
Before we discuss the CO2 lewis structure or lewis dot structure for CO2, we need to know the basics of lewis dot structure. Lewis dot structure works on the octet rule, which means that all the atoms would have eight electrons in their valence shell except hydrogen. Lewis’s theory is based that atoms share the electrons completing their octet and getting stability. The octet rule is to break for the central atom because the octet rule is not a universal rule, and in the central atom, more than or equal to 8 electrons in their valence shell.
Important Rules to Draw Lewis’s Structure
Rules For Selecting Central Atom
- The central atom is that which has least number in a compound or least electronegative or largest size or highest atomic number.
- Octect of corner atom must be complete.
- The central atom can have 8 electrons or more than 8 electrons in valence shell.
- The central atom tries to remain in maximum covalency. Covalancy means how many electrons share by atom or how many bond formed by atom.
- Oxygen has maximum 2 covalency.
- Sulphur has maximum 6 covalency.
- Nitrogen has maximum 5 covalency but it formed only four bond. Nitrogen can never be pentavalent.
- Phosphorus has maximum 5 covalency.
- Carbon has maximum 4 covalency.
- Silicon has maximum 4 covalancy.
- Chlorine has maximum 7 covalency.
These atoms can show covalency less than them, but they try to show their maximum covalency.
- Oxygen show 1 and 4 covalency.
- Sulphur show 2, 4 and 6 covalency.
- Nitrogen show 3 and 4 covalency.
- Phosphorus show 3 and 5 covalency.
- Carbon show only 4 covalency.
- Silicon show only 4 covalancy.
- Chlorine show 1, 3, 5 and 7 covalency.
5. If a positive charge is there (H3O+, NH4+), it will be on the central atom.
6. If a negative charge is there (SO42-, CO32-), it will be on the corner atom.
Calculations
First of all, we calculate Q (total electrons), equal to the valence electron of all-atom plus the number of negative charges minus the number of positive charges.
Q = Valance electron of all-atom + no of -ve charge – no of +ve charge
Then we find bond pair electrons equal to the number of bonds multiplied by two.
B.P e– = 2 × no of bonds
Then we find lone pair electrons by subtracting the bond pair electrons from total electrons Q.
L.P e– = Q – B.P e–
Lone pair electrons are used for both lone pairs as well as a double bond or triple bonds.
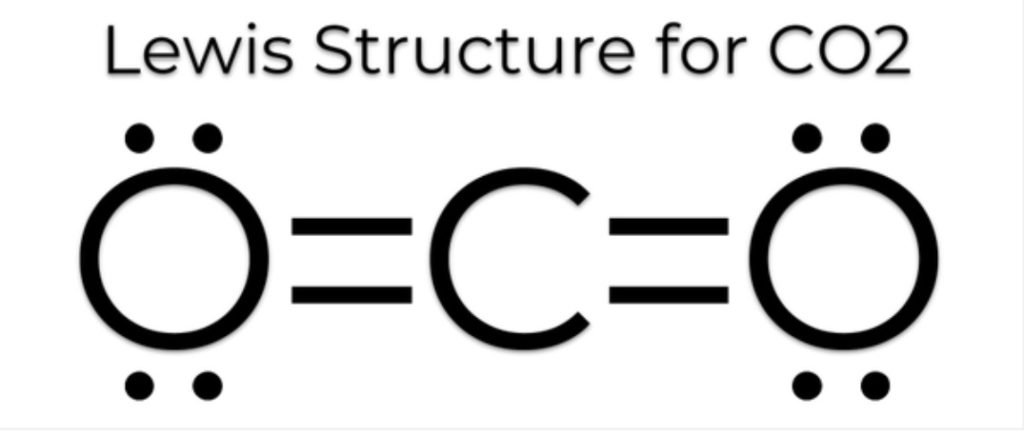
CO2 Lewis Structure
Now we learn how to draw a CO2 lewis structure, first, we find the total number of electrons by using the formula of Q.
Q = Valance electron of all-atom + no of -ve charge – no of +ve charge
Q = 16 + 0 – 0
Q = 16
B.P e– = 2 × no of bonds
B.P e– = 2 × 2
B.P e– = 4 e–
L.P e– = Q – B.P e–
L.P e– = 16 – 4
L.P = 12
Two lone pairs of both oxygen form bonds to complete the valency of carbon.
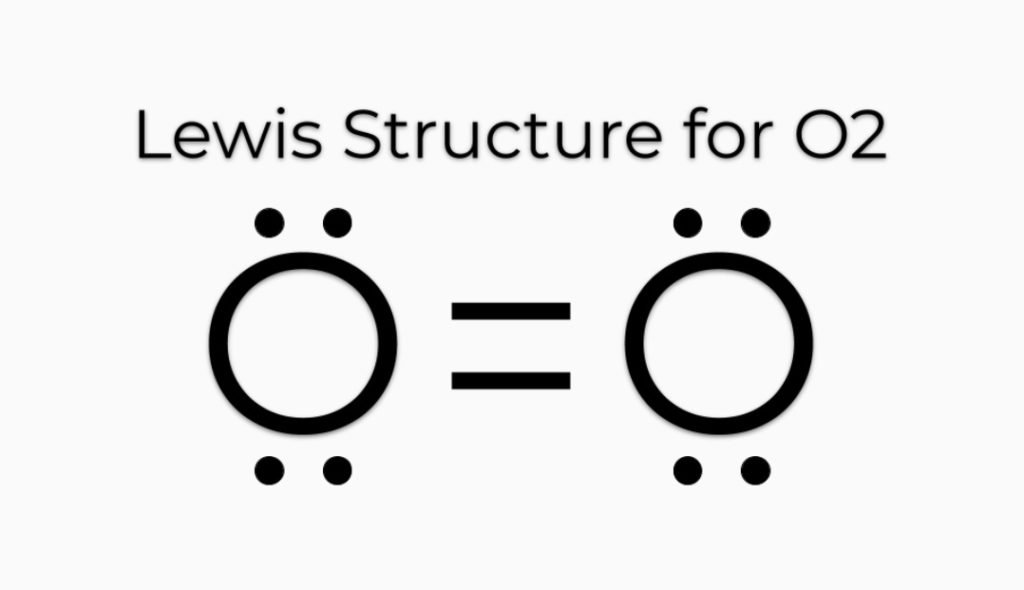
O2 Lewis Structure
O2 Lewis structure, oxygen is the diatomic molecule, and hence two atoms of the elements combine to form dioxygen. The oxygen has six electrons in its valance shell, and they try to gain or share two electrons to complete their octet and get stability.
By using the formula of Q, we can calculate the total electrons
Q = Valance electron of all atom + no of -ve charge – no of +ve charge
Q = (12 + 0 – 0)
Q = 12
B.P e– = 2 × no of bonds
B.P e– = 2 × 1
B.P e– = 2 e–
L.P e– = Q – B.P e–
L.P e– = 12 – 2
L.P e– = 10
First, we start to complete the octet by using the lone pair.
