
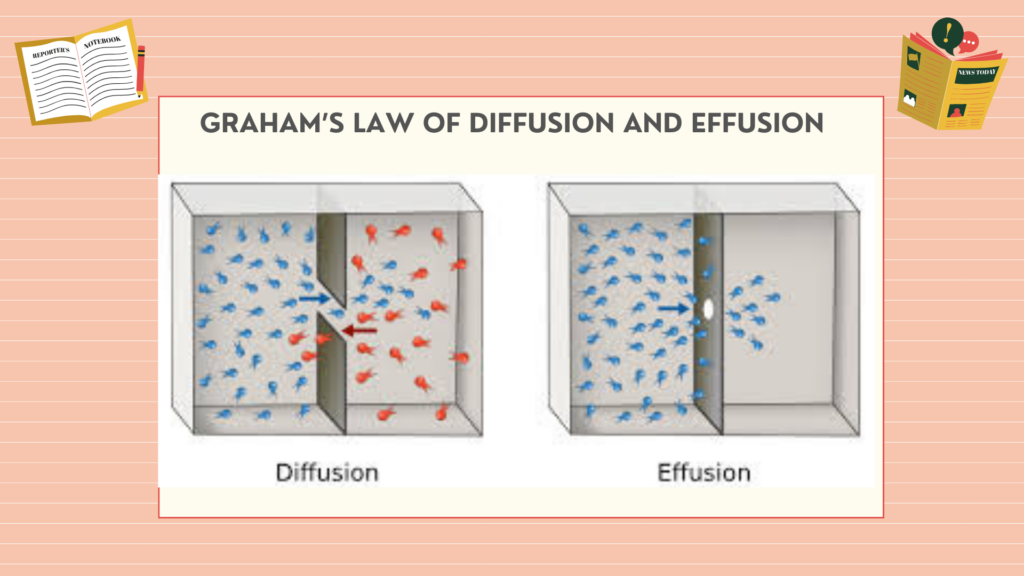
Graham’s law of diffusion explains the rate of diffusion. The movement of molecules (gases) from their higher pressure to their lower pressure is called diffusion.
For example, a drop of ink is added to the water glass, and a drop diffuses into the whole glass from higher pressure to lower pressure.
Graham’s law of Effusion is the movement of gas particles through a small hole.
For example, when the tire gets punctured, the air inside the tire effuse through a tiny hole. Graham’s law of diffusion is also valid for effusion.
Graham’s Law of Diffusion
According to this law, the rate of diffusion is inversely proportional to the under root of the molar mass of gas provided the pressure, temperature, and area remain constant.
Rate of diffusion (r) ∝ 1 / √molar mass
- The more the molar mass of the gas, the lesser the rate of diffusion.
- The higher the gas, the lesser the rate, or vice versa.
Among hydrogen and oxygen gas, which is more diffuse?
The molar mass of hydrogen and oxygen gas is 2 g and 32 g respectively. So H2 is five times more diffuse than O2 (provided P, T, and the area remains constant). The rate of diffusion depends upon the pressure, temperature, area, and molar mass.
r ∝ P A / √T M
Where P is pressure, A is the area, T is temperature, and M is the molar mass of the gas. But it is not Graham’s law because as we know that according to this law, temperature, pressure, and the area remains constant so, r ∝ 1 / √M
Case 1
- P = Constant
- A = Constant
- T =Constant
Let us have two gases, gas 1 and gas 2. The rate of diffusion of both gases is given,
r1 ∝ 1 / √M1 ———- (1)
r2 ∝ 1 / √M2 ———- (2)
Combine eq (1) and (2)
r1 / r2 = √ M2 / M1 ———- (3)
Where,
- r1 = rate of diffusion of gas 1
- r2 = rate of diffusion of gas 2
- M1 = molar mass of gas 1
- M2 = molar mass of gas 2
Graham’s law is also explained by the density of the gas. As we know,
density of gas = d = molar mass / 2
Let us two gases having density d1 and d2 so,
d1 = M1 / 2
M1 = 2 d1
d2 = M2 / 2
M2 = 2 d2
Putting the values of M1 and M2 in eq (3)
r1 / r2 = √ 2d2 / 2d1
r1 / r2 = √ d2 / d1
So Graham’s law of diffusion also states that
“The rate of diffusion is inversely proportional to the square root of the density of gas”
Practice problems for Applying graham’s law.
