
This blog post will discuss “is NH3 acid or base? “. NH3 is also known as Ammonia, and it is also present in the gaseous state. Ammonia is a very technical and complicated molecule to be studied. This article will also learn different theories that prove NH3 acid or base. Some theories propose Ammonia’s acidic and fundamental nature due to many factors like removal of or acceptance of proton H+ or OH–, ka and kb values, their dissociation constant, and the presence of lone pair.
Ammonia is a gas that has a pungent smell, is colorless, and is non-flammable gas. If you discuss its composition, it is made up of one nitrogen atom and three hydrogen atoms. Due to a very electronegative atom and dissimilar atoms N and H., it is Polar because all-atom does not cancel out their effects and causes some of the dipole moment. According to the valence shell electron pair repulsion theory (VSEPR), its generic formula is AB3 type, and according to hybridization, it possesses sp3 hybridization and Trigonal bipyramidal geometry. It has a bond angle of 107.5 degrees due to the repulsion of lone pairs.
The actual angle of this geometry is 109.5 degrees. Ammonia can have strong hydrogen bonding between nitrogen and hydrogen atoms, so it can easily be liquefied. Ammonia is used as an active ingredient in agricultural fertilizers and is an excellent nitrogen source in plants and animals. It is a highly toxic gas, and its exposure can cause damage to the lungs or even cause death. It possesses many industrial uses, i.e., refrigerant, disinfectant chemicals, and preparation of fertilizers.
Is NH3 Acid or Base / Theories
Ammonia (NH3) is a highly complex compound to study because NH3 can act as a weak base, an acid, and an amphoteric species. Different theories of acid, and base concept, i.e., Arrhenius, Lowry-Bronsted, and Lewis’s idea of acid and base. There is an exception between all these concepts of acid and base. Some ideas explain its acidic or basic nature in water or aqueous medium; some explain its nature on behalf of the presence of lone pair. And some explanation on behalf of removal and acceptance of proton H+ ions.
NH3 is a weak base; however, its pH is 11 and acts as an amphoteric species. Amphoteric species is the species that behave as an acid and the base. Its basicity value (pkb) is 4.75, and it is miscible soluble in water. Three concepts of acid and bases will explain Ammonia’s acidic or basic nature.
1. Arrhenius Concept of Acids and Bases
This concept is only valid for the dissociation that occurs in the aqueous medium. According to the definition of Arrhenius acid and base:
- Acid is a substance that can donate a proton or H+ ion on dissociation in aqueous media, and the base is a substance that can donate OH- ions on the dissociation in the aqueous media.
- In other words, it is to be said that donation of H+ and OH- ions by an acid or a base cause increase in the concentration of H+ and OH- ions in the water.
According to the Arrhenius concept of acid and bases, NH3 acts as an acid because it forms conjugate base NH2+and the hydronium ion H3O+.
NH3 + H2O → NH2+ + H3O
It is concluded by this reaction that it will not act as Arrhenius base because the condition for Arrhenius base is not satisfied. The requirement states that the Arrhenius base will donate OH- ions in the aqueous media. In the formula of Ammonia, there is no unit of Oh- ion, just like NaOH, KOH, etc. So, it will not act as Arrhenius Base.
2. Lowry-Bronsted concept of Acid and Base
Lowry and Bronsted proposed the concept of acid and base, and they did not explain this concept in aqueous media. According to the Lowry-Bronsted idea of acid and base:
- Acid is a substance that donates a proton to another species, but the base is a substance that accepts a proton from Lowry-Bronsted acid.
- Acid is a strong species if its conjugate base is stable, and the base is a strong species only when its conjugate acid is much more stable.
Consider the following reaction:
HCl + NH3 → NH4+ + Cl–
In the reaction mentioned above, NH3 accepts a proton from HCl. So, according to the definition of this concept, NH3 acts as a base, and its conjugate acid is stable. So, this theory explains the basic nature of Ammonia NH3. It also acts as an acid, called an amphoteric species. “Amphoteric species is the species that behave as an acid as well as the base.”
3. Lewis Concept of Acid and Base
Lewis’s concept was given by the US scientist G. N Lewis in 1923, and he proposed this concept in his name. According to the idea of acid and base, Lewis acid and base can be defined as:
- Lewis Acid is a substance that can accept a pair of electrons from a donor and base is a substance that can donate a pair of electrons to an acceptor.
- Lewis Acid acts as an acceptor & lewis base acts as a donor. Lewis also proposed a theory based on pair of electrons.
- It is also concluded that Lewis acid is a species that is electron deficient but Lewis base is the specie that is rich in a number of electrons.
Consider the following reaction between Ammonia and boron trifluoride. In this reaction, NH3 acts as a Lewis Base because it donates the lone pair to the molecule of BF3 by a coordinate covalent bond. So, BF3 acts as a Lewis acid due to the acceptance of the electron pairs. To show the acceptance and donating of electron pairs on the paper we first have to make the Lewis structures of NH3 and BF3.
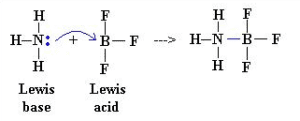
It is a neutralization process in which Lewis acid and base react with each other to form an adduct species. Nitrogen attacks on the 2p – orbital of boron to form a coordinate covalent bond.
Stability according to conjugate acid-base pair
Conjugate acid is similar to the Lowry bronsted base which can accept the proton and form when a Lowry bronsted acid donates a proton. But on the other hand, the conjugate base is similar to the Lowry bronsted acid which can donate a proton & form when a Lowry bronsted base accepts a proton. It is to be noted that:
- A strong acid produces a weak conjugate base.
- A strong base produces weak conjugate acid.
- A weak acid produces a strong conjugate base.
- A weak base produces strong conjugate acid.
As in the following reaction, it is noticed that NH3 is a weak base because it accepts a proton from acid, so it forms the conjugate acid. So, a weak base forms strong conjugate acid which satisfies that it is a strong base. NH4+ is a strong conjugate acid so by this concept NH3 is a weak base.
NH3 + H2O → NH4+ + OH–
Properties of Ammonia
Ammonia possesses many of the following properties that are discussed below:
- It has a molar mass of 17.031 g/mol.
- The boiling point of NH3 is -33.34 degrees and its melting point is -77.73 degrees.
- The dipole moment of NH3 is 1.42 D.
- Ammonia is lighter than air so can easily be liquified due to strong hydrogen bonding in nitrogen and hydrogen.
- The ammonia molecule undergoes nitrogen inversion at room temperature.
Uses of Ammonia
- Ammonia is used to manufacture many pharmaceutical products.
- It plays an important role in the agricultural field as a fertilizer.
- Many other chemicals like plastics and dyes are manufactured by using Ammonia.
- For the purification of water supply and as refrigerant its role is not ignorable.
- In the fermentation process, it is used to change the pH.
- Act as an antimicrobial agent and cleaning purposes.
- Also used to manufacture synthetic fiber like nylon and thread.
- It is also used as the precursor of the nitrogenous compound.
Conclusion
It is concluded that according to Arrhenius NH3 acts as an acid but other two concepts Lowry-bronsted and Lewis suggested that NH3 acts as a weak base because it possesses a strong conjugated acid and a lone pair on it. Also, NH3 is discussed as a whole compound in this article.