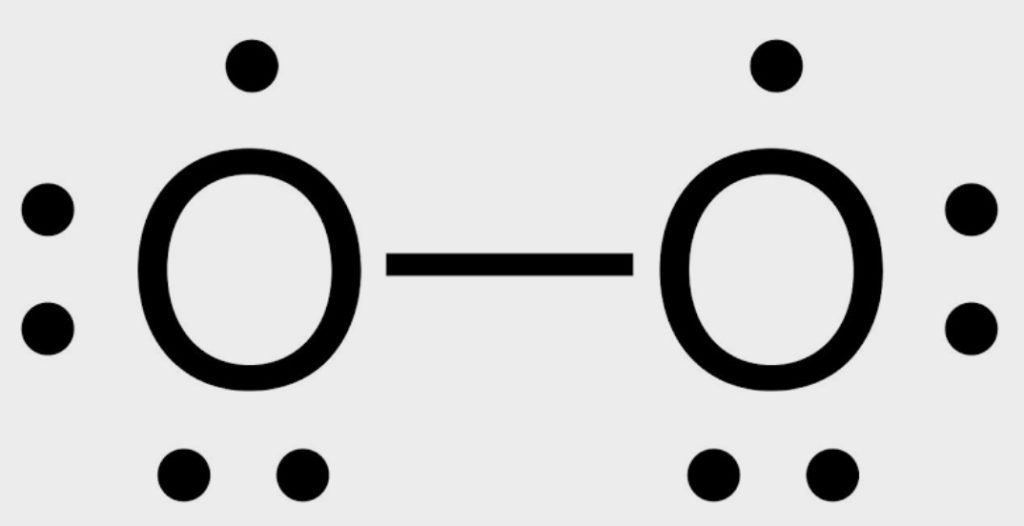
The octet of both oxygens is not complete. As we know, the lone pairs are also used for multiple bonds (double or triple) so, both oxygens share the electron and form a double bond.
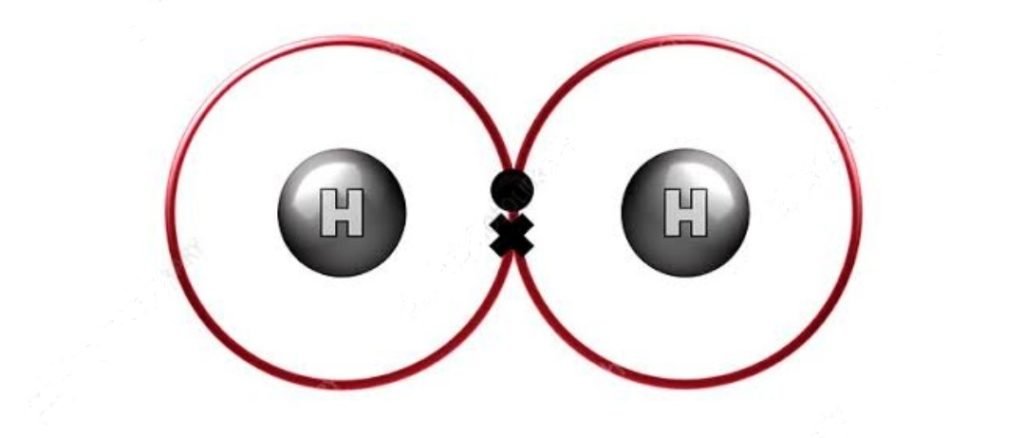
H2 Lewis Structure
H2 lewis structure, first, we calculate Q.
Q = Valance electron of all atom + no of -ve charge – no of +ve charge
Q = (2 + 0 – 0)
Q = 2
B.P e– = 2 × no of bonds
B.P e– = 2 × 1
B.P e– = 2 e–
L.P e– = Q – B.P e–
L.P e– = 2 – 2
L.P e– = 0
H3O+ Lewis Structure
H3O+ lewis structure, first, we calculate Q.
Q = Valance electron of all atom + no of -ve charge – no of +ve charge
Q = (9 + 0 – 1)
Q = 8
B.P e– = 2 × no of bonds
B.P e– = 2 × 3
B.P e– = 6 e–
L.P e– = Q – B.P e–
L.P e– = 8 – 6
L.P e– = 2
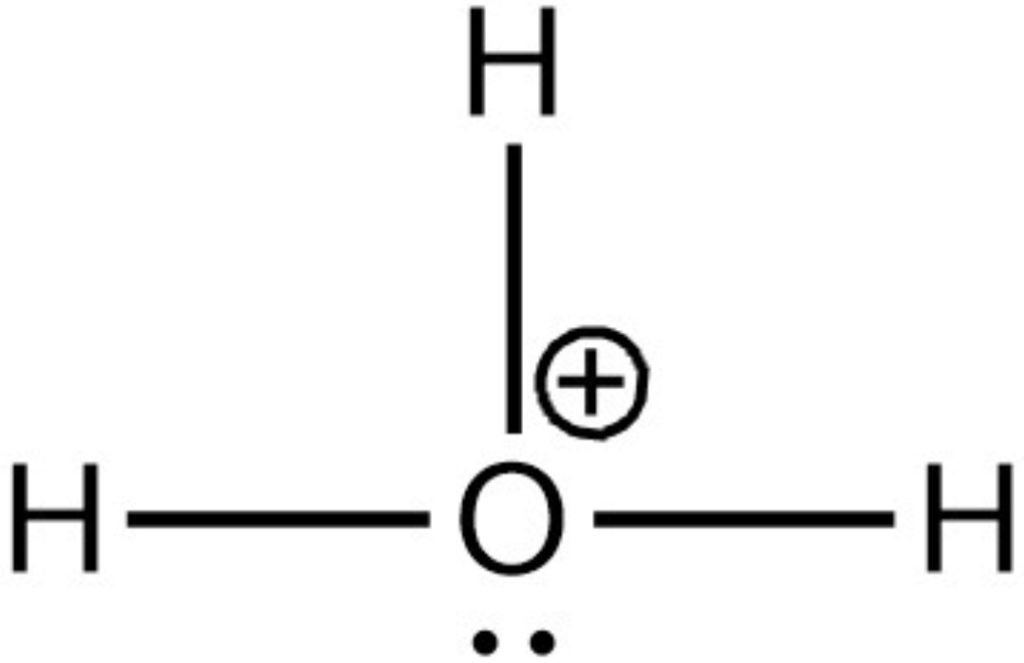
- All the three hydrogens has completed their duplet.
- Oxygen has 6 electrons firstly, so, we use two lone pairs to complete the octet of the central atom.
- The positive charge is always on the central atom.
NH4+ Lewis Structure
NH4+ lewis structure, first, we calculate Q.
Q = Valance electron of all-atom + no of -ve charge – no of +ve charge
Q = (9 + 0 – 1)
Q = 8
B.P e– = 2 × no of bonds
B.P e– = 2 × 4
B.P e– = 8 e–
L.P e– = Q – B.P e–
L.P e– = 8 – 8
L.P e– = 0

- All the four hydrogens has complete their doplet.