Find the ratio of the rate of diffusion of helium and methane gas under similar pressure, temperature, and area.
r1 / r2 = √ M2 / M1
rHe / rCH4 = √ MCH4 / MHe
The molar mass of He = 4
The molar mass of CH4 = 16
rHe / rCH4 = √ 16 / 4
rHe / rCH4 = 2 / 1
rHe : rCH4 = 2 : 1
Find the ratio of the rate of diffusion of He, CH4, and SO2.
r ∝ 1 / √M
r = k / √M
The molar mass of He, CH4, and SO2 is 4, 16, and 64 respectively.
rHe : rCH4 : rSO2= He : CH4 : SO2
rHe : rCH4 : rSO2= k / √MHe : k / √MCH4 : k / √MSO2
rHe : rCH4 : rSO2= k / √4 : k / √16 : k / √64
rHe : rCH4 : rSO2= 1 / 2 : 1 / 4 : 1 / 8
Multiply by 8
rHe : rCH4 : rSO2= 4 : 2 : 1
- He is lighter gas so the rate of diffusion is high.
- SO2 is heavier gas so the diffusion rate is low.
The rate of diffusion of an unknown gas is 1.73 times smaller than the rate of diffusion of O2. Find the molar mass of an unknown gas.
r1 / r2 = √ M2 / M1
rx / rO2 = √ MO2 / Mx
Let the rate of diffusion of O2 is 1.73
1 / 1.73 = √ 32 / Mx
1 / √3 = √ 32 / Mx
squaring on both sides
1 / 3 = 32 / Mx
Mx = 96 g
The mass of an unknown gas is 96 g.
Meaning of Rate of Diffusion
1) In terms of the volume of gas:
“Volume of gas diffuse of effuse per unit time”
r = volume of gas diffused or effused / time taken
r1 / r2 = (v1/t1) / (v2/t2) = √ M2 / M1
2) In terms of distance travels:
r = distance traveled by the gas/time taken
r1 / r2 = (d1/t1) / (d2/t2) = √ M2 / M1
3) In terms of moles:
r = no of moles effuse or diffuse / time taken
r1 / r2 = (n1/t1) / (n2/t2) = √ M2 / M1
20 ml of He diffused in 5 sec. Find the volume of O2 diffused through the same hole in 10 sec.
r1 / r2 = (v1/t1) / (v2/t2) = √ M2 / M1
rHe / rO2 = (vHe/tHe) / (vO2/tO2) = √ MO2 / MHe
rHe / rO2 = (20/5) / (vO2/10) = √ 32 / 4
rHe / rO2 = 4×10 / vO2 = √8
vO2 = 40 /√8
vO2 = 14.14 ml
24 g of H2 diffused in 5 sec. Find the mass of O2 diffused in 10 sec through the same hole.
No of moles of H2 = m/M = 24/2 = 12 g
No of moles of O2 = m/M = x/32
r1 / r2 = (n1/t1) / (n2/t2) = √ M2 / M1
(12/5) / (x/32 × 10) = √ 32 / 2
(12/5) / (x/32 × 10) = 4
(12/5) / (x/320) = 4
12/5 × 320/x = 4
3840 / 5x = 4
x = 192 g
“x” ml of H2 effused through a hole in 5 sec. Find the time taken by the same volume of O2 to effuse through same hole.
r1 / r2 = (v1/t1) / (v2/t2) = √ M2 / M1
rH2 / rO2 = (vH2/t) / (vO2/t2) = √ MO2 / MH2
(x/5) / (x/t) = √ 32 / 2
(x/5) × (t/x) = 4
t / 5 = 4
t = 20 sec
Let us a cylinder, we effuse NH3 from one end and effuse HCl from another end of the cylinder. When both gases combine dense white fumes (NH4Cl) is formed. Where the dense white fumes is formed?
