
Le Chatelier’s principle helps to understand how equilibrium accommodates or cancels the effect of changing variables temperature pressure and concentration. When a system at equilibrium is disturbed by changing variables of pressure temperature and concentration the system will try to minimize the effect of that change.
First of all, we will discuss equilibrium before we start Le-Chatelier’s Principle.
What is a chemical equilibrium state?
The chemical state at which the rate of forwarding reaction is the same as the rate of the reverse reaction, the composition of the reaction mixture remains constant this state is called chemical equilibrium state.
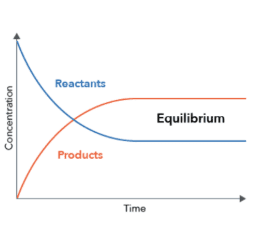
Le Chatelier’s principle:
Discovery:
A French Chemist Henry Le Chatelier in 1884 observed the effect of concentration, temperature, and pressure on a large number of systems in equilibrium. He gives his observations in the form of the generalized principle.

According to this principle:
If a system at equilibrium is disturbed by changes in variables such as temperature, pressure, and concentration. The system tries to adjust itself to decrease or minimize the effect of change as long as possible.
Importance:
This principle applies to all general and physical equilibria. Some applications of this principle are discussed in detail are given below.
Applications of Le Chatelier’s Principle:
Some applications of the Le-Chatelier principle are discussed here with the changes of variables temperature, pressure, and concentration.
1.Synthesis of Ammonia:
The synthesis of Ammonia has great importance in the industry.AS ammonia is used for the manufacturing of Nitrogen fertilizers such as Urea, ammonium nitrate, etc.
Process:
In the synthesis of ammonia by Haber’s Process, a mixture of nitrogen and hydrogen in the ratio of 1:3 by volume is passed through an iron coil lined inside with finely divided iron maintained at a constant temperature of 450 ℃.
It is exothermic process and equilibrium is given as:
N2 (g) + 3H2 (g) ⇋ 2NH3 (g) ∆H=-93.74KJ/mol
Initial Concentration:
1 3 0
Equilibrium Concentration Per unit volume:
1-α/V 3-3α/V 2α/V
Equilibrium constant(Kc):
Rate of forward Reaction:
Rf = Kf [N2 ] [H2]3
Rate of reverse reaction:
Rr =kr [NH3 ]2
Expression for equilibrium:
Kc= Rr / Rf
Kc = [NH3 ]2/[N2 ] [H2]3
Kc=[ 2α/V ]2 /[1-α/V][3-3α/V]
kc=4α2V2 /27(1- α )4
So this is the equilibrium constant expression of the reaction for the synthesis of ammonia.
Effects of changing pressure, temperature, and concentration:
Now we will study how the changes in variables like temperature, pressure, and concentration affect net production.
Effect of changing Pressure:
- The effect of change of pressure is relatively small on reactions that involve solids or liquid.
- In the case of a reaction involving gases, a very small change in pressure will affect the equilibrium state of the reaction largely.
- In the case of ammonia formation by Haber’s process, the forward reaction is favored by a decrease in the volume of the reaction mixture. By decreasing volume, the volume becomes four to two volumes.
- If the pressure is increased further on the system the volume will decrease furthermore.
so;
P α 1/V
And according to this Principle;
The reaction will move in the direction where there are volume decreases.
In other words, the formation of ammonia is favorable at high pressure. The reason behind this is that Haber’s process for the synthesis of ammonia is maintained at a high pressure of 200-600 atm.
Effect of changing the concentration:
Case 1:
If we increase the concentration of one of the components at equilibrium.
Then according to Le-Chatelier’s Principle, the equilibrium will shift in that direction to minimize the concentration of that substance.
Case 2:
If we decrease the concentration of one of the components at equilibrium.
Then according to this, the equilibrium will shift in that direction to increase the concentration of that substance in other words we say that to maintain equilibrium.
Example:
If we added more N2 Or H2 to the equilibrium mixture for the synthesis of ammonia. According to Le Chatelier’s principle then more ammonia is produced.
