 2.303/K log10 (100 / 80) ———- (1)
A° = 100
60% reaction is complete so the remaining concentration is A = 40
t = ?
t = 2.303/K log10 (100 / 40) ———- (2)
Divide equation (2) by (1) we get
t/5 = log10 10 – log 4 / log10 10 – log 8
t/5 = log10 10 – log 22 / log10 10 – log 23
t/5 = 1 – 2 log 2 / 1 – 3 log 2
t/5 = 1 – 2 (0.3) / 1 – 3 (0.3)
t/5 = 0.4 / 0.1
t = 4 × 5
t = 20 min
In first order reaction, “A” gives a product. The concentration of “A” changes from 0.1 M to 0.025 M in 40 mins. Find the rate of reaction when the concentration of “A” is 0.01 M.
Rate of reaction =?
Rate of reaction = r = K [A]1
First to find the rate constant, to find the K
t = 2.303/K log10 (A° / A)
K = 2.303/40 log10 (0.1 / 0.025)
K = 2.303/40 log10 4
K = 2.303/40 2 log10 2
K = (2.303) (2) (0.3) / 40
K = (2.303) (2) (3) / 400
K = (2.303) (6) / 400
K = 13.818 / 4 × 100
K = 3.454/100
Rate = K [A]1
Rate = 3.454/100 × 0.01 M
Rate = 3.454/100 × 1/100
Rate = 3.454/10000
Rate = 3.4 × 10-4
Important Points to Remember
- The time taken for 75% completion of the first-order reaction is equal to 2 times taken for 50% completion. (t75% = 2 t50%)
- The time taken for 99.9% completion of the first-order reaction is equal to 10 times taken for 50% completion. (t99.9% = 10 t50%)
- The time taken for 25% completion of the first-order reaction is equal to 0.415 times taken for 50% completion. (t25% = 0.415 t50%)
- The time taken for 87.5% completion of the first-order reaction is equal to 3 times taken for 50% completion. (t87.5% = 3 t50%)
Prove that t75% = 2 t50%.
t = 2.303/K log10 (A° / A)
Let the initial concentration (A°) of reactant is 100, when 50% reaction is goes to completion the remaining concentration of reactant is 50.
t50% = 2.303/K log10 (100 / 50) ———- (1)
When the reaction is 75%complete, the remaining concentration is 25.
t75% = 2.303/K log10 (100 / 25) ———- (2)
Divide (2) by (1)
t75% / t50% = (2.303/K log10 (100 / 25)) / (2.303/K log10 (100 / 50))
t75% / t50% = 2 log10 2 / log10 2
t75% / t50% = 2
t75% = 2 t50%
Prove that t99.9% = 10 t50%.
Let the initial concentration (A°) of reactant is 100, when 50% reaction goes to completion the remaining concentration of reactant is 50.
t50% = 0.693 / K ——— (1)
when 99.9% reaction goes to completion, the remaining concentration of the reactant is 0.1.
t99.9% = 2.303/K log10 (100 / 0.1)
t99.9% = 2.303/K log10 (1000 / 1)
t99.9% = 2.303/K log10 103 ———- (2)
Divide (2) by (1)
t99.9% / t50% = (2.303/K log10 103) / (0.693 / K)
t99.9% / t50% = 2.303 × 3 / 0.693
t99.9% / t50% = 6.909 / 0.693
t99.9% / t50% =10
t99.9% = 10 t50%
Examples of First Order Reaction
- Nuclear physics reaction (Radioactive decay)
- Growth and decay of bacteria
- Inversion of cane sugar in an acidic medium
- Acidic hydrolysis of ester (esterification)
- Dissociation of N2O5
- Dissociation of H2O2
Conclusion
These reactions hold significant importance in fields such as medicine and environmental science. By comprehending them, you can anticipate the duration of a drug’s effectiveness or how pollutants break down.
Therefore, having an understanding of first order reactions can assist you in analyzing processes that involve growth or decay. You may utilize this knowledge to investigate and tackle problems in various areas of chemistry or everyday life.
FAQs
Q1: How can I determine if a reaction follows a first-order kinetics?
First Order Reactions are characterized by their linear dependence on reactant concentration over time. If plotting the natural logarithm (ln) of reactant concentration against time results in a straight line, it indicates that the reaction follows first-order kinetics.
Q2: Can you provide an example of a real-world application where understanding first-order reactions is important?
One practical application is radioactive decay, where isotopes decay at a constant rate following first-order kinetics. Understanding this concept is crucial in fields such as nuclear medicine, carbon dating, and environmental science.
Q3: Are there any limitations to using half-life in determining reaction progress?
While half-life provides valuable information about reaction rates, it should be noted that it only measures the time required for one-half of the reactant to be consumed. It does not give insight into other factors that may affect overall reaction progress.
Q4: How does temperature affect the half-life of a first-order reaction?
Increasing the temperature generally decreases the half-life of a first-order reaction. This is due to the fact that higher temperatures increase the rate constant, leading to faster reaction rates and shorter half-lives.
Q5: Can you explain why the half-life formula for zero order reactions differs from that of first-order reactions?
In zero order reactions, the concentration of reactant does not affect the rate of reaction. Therefore, the half-life formula is derived based on initial reactant concentration and rate constant. In contrast, first-order reactions exhibit a linear relationship between concentration and time, leading to a different mathematical derivation for their half-life formula.
Feel free to explore these FAQs for further insights into first-order reactions and half-life concepts.
References:
- Chemistry World. (2022, August 15). First Order Reaction: Half-life Calculations. Royal Society of Chemistry.



Leave a Reply