NH3 + HCl → NH4Cl (dense white fumes)
As we know that, lighter the gas more the rate of diffusion, or vice versa.
r ∝ 1 / √M
The molar mass of NH3 and HCl is 17 g and 35.5 g so the rate of diffusion of NH3 is more so the dense white fumes formed near origin of HCl gas.
Case 2
- P = not constant
- A = Constant
- T = Constant
As we know that
r ∝ P A / √T M (If A and T is constant)
r ∝ P / √ M
Graham’s law of diffusion when pressure is not constant is:
r1 / r2 = P1/P2 √ M2 / M1
20 ml of He diffused through a hole in 10 sec at a pressure of 2 atm. Find the volume of CH4 diffused in same time at a pressure of 1 atm.
r1 / r2 = (v1/t1) / (v2/t2) = P1/P2 √M2 / M1
rHe / rCH4 = (vHe/t) / (vCH4/t2) = P1/P2 √MCH4 / MO2
(20/10) / (vCH4/10) = 2/1 √16 / 4
20 / vCH4 = 2 √4
20 / vCH4 = 2 × 2
V = 20 / 4
V = 5 ml
Let us a cylinder containing helium gas and methane gas having number of moles 2 and 1 respectively. Find the ratio of the rate if diffusion intially.
r1 / r2 = P1/P2 √ M2 / M1
According to Dalton’s law, the more the number of moles, the more will be pressure of the gas.
P1 / P2 = n1 / n2
rHe / rCH4 = nHe/nCH4 √ MCH4 / MHe
rHe / rCH4 = 2/1 √16 / 4
rHe / rCH4 = 2 × 2 / 1
rHe : rCH4 = 4 : 1
Factors Influencing the Rates of Diffusion and Effusion
Temperature Effects
Temperature plays a crucial role, As the temperature increases, so does the molecular motion within a material. This increased motion leads to faster rates of diffusion and effusion. Consequently, higher temperatures result in enhanced effusion rates.
Particle Size
The size of particles also influences both diffusion and effusion rates. Smaller particles have shorter distances to travel, which facilitates their movement through materials or across porous barriers.
In comparison, larger particles encounter more obstacles along their path due to their increased size. Therefore, smaller particle sizes contribute to faster rates of diffusion and effusion.
Pressure Differences
Higher pressure on one side creates a greater force pushing the molecules towards the other side, resulting in faster diffusion or effusion rates.
For instance, when there is a significant difference in pressure between two compartments separated by a porous barrier, molecules will move more rapidly from the high-pressure region to the low-pressure region.
Concentration Gradient
A concentration gradient refers to a difference in concentration levels between two regions. When there is a high concentration on one side and a low concentration on the other side of a barrier, molecules tend to move from areas of higher concentration to areas of lower concentration until equilibrium is reached. This movement occurs through diffusion or effusion processes depending on whether there are porous barriers involved or not.
Real-Life Examples and Applications of Graham’s Law
Helium-filled Balloons Deflating Quicker than Air-Filled Ones
One fascinating example that demonstrates the practical application of Graham’s Law is the deflation rate of helium-filled balloons compared to air-filled ones.
When we release a helium-filled balloon, we notice that it deflates much faster than an ordinary air-filled balloon. This phenomenon can be explained by Graham’s Law, which states that the rate of diffusion or effusion of a gas is inversely proportional to the square root of its molar mass.
Helium, being the second lightest element on the periodic table, has a significantly lower molar mass compared to the mixture of gases present in our atmosphere.
As a result, helium molecules diffuse or escape through the balloon material at a much higher rate than heavier air molecules. Consequently, this causes the helium-filled balloon to deflate quicker as more gas particles escape into the surrounding environment.
Designing Efficient Gas Masks for Harmful Gases Removal
Another noteworthy application of Graham’s Law lies in designing effective gas masks used for protection against harmful gases.