This is because by increasing the concentration of N2 Or H2 the equilibrium will shift in that direction to favor the reaction that proceeds with decreasing the number of molecules of N2 Or H2.
Hence by adding N2 Or H2 ammonia production will increases.
Effect of changing temperature:
By changing temperature equilibrium is also affected. For example the following reaction.
N2 (g) + 3H2 (g) ⇋ 2NH3 (g) + heat
In this reaction, a large amount of heat is produced.
The above reaction is exothermic in nature.
By increasing temperature:
By increasing temperature, the equilibrium shift in the reverse direction, and dissociation of ammonia occurs. There is a low yield of ammonia by increasing temperature.
By decreasing temperature:
Decreasing temperature the equilibrium will shift to the forward direction as the production of ammonia increases. So the production of ammonia is enhanced by decreasing the temperature.
The optimum temperature for the production of ammonia:
The optimum temperature for high production of ammonia is 450-500℃.
2.Formation of Sulphur Trioxide:
The formation of sulfur trioxide is another application of Le Chatelier’s Principle.
SO2 on reacting with oxygen gives SO3 the following reaction is given.
2SO2 (g) + O2 (g) ⇋ 2SO3 (g) ∆H=-189.11KJ/mol
- This is an exothermic reaction.
This reaction is taking place with a decrease in volume.
How the production will increase?
The net production will increase by ;
Increasing the concentration of any of the reactants favors the formation of SO3.AS by increasing pressure formation of SO3 will also increase. By decreasing temperature the synthesis of SO3 is increased. Because by increasing temperature the dissociation of SO3 occurs.
so production of SO3 is enhanced by :
- High concentration of SO2 and O2.
- High pressure.
- Low temperature (450℃)
3.Formation of Nitric Oxide:
The formation of Nitric Oxide is another application of this Principle. The reaction for the formation of Nitric Oxide is given below and how we will increase the production of Nitric oxide is also disscused here.
N2 + O2 ⇋ 2NO ∆H=180.74KJ/mol
- This is an endothermic reaction.
There is no change of volume that occurs during the formation of nitric oxide. So we can say that there is no effect of changes of equilibrium pressure. By increasing temperature the equilibrium will shift in that direction where heat is absorbed. The production of NO is increased by increasing temperature and by increasing the concentration of reactants.
So the production of NO will be enhanced by:
- Increasing temperature
- Increasing concentration of reactants
Buffer Solutions:
Buffers’ solutions also follow that principle. Buffer solutions are those which resist any change in pH. And buffer solutions work on it to resist any change in its pH.
Numerical Problem:
For the reversible reaction N2 (g) + 3H2 (g) ⇋ 2NH3 (g) at 500 ℃ .The value of Kp with partial pressure in atm is 1.44×10-5 at low pressure, the gases behave almost ideally. Calculate the corresponding value of Kc with concentrations in moles per liter.
Given:
T=500+273=773K
∆n=2-4=-2
R=0.082 litre atm /deg mol
Kp=1.44×10-5
solution:
Kp=Kc(RT)∆n
putting values…
1.44×10-5 =Kc(0.082 × 773)-2
Kc= 1.44×10-5 / (0.082 × 773)-2
Kc=5.79 × 10-2
Conclusion:
There is a complete overview of this principle. How we can use this principle to increase our industrial production. It has significant importance in industries.
Related Questions:
How Le Chatelier Principle used in industries?
This principle has significant importance in industries as the reaction scheme involves variables like pressure temperature, the concentration of species a change in each variable leads to equilibrium shifting and results in undesired production.
Does this principle apply to solids?
Le Chatelier’s principle is that if we apply any stress on a system at equilibrium. Then equilibrium will shift to that direction to remove stress. It does not apply to solids. Because adding or removing a solid from a system at equilibrium does not affect the position of equilibrium.
Give an example to prove that it does not apply to solids?
For example the following reaction at equilibrium;
2HgO(s) ⇋ Hg(l) + O2(g)
You might think that if you add more HgO to equilibrium then equilibrium will be shifted to the right. But you would certainly have more HgO on left but the density of HgO does not change. So by adding or removing a solid from a system at equilibrium does not affect the position of equilibrium.



Leave a Reply