- The central atom nitrogen has completed its octet.
- The positive charge is always on the central atom.
NO3-1 Lewis Structure
NO3-1 lewis structure, first, we calculate Q.
Q = Valance electron of all atom + no of -ve charge – no of +ve charge
Q = (23 + 1 – 0)
Q = 24
B.P e– = 2 × no of bonds
B.P e– = 2 × 3
B.P e– = 6 e–
L.P e– = Q – B.P e–
L.P e– = 24 – 6
L.P e– = 18
We start to complete the octet of corner atoms by using lone pairs.
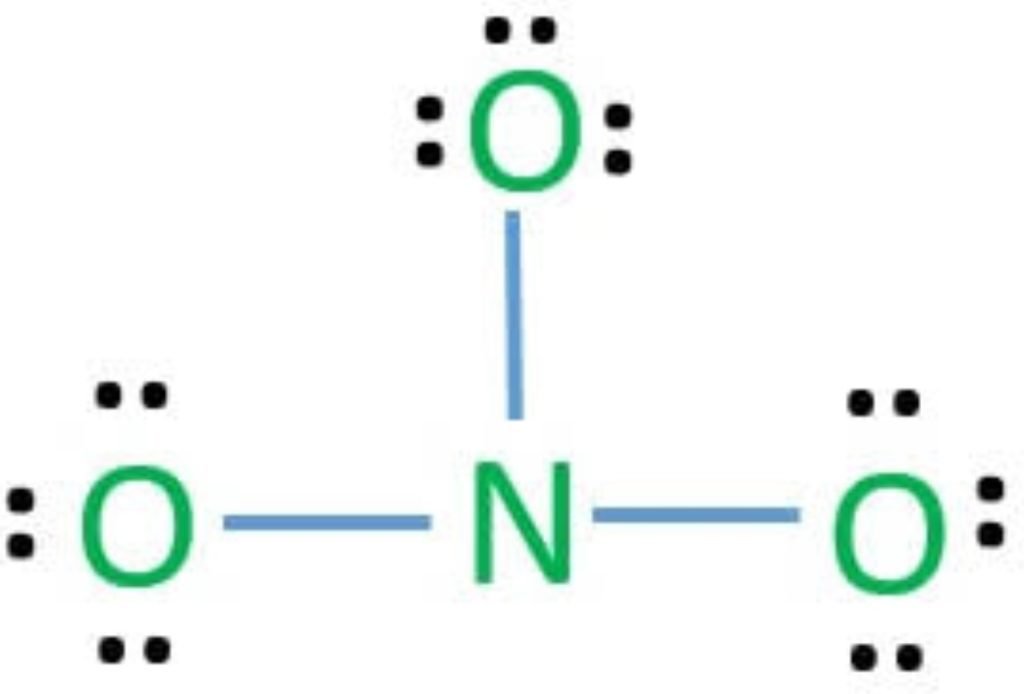
The octet of all three oxygens is complete, but the octet of nitrogen is not complete, so 2e-s of one oxygen share with nitrogen and make a double bond.
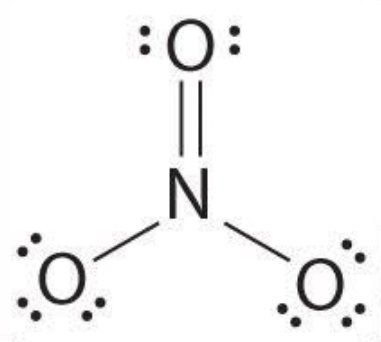
As we know, the negative charge is on the corner atoms, and there are three oxygen atoms on the corner so, we decided which atom has a negative charge with the help of the formal charge formula.
Formal Charge
F.C = Valance electrons – lone pair electrons – bond pair electrons/2
F.C(O1) = 6 – 4 – 4/2 = 6 – 4 – 2 = 0
F.C(O2) = 6 – 6 – 2/2 = 6 – 6 – 1 = -1
F.C(O3) = 6 – 6 – 2/2 = 6 – 6 – 1 = -1
F.C(N) = 5 – 0 – 8/2 = 5 – 0 – 4 = +1
So, the exact NO3-1 lewis dot structure is:
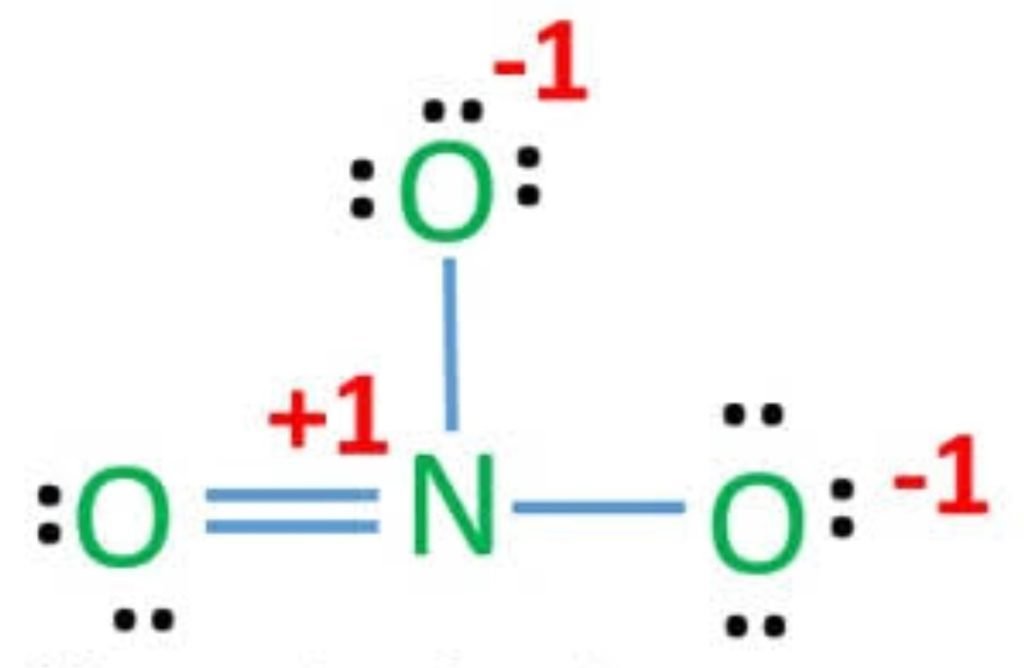
Note:
One of the negative charges on oxygen is cancelled with a positive charge on nitrogen.
NO2-1 Lewis Structure
NO2-1 lewis structure, first, we calculate Q.
Q = Valance electron of all atom + no of -ve charge – no of +ve charge
Q = (17 + 1 – 0)
Q = 18
B.P e– = 2 × no of bonds
B.P e– = 2 × 2
B.P e– = 4 e–
L.P e– = Q – B.P e–
L.P e– = 18 – 4
L.P e– = 14
By using lone pairs, we complete the octet of corner atoms. The octet of corner atoms is complete, but the octet of central atom nitrogen is not. So, one of two electrons of oxygen is shared with nitrogen and makes a double bond.
