
History:
Noble gases contain helium, neon, argon, krypton, Xenon, and radon. None of the noble gases was known when Mendeleev proposed his periodic table. In 1892, The English Scientist Willaim Ramsay became interested in the discovery.

| Born | 2 October 1852, Glasgow, UK |
| Died | 23 July 1916, High Wycombe, UK |
| Known for | Noble gases |
| Discovered | Argon, Neon, Xenon, Krypton |
William Ramsay’s work led to groundbreaking advances in thermodynamics and nuclear physics. While searching for Argon, Ramsay found Helium, which had been previously thought to exist only in the sun. Ramsay’s 1896 book ”The gases of the Atmosphere” predicted the existence of at least three more noble gases. Reducing air to low temperature at high pressure, his team proceeded to identify Neon, Krypton, and Xenon, reshaping the periodic table of elements forever. It is considered the greatest chemical discovery of his time. Sir William Ramsay was a Scottish chemist who discovered the noble gases and received the Noble Prize in Chemistry in 1904.
Discovery of Argon-The first Noble gas:
In 1898, William Ramsay and Lord Rayleigh discovered Argon. Sir Lord Rayleigh is a professor of physics in the cavendish laboratory England, accurately determines the atomic masses of the elements. Lord Rayleigh obtain Nitrogen from two methods.
- By Thermal decomposition of ammonia
2NH3 → N2 + 3H2
- By seperated Nitrogen from air. (He get Nitrogen, Oxygen, Carbon dioxide, Water vapours from air)
The nitrogen obtained from the air had a slightly higher density than that prepared by chemical reactions. William Ramsay reacts with red hot magnesium as a result, magnesium nitride is formed. After careful investigation, he concluded that higher density must be due to the presence of unknown gas. When he separated this gas from the air, he found that it was completely unreactive. William Ramsay and Lord Rayleigh get the spectrum of unknown gas with the help of Sir William Crookes. (Sir William Crookes is the inventor of discharge tube)
William Ramsay and Lord Rayleigh match the emission spectrum of an unknown element with any known element, its spectrum is different. They realize that it was a new element and called Argon. Argon is a Greek word that means lazy gas. (Atomic mass=39.5amu)
Discovery of Helium:
Helium was discovered by Frankland and Lockyer in 1868 in a new bright-line, D3 in the yellow region of the son’s atmosphere. Lockyer and Frankland observed a new yellow light in the chromosphere of the sun, during a total solar eclipse. This line did not identical with D1 and D2 lines of sodium and Lockyer called it the D3 line. This shows the presence of a new element. It was named helium. The word helium is derived from the Greek word helios, meaning sun.
On the earth, it was discovered by Ramsay. William Ramsay’s work led to groundbreaking advances in thermodynamics and nuclear physics. While searching for Argon, Ramsay found Helium, which had been previously thought to exist only in the sun. In 1898, Ramsay isolate Helium from Uranium ore. He was convinced that there were other unreactive gases and they were all members of one group in the periodic table.
Discovery of Xenon, Neon, Krypton:
After this, three more elements Xenon, Neon, and Krypton are discovered in just three months. These gases are discovered by Ramsay and his student Moriss Traverse. They used the refrigerator machine to first produce liquid air (liquification of gas). Applying a technique called fractional distillation. Allow the liquid to warm up gradually collected components at different temperatures.
Introduction of Noble gases:
What do party balloons, neon signs, and certain light bulbs have in common they are all filled with a noble gas in this.

Noble gases are group 18 elements consists of Helium, Neon, Argon, Krypton, Xenon, and Radon. All these are gases and chemically unreactive. They form very few compounds because of this they are termed noble gases. These elements are notable for having a full valance shell of electrons. Helium has two electrons in its valance shell where all other noble gases each have eight valance electrons. The noble gases all have full valance shells which makes them very stable. Noble gases are also known as rare gases because only 1% of noble gases occur in the atmosphere.
Are all noble gases are inert gases?
In the past, chemists thought that noble gases could not react with other elements that’s why they were called inert or unreactive gases. He, Ne, Ar are inert gases due to the less shielding effect and have a strong force of attraction of nucleus on outermost shell electrons. However, we know today that some noble gases can indeed react to form some stable compounds. Kr, Xe, Rn reacts with oxygen and fluorine under extreme conditions. so, all inert gases are noble gases but all noble gases are not inert.
Occurrence:
All the noble gases except radon occur in the atmosphere. Their atmospheric abundance in dry air is nearly 1% by volume of which argon is the major constituent. Helium and sometimes neon is found in minerals of radioactive origin. e.g. pitchblende, monazite, cleveite. The main commercial source of helium is natural gas. Xenon and radon are the rarest elements of the group. Radon is obtained as a decay product of 226Ra.