The principle behind these masks is to efficiently remove toxic substances from the air before they reach our respiratory system. By understanding Graham’s Law, scientists and engineers can select materials and design structures that allow for optimal diffusion and effusion rates.
Gas masks incorporate filters made from porous materials with microscopic pores that enable gas molecules to pass through while trapping harmful particles or chemicals. These filters are engineered based on specific molecular sizes and weights associated with different toxic substances.
By utilizing Graham’s Law, experts determine which materials and pore sizes will allow for efficient removal of hazardous gases while maintaining breathability.
Controlled Diffusion in Perfume Manufacturing
The controlled diffusion of fragrance compounds plays a vital role in perfume manufacturing processes. Perfumers need precise control over how quickly scents evaporate from their products so that they can create long-lasting and appealing fragrances.
Graham’s Law provides a fundamental understanding of how various fragrance molecules diffuse through the air.
Perfume manufacturers use this knowledge to select specific compounds with desired diffusion rates. For instance, top notes in perfumes are designed to evaporate quickly, creating an initial burst of fragrance upon application.
On the other hand, base notes are composed of heavier molecules that evaporate slower, providing a lasting scent throughout the day. By carefully considering the molar masses and diffusion rates of different fragrance components, perfumers can create well-balanced and captivating scents.
Summarizing Key Insights from Graham’s Law
Now that we have explored the concept of Graham’s Law and its relationship to diffusion and effusion in gases, let’s summarize the key insights we’ve gained.
Graham’s Law states that the rate of diffusion or effusion of a gas is inversely proportional to the square root of its molar mass.
In simpler terms, lighter gases diffuse or effuse faster than heavier ones. This fundamental principle has important applications in various fields, such as understanding gas behavior, predicting gas mixtures’ behavior, and even measuring the molecular weights of unknown substances.
By calculating rates of diffusion or effusion using Graham’s Law, we can gain valuable insights into how different factors influence these processes. We have also seen real-life examples where Graham’s Law plays a crucial role, like in perfume manufacturing and air quality monitoring.
Frequently Asked Questions (FAQs)
What is the difference between diffusion and effusion?
Diffusion refers to the process by which particles spread out from an area of high concentration to an area of low concentration. It occurs when gases mix together naturally due to their random motion.
On the other hand, effusion specifically refers to the escape of gas molecules through a small opening into a vacuum or another region with lower pressure.
How does temperature affect diffusion and effusion rates?
Temperature has a direct impact on both diffusion and effusion rates. As temperature increases, gas molecules gain more kinetic energy and move faster. This increased speed leads to higher rates of diffusion and effusion.
Can Graham’s Law be applied to all types of gases?
Graham’s Law applies specifically to ideal gases under ideal conditions. It assumes that there are no intermolecular forces between gas molecules and that their volumes are negligible.
In reality, some gases may deviate from the ideal behavior due to factors like molecular size or attractive forces.
Is Graham’s Law applicable only to gases?
Yes, Graham’s Law is applicable only to gases. It describes the behavior of gas particles and their rates of diffusion or effusion.
Liquids and solids have different mechanisms of particle movement and do not follow the same principles as gases.
How can understanding Graham’s Law be useful in everyday life?
Understanding Graham’s Law can have practical applications in various aspects of everyday life.
For example, it helps us understand how odors spread through a room, how gas leaks occur, or even how certain medications are delivered through inhalers.
By grasping this concept, you’ll gain insights into the behavior of gases that surround us every day.
References
Chemistry textbooks, such as “Chemistry: The Central Science” by Brown, LeMay, Bursten, and Murphy.



Comments (2)
ısıtıcısays:
September 27, 2024 at 3:52 amFor the reason that the admin of this site is working, no uncertainty very quickly it will be renowned, due to its quality contents. ısıtıcı
2. el güvenlik kamera fiyatlarısays:
September 27, 2024 at 9:26 amHi there to all, for the reason that I am genuinely keen of reading this website’s post to be updated on a regular basis. It carries pleasant stuff. 4 lü ıp kamera seti