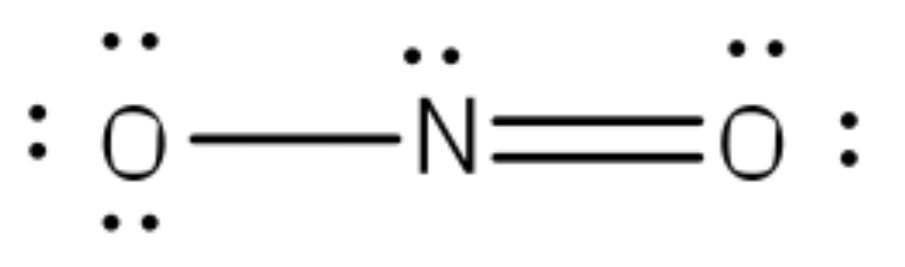
Formal Charge
F.C = Valance electrons – lone pair electrons – bond pair electrons/2
F.C(O1) = 6 – 4 – 4/2 = 6 – 4 – 2 = 0
F.C(O2) = 6 – 6 – 2/2 = 6 – 6 – 1 = -1
So, the exact NO2-1 lewis dot structure is:
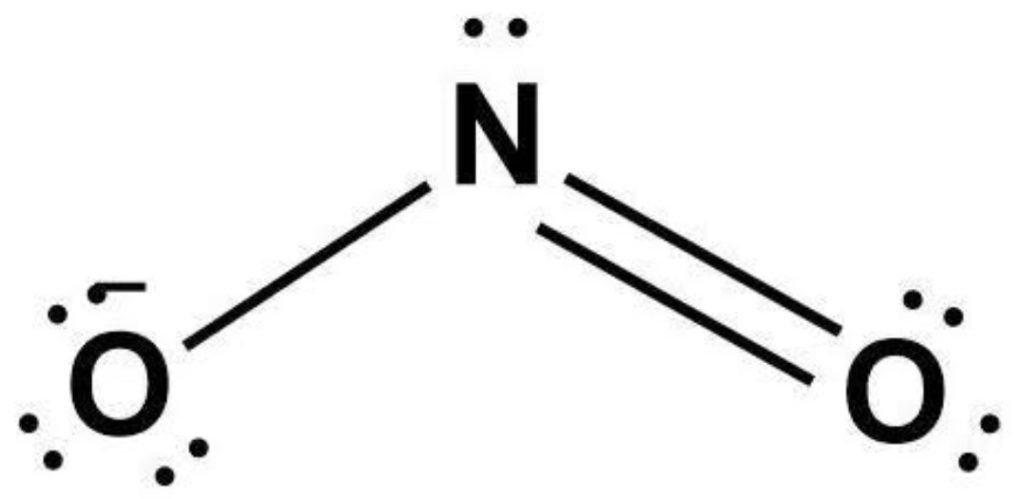
SO3 2- Lewis Structure
SO3 2- lewis structure, first, we calculate Q.
Q = Valance electron of all-atom + no of -ve charge – no of +ve charge
Q = (24 + 2 – 0)
Q = 26
B.P e– = 2 × no of bonds
B.P e– = 2 × 3
B.P e– = 6 e–
L.P e– = Q – B.P e–
L.P e– = 26 – 6
L.P e– = 20
We complete the octet of corner atoms by using lone pairs.

In the above structure, sulfur also has one lone pair. The octet of all three oxygen has complete (8 electrons). The octet of sulfur is also complete, but the structure is not actual because sulfur shows six covalencies. In the above structure, sulfur shows five covalencies so, one of the two electrons of oxygen is shared with sulfur and makes a double bond.
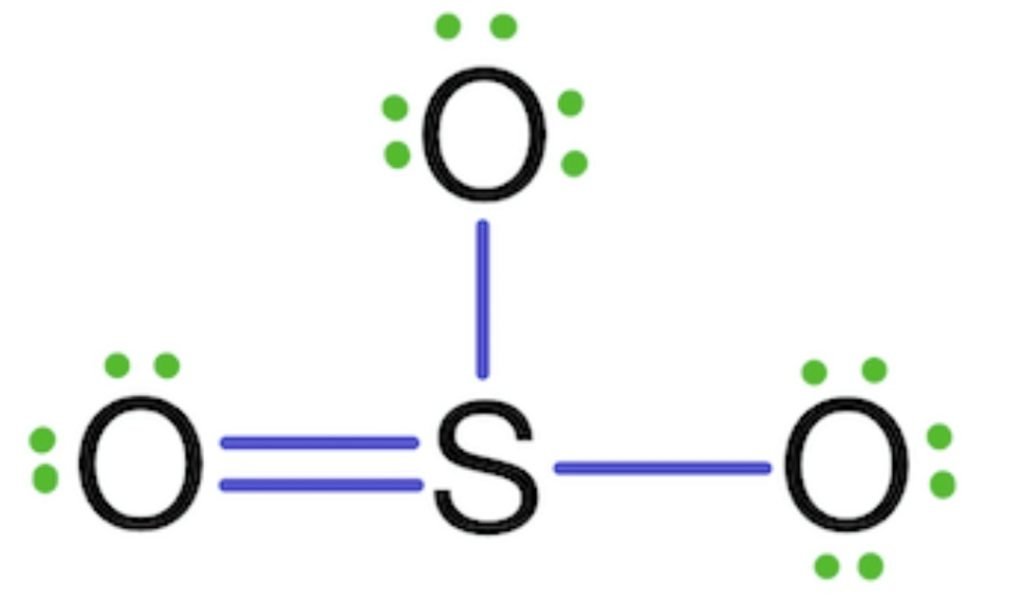
Formal Charge
F.C = Valance electrons – lone pair electrons – bond pair electrons/2
F.C(O1) = 6 – 4 – 4/2 = 6 – 4 – 2 = 0
F.C(O2) = 6 – 6 – 2/2 = 6 – 6 – 1 = -1
F.C(O3) = 6 – 6 – 2/2 = 6 – 6 – 1 = -1
F.C(S) = 6 – 2 – 8/2 = 6 – 2 – 4 = 0
So, the exact SO3 2- lewis dot structure is:
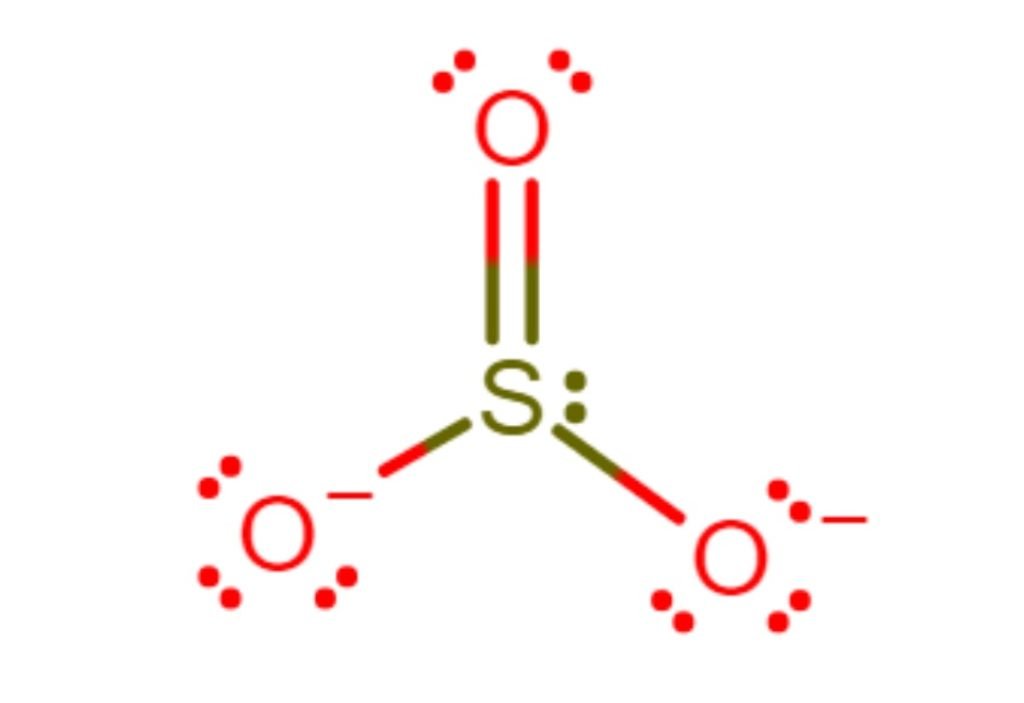
SO4 2- Lewis Structure
SO4 2- lewis structure, first, we calculate Q.
