
Hund’s rule, Aufbau’s principle, and Pauli’s exclusion rules are necessary for the filling of electrons. The distribution of electrons in the shells and subshell are done due to these three rules and concepts. Before the discussion of Hund’s rule, let us consider the base of Aufbau’s principle.
It tells us that electrons are filled in the lower energy orbit and then in the higher energy orbit. So, this rule is only applicable for the s-orbital but didn’t explain the filling of p-orbitals as it has three degenerate orbitals px, py, and Pz. To solve this mystery we will study Hund’s rule.
State Hund’s Rule?
According to Hund’s rule electrons are filled in the following two ways:
- Before occupying double-bonded electrons in the subshell, electron filling must be single-bonded.
- To maximize the total spin all the single-bonded electrons in different orbitals have the same spin.
Electrons were more likely to live in lower energy orbit because the orbit in low energy has greater stability. So, electrons are most probable to live in low energy state. But electrons are present in the same spin if they are unpaired.
So, these unpaired electrons have reduced the repulsive forces. In the ground state, if electrons are in paired form and the orbital is half-filled, they will never pair up to leave a vacant orbital to become partially filled because a half-filled is more stable than a partially filled atom.
HALF-FILLED >> PARTIALLY-FILLED
When another atom comes close to the first atom it will act as a magnet. If it is electron-deficient species then it will attract to it. But on the other hand, if it is an electron-rich atom it will repel each other.
Examples regarding Hund’s Rule
1. Nitrogen atom:
Let us consider an example of the nitrogen atom. Nitrogen has an atomic number of 7 (Z=8). So, its electronic configuration involves the arrangement of electrons in shells and sub-shells.
Nitrogen = (Z=7) ⇒ 1s2, 2s2, 2p3
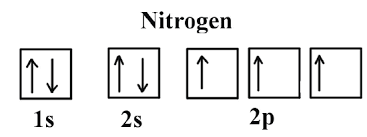
In this example, 1s and 2s are filled according to principle but the p-orbital is filled according to Hund’s rule. As we know, 3 electrons in 3 p-orbitals make it stable to others because its filling makes it half-filled stable.
2. Oxygen atom:
Now we will discuss the example of oxygen having the atomic number 8. So,
oxygen = (Z=8) ⇒ 1s2, 2s2, 2p4
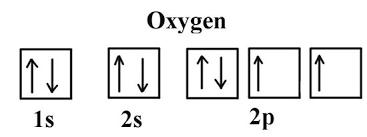
We have filled electrons in s by using Aufbau’s principle but in p-orbital it is filled first in px, then in py, and in the last Pz. But there is the exception that the 4th electron will enter px orbital after filling in Pz. When all orbitals are half-filled. So, px is paired with opposite spin to reduce maximum repulsion.
Explanation of Hund’s Rule
According to Hund’s rule electrons are filled by two rules stated above. The electron can fill in the lower energy orbit as well as empty orbitals. So, to minimize the repulsive forces electron should fill in the different orbitals to become unpaired. But the question arises that why an atom follows the rule of minimum repulsion?? IT IS BECAUSE THE FURTHER STUDIES HAVE PROVED THAT, QUANTUM MECHANICAL CLACULATONS PROVED THAT UNPAIRED ELECTRONS HAVE MORE Zeff AND SHIELDING EFFECT.
As the second rule discussed the same spin. To reduce repulsion and reduce spin multiplicity electrons must pair up in a single degenerate orbital with opposite spin. Since the opposite spin is shown by (⇅).
Hund’s rule related to Maximum multiplicity:
This rule for maximum multiplicity is based on the observation of atomic spectra. This rule determines the energy of orbitals by the spin multiplicity numbers. If degenerate orbitals of equal energy are present then electrons must fill in them by a single spin. This rule is proposed by Friedrick Hund in 1925.
If we talk about atoms, the spin multiplicity is 2s+1. s is the spin present in an orbital. The spin value of each electron present in an orbital is +1/2. The low energy state and increased stability occur due to the high spin state. There is given the wrong explanation of Low energy of high spin that different occupied spatial orbitals create a large volume distance between two electrons but the quantum mechanical calculation has proved that the actual reason for an increase in the stability is that the decrease in screening effect between electron by the nucleus.
So, this is the reason that explained why electrons filled in a lower energy state???
Relations of Hund’s rule by Aufbau principle
Hunds rule and the Aufbau principle have dissimilarities between them. Hund’s rule states that before an electron is doubled it should be single in different orbitals and Hund’s rule also gives the rule of spin multiplicity. But Aufbau’s principle states that electrons must fill in the lower energy state first and then in the lower energy state. It will follow the n+1 rule.
Relation of Hund’s rule to Pauli’s Exclusion
Hund’s rule is discussed above but Pauli’s exclusion principle states that two electrons in an atom do not have the same four types of quantum numbers.
What are the limitations of Hund’s rule??
Hund’s rule has no limitations except one. This rule does not predict the low energy configuration.



Leave a Reply