

What is the halogenation of alkenes?
Halogenation is a process in which halogens which are Cl, Br, I, F are introduced in a molecule. In the halogenation of alkenes, the halogen atoms are added to the π bond system. A s the pi bond in the region of high charge density so it acts as the nucleophilic center. Now we will discuss the mechanism and steps involved in the halogenation of alkenes.
Mechanism of halogenation:
Alkenes and halogens are non polar. But under certain conditions, there can be the formation of the induced dipole, which causes the creation of attractive forces between the molecules. the mechanism of halogenation of alkenes involves the following steps:
Step 1 of halogenation of alkenes:
In the first step, the bond present between Br-Br is polarized. The covalent bond breaks. The Br with a positive charge forms a cyclic intermediate with two carbons of alkene.
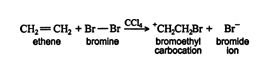
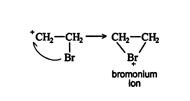
Step 2 of halogenation of alkenes:
In this step, the bromide anion having a negative charge attacks one of the carbon atoms of the brominium ion. Bromide anion attacks from the backside of the ring. The reason for this is that due to the formation of brominium ion it blocks the entire side and causes steric hindrance. This is why a trans addition is possible only.
As a result, the ring opens and a trans vicinal dihalide is formed.
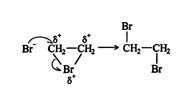
Commonly used halogens:
Mostly Br, Cl is used for these halogenation reactions. As my reaction is too slow due to its atom size. and F reaction is too fast and can be explosive.
Some examples:

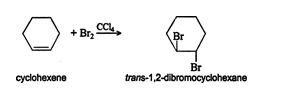
Due to the reason that bromide anion can attack any of the carbon from another side, it creates a variety of products.



Leave a Reply