Q = Valance electron of all-atom + no of -ve charge – no of +ve charge
Q = (30 + 2 – 0)
Q = 32
B.P e– = 2 × no of bonds
B.P e– = 2 × 4
B.P e– = 8 e–
L.P e– = Q – B.P e–
L.P e– = 32 – 8
L.P e– = 24
We complete the octet of corner atoms by using lone pairs.
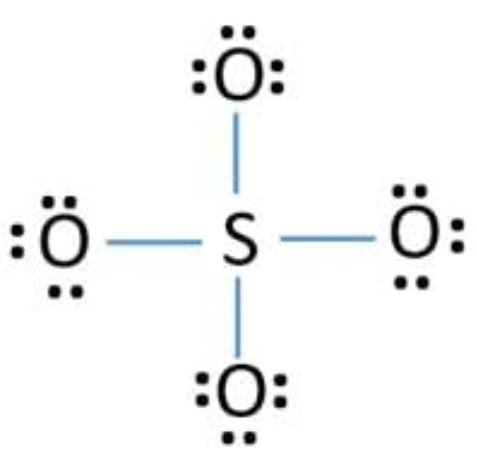
The octet of all four oxygen has complete 8 electrons. The octet of sulfur is also complete, but the structure is not so good because sulfur show six covalency, but in the structure, sulfur shows four covalencies, so oxygens are shared in lone pairs with sulfur and make two double bonds.
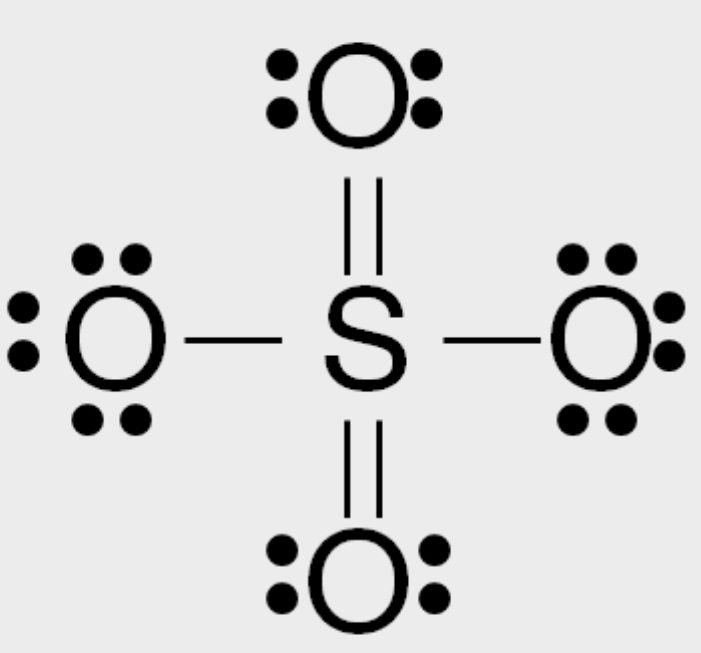
Formal Charge
F.C = Valance electrons – lone pair electrons – bond pair electrons/2
F.C(O1) = 6 – 4 – 4/2 = 6 – 4 – 2 = 0
F.C(O2) = 6 – 4 – 4/2 = 6 – 4 – 2 = 0
F.C(O3) = 6 – 6 – 2/2 = 6 – 6 – 1 = -1
F.C(O4) = 6 – 6 – 2/2 = 6 – 6 – 1 = -1
F.C(S) = 6 – 0 – 12/2 = 6 – 0 – 6 = 0
So, the exact SO4 2- lewis dot structure is:
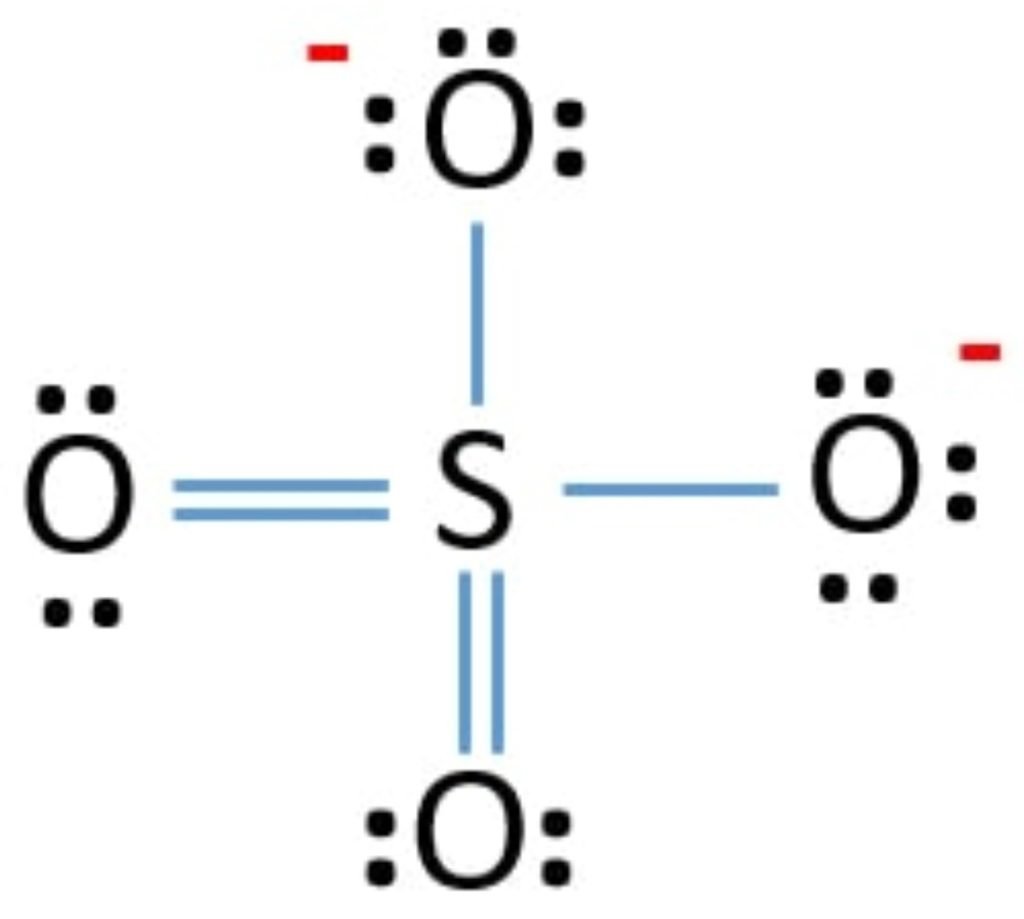
SO3 Lewis Structure
SO3 lewis structure, first, we calculate Q.