88226Ra → 86222Rn + 24He
| Property | He | Ne | Ar | Kr | Xe | Rn* |
|---|---|---|---|---|---|---|
| Atomic number | 2 | 10 | 18 | 36 | 54 | 86 |
| Atomic mass | 4.00 | 20.18 | 39.95 | 83.80 | 131.30 | 222.00 |
| Electronic configuration | 1s1 | [He] 2s2 2p6 | [Ne] 3s2 3p6 | [Ar] 4s2 4p6 | [Kr] 5s2 5p6 | [Xe] 6s2 6p6 |
| Atomic radius (pm) | 120 | 160 | 190 | 200 | 200 | – |
| Ionization enthalpy (KJ/mol) | 2372 | 2080 | 1520 | 1351 | 1170 | 1037 |
| Electron gain enthalpy (KJ/mol) | 48 | 116 | 96 | 96 | 77 | 68 |
| Density (at STP g/cm3 ) | 1.8×10-4 | 9.0×10-6 | 1.8×10-3 | 3.7×10-3 | 5.9×10-3 | 9.7×10-3 |
| Melting point (K) | – | 24.6 | 83.8 | 115.9 | 161.3 | 202 |
| Boiling point (K) | 4.2 | 27.1 | 87.2 | 119.7 | 165 | 211 |
| Atmospheric content (% by volume) | 5.24×10-6 | – | 1.82×10-4 | 0.934 | 1.14×10-6 | 8.7×10-6 |
Ionization Enthalpy:
Due to stable electronic configuration, these gases exhibit very high ionization enthalpy. However, it decreases down the group with an increase in atomic mass.
Atomic radii:
Atomic radii increase down the group with an increase in atomic number.
Electron gain enthalpy:
Since noble gases have a stable electronic configuration, they have no tendency to accept the electrons and therefore have large positive values of electron gain enthalpy.
Physical properties:
All noble gases are monoatomic. They are colourless, odourless and tasteless. They are sparingly soluble in water.
Melting point and Boiling point α Van der Waal forces
Van der Waal forces α Molecular weight
As we move from top to bottom in a group, molecular weight increases, therefore melting point and boiling point also increases. They have a very low melting point and boiling point because the only type of interatomic interaction in these elements is weak dispersion forces. Helium has the lowest boiling point at any known substance. It has an unusual property of diffusing through most commonly and laboratory materials such as rubber, glass or plastic. Some important points about noble gases are following:
- Noble gases or rare gases are placed in the zero groups.
- These have eight electrons in the valance shell of each element except helium.
- Noble gases are isolated from air by fractional distillation.
- Helium is present on earth as radioactive decay, the second most abundant element in the universe.
- Neon is 1/65000 the part of the earth.
- Neon gives a reddish glow in the gas discharge tube.
- Neon has 40 times more refrigeration capacity than helium.
- Xenon is present in the air at 0.08 ppm.
- Radon is a product of α-decay of radium.
- Boiling points increase down the group due to an increase in London forces.
Chemical properties:
In general, noble gases are unreactive or least reactive. The inertness of noble gases is due to two reasons:
- The noble gases have complete their octet (ns2 , np6 ) except Helium.
- They have high ionization energy and more positive electron gain enthalpy.
For few years, all attempts to force noble gases to react to form stable compounds were unsuccessful. After that in 1962, Neil Bartlett observed the reaction of a noble gas. First, he prepared a red compound which is formulated as O2+pt F6–. Then he realizes that the first ionization enthalpy of molecular oxygen is 1175 KJ/mol which is almost identical to that of Xenon (1170 KJ/mol). He tries his best to prepare some type of compounds with Xenon and was successful in preparing another red colour compound Xe+pt F6– by reacting ptF6 and Xenon. After this discovery, a number of Xenon compounds have been synthesised mainly with most electronegative elements like Fluorine and Oxygen.
The compound of Krypton is fewer. Only one compound of Krypton is discovered (KrF2 ). The compounds of radon have not been isolated by only identified e.g. (RnF2 ) by radiotracer technique. No compounds of Argon, Neon, and Helium are yet known.
Xenon fluorine compounds:
Xenon forms three binary fluorides XeF2, XeF4 and XeF6 by using the direct reaction of elements under appropriate experimental conditions.
Xenon Difluorine XeF2 :
When xenon is in excess, XeF2 is prepared by heating xenon and fluorine in a nikal tube at 673K and 1bar pressure.