Q = Valance electron of all-atom + no of -ve charge – no of +ve charge
Q = (24 + 0 – 0)
Q = 24
B.P e– = 2 × no of bonds
B.P e– = 2 × 3
B.P e– = 6 e–
L.P e– = Q – B.P e–
L.P e– = 24 – 6
L.P e– = 18
We complete the octet of corner atoms by using lone pairs.
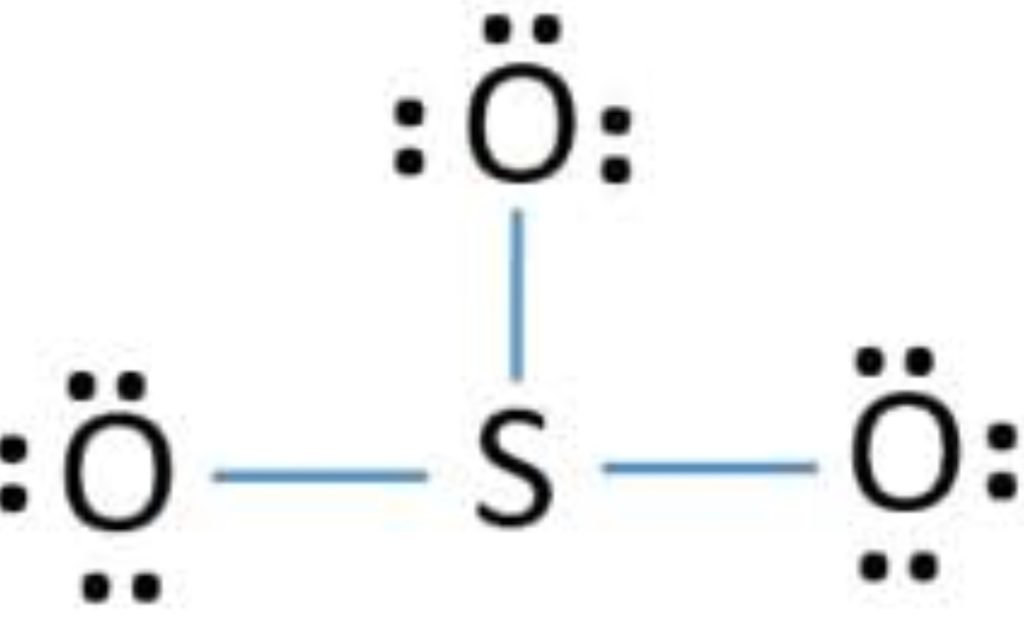
The octet of all three oxygen is complete, but the sulfur is not (6 electrons) so, the two electrons of oxygen are shared with sulfur and form a double bond.
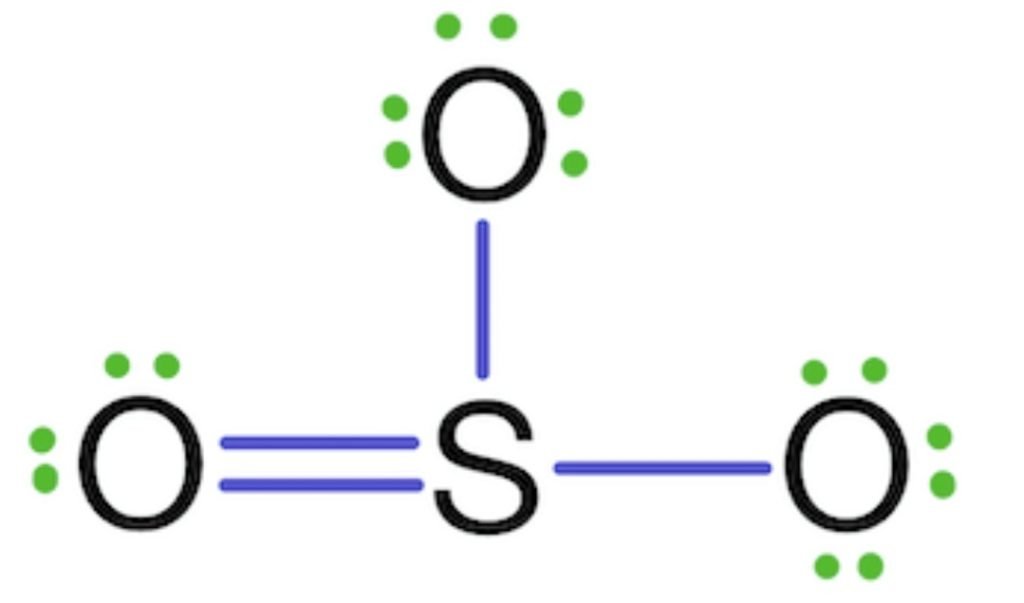
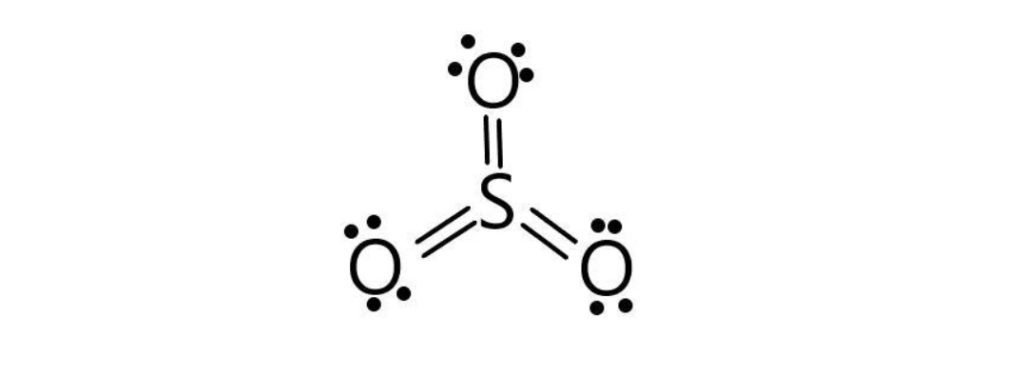
The octet of sulfur is complete, but as we know, sulfur shows six covalencies, but in the given above structure, sulfur shows four covalencies, so the two lone pairs of two oxygens are shared with sulfur form double bonds.
Formal Charge
F.C = Valance electrons – lone pair electrons – bond pair electrons/2
F.C(O1) = 6 – 4 – 4/2 = 6 – 4 – 2 = 0
F.C(O2) = 6 – 4 – 4/2 = 6 – 4 – 2 = 0
F.C(O3) = 6 – 6 – 4/2 = 6 – 6 – 2 = 0
F.C(S) = 6 – 0 – 12/2 = 6 – 0 – 6 = 0
So, the exact SO3 Lewis dot structure is:

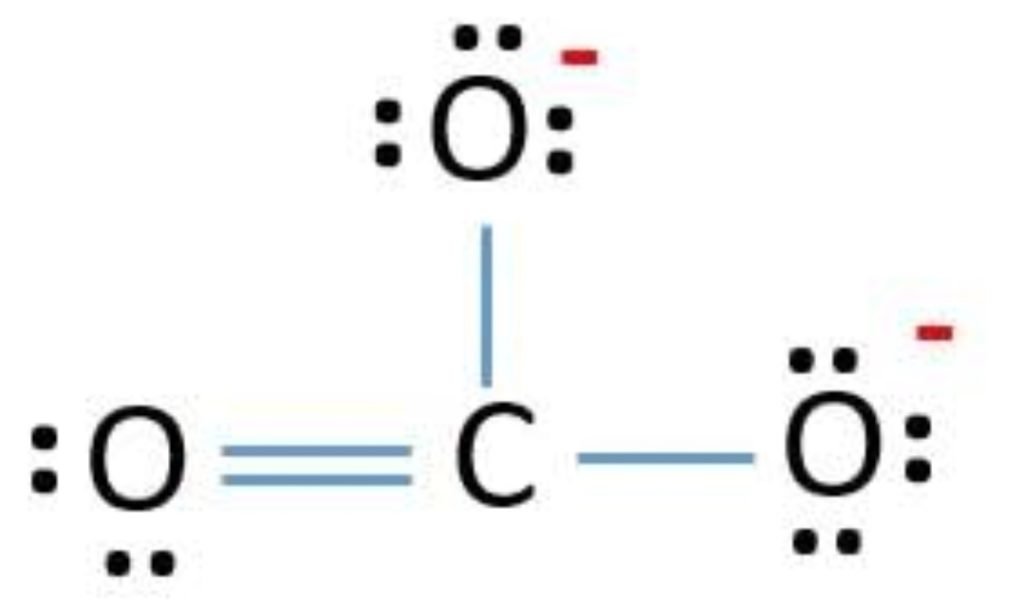
CO3 2- Lewis Structure
CO3 2- lewis structure, first, we calculate Q.
Q = Valance electron of all-atom + no of -ve charge – no of +ve charge
Q = (22 + 2 – 0)
Q = 24
B.P e– = 2 × no of bonds
B.P e– = 2 × 3
B.P e– = 6 e–
L.P e– = Q – B.P e–
L.P e– = 24 – 6
L.P e– = 18
We complete the octet of corner atoms by using lone pairs.
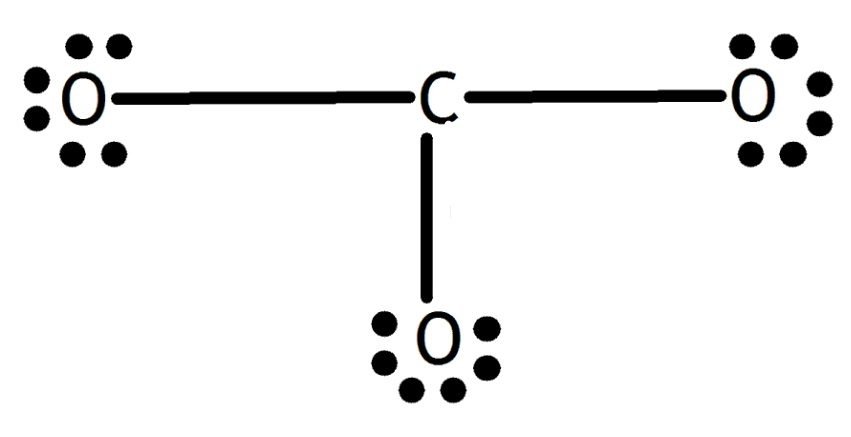
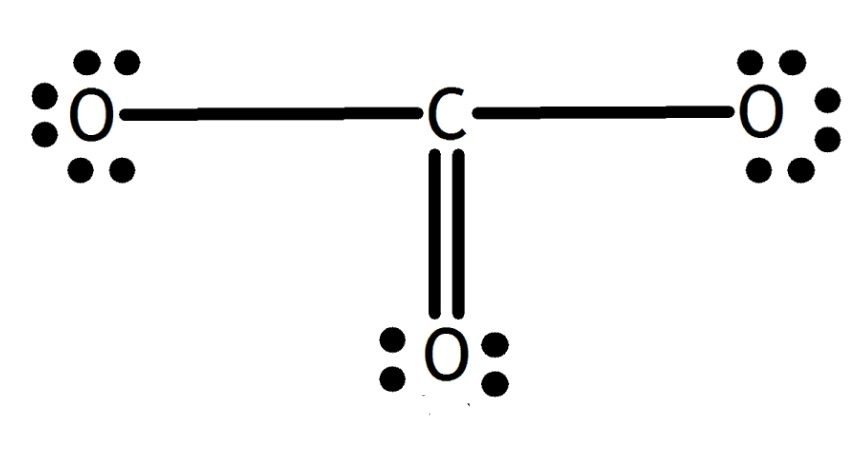
The octet of all three oxygen is complete, but the central atom carbon is not (6 electrons). So, one lone pair of oxygen is shared with carbon and forms a double bond.