Xe(g) + F2 → XeF2 (S)
It can be obtained by irradiating a mixture of Xe and Fluorine with light from a high-pressure mercury arc. It can be made by the action of dioxygen difluoride on xenon at -118℃.
Xe + O2F2 → XeF2 +O2
Xenon tetrafluoride XeF4 :
XeF4 is prepared by heating a mixture of xenon and fluorine (1∶5) in a nikal vessel at 873K and 7bar pressure.
Xe(g) + 2F2 → XeF4 (s)
when XeF4 react with xenon, XeF2 produces.
XeF4 + Xe → 2XeF2
Xenon herxafluoride XeF6 :
XeF6 is prepared by heating a mixture of xenon and fluorine (1∶20) in a nikal vessel at 573K and 70bar pressure.
Xe(g) + 3F3 → XeF6 (S)
XeF6 can also be prepared by the interaction of XeF4 and O2F2 at 143k.
XeF4 + O2F2 → XeF6 + O2
XeF2, XeF4 and XeF6 are colourless crystalline solids and sublime readily at 298K. They are powerful fluorinating agents. They are hydrolysed even by traces of water. For example, XeF2 is hydrolysed to give Xe, HF, and O2.
2XeF2 (S) + 2H2 O → 2Xe(g) + 4HF(aq) + O2 (g)
The structure of three xenon fluorides can be deduced from the VSEPR theory. XeF2 and XeF4 have linear and square planner structures respectively. XeF6 has six bond pairs and one lone pair thus, have a distorted octahedral structure.
Xenon fluorides react with fluoride ion acceptors to form cationic ions and fluoride ion donors to form fluoroanions.
XeF2 + PF5 → [XeF]+ + [PF6]–
XeF4 + SbF5 → [XeF3]+ + [SbF6]–
XeF6 + MF → M+ + [XeF7 ]– (M= Na, K, Rb, Cs)
Xenon oxygen compounds:
Xenon trioxide XeO3:
Hydrolysis of XeF4 and XeF6 with water gives XeO3.
6XeF4 + 12H2O → 4Xe + 2XeO3 +24HF + 3O2
XeF6 + 3H2O → XeO3 + 6HF
Partially hydrolysis of XeF6 gives oxyfluorides XeOF4 and XeO2F2.
XeF6 + H2O → XeOF4 + 2HF
XeF6 + 2H2O → XeO2F4 + 4HF
XeO3 is a colourless explosive solid and has a pyramidal molecular structure. XeOF4 is a colourless volatile liquid and has a square pyramidal molecular structure.
Xenon tetraoxide XeO4 :
XeO4 is obtained from the action of sulphuric acid on barium peroxenate.
Ba2XeO6 + 2H2SO → XeO4 + 2BaSO4 + 2H2O
Hybradization of Xenon compounds:
Hybradization of XeF2 :

| Name of molecule | Xenon difluoride |
| Hybradization type | sp3 d |
| Geometry | Trigonal bipyramidal |
| Shape | Linear |
| Bond pair | 2 |
| Lone pair | 3 |
Hybradization of XeF4:
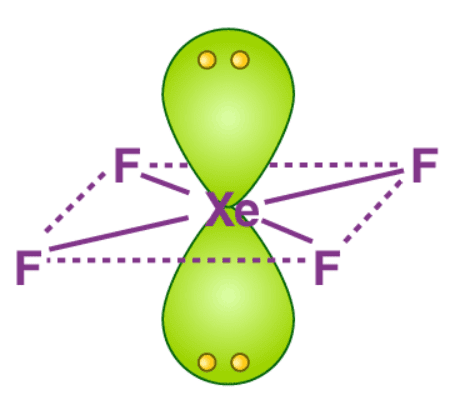
| Name of molecule | Octahedral |
| Hybradization type | sp3 d2 |
| Geometry | Octahedral |
| Shape | Square planner |
| Bond pair | 4 |
| Lone pair | 2 |
Hybradization of XeF6 :
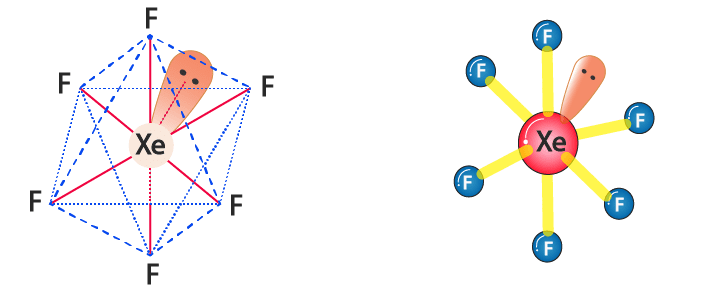
| Name