Formal Charge
F.C = Valance electrons – lone pair electrons – bond pair electrons/2
F.C(O1) = 6 – 4 – 4/2 = 6 – 4 – 2 = 0
F.C(O2) = 6 – 6 – 2/2 = 6 – 6 – 1 = -1
F.C(O3) = 6 – 6 – 2/2 = 6 – 6 – 1 = -1
F.C(C) = 4 – 0 – 8/2 = 4 – 0 – 4 = 0
So, the exact CO3 2- Lewis dot structure is:
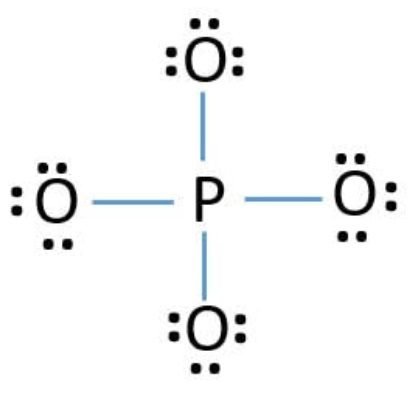
PO4 3- Lewis Structure
PO4 3- lewis structure, first, we calculate Q.
Q = Valance electron of all-atom + no of -ve charge – no of +ve charge
Q = (29 + 3 – 0)
Q = 32
B.P e– = 2 × no of bonds
B.P e– = 2 × 4
B.P e– = 8 e–
L.P e– = Q – B.P e–
L.P e– = 32 – 8
L.P e– = 24
We complete the octet of corner atoms by using lone pairs.
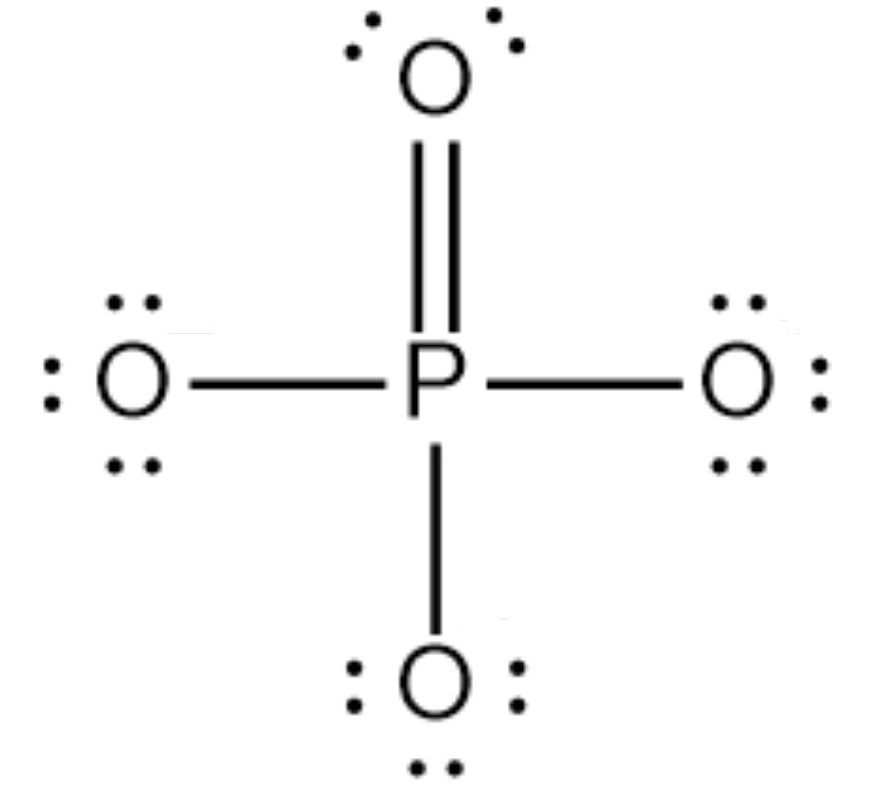
The octet of all four oxygens is complete, and the octet of phosphorus has also been complete. Still, phosphorus shows a maximum of 5 covalencies so, one of the two electrons of oxygen share with phosphorus and forms a double bond.
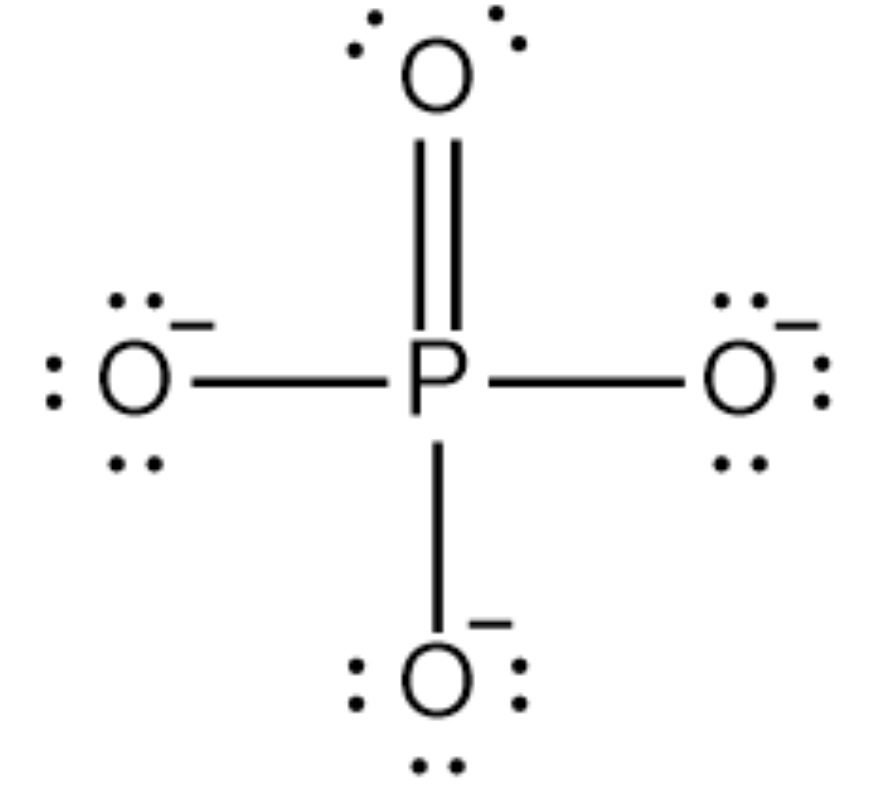
Formal Charge
F.C = Valance electrons – lone pair electrons – bond pair electrons/2
F.C(O1) = 6 – 4 – 4/2 = 6 – 4 – 2 = 0
F.C(O2) = 6 – 6 – 2/2 = 6 – 6 – 1 = -1
F.C(O3) = 6 – 6 – 2/2 = 6 – 6 – 1 = -1
F.C(O4) = 6 – 6 – 2/2 = 6 – 6 – 1 = -1
F.C(P) = 5 – 0 – 10/2 = 5 – 0 – 5 = 0
So, the exact PO3 3- Lewis dot structure is:
Note: Trick of Charge
- When oxygen shows 2 co-valency then there will be no charge on oxygen.
- When oxygen shows 1 covalency then oxygen has a -1 charge.
- When oxygen shows 3 co-valency then oxygen has a +1 charge. When nitrogen show 4 covalency then nitrogen has a +1 charge. Otherwise, the formula of formal charge is used to find out the charge on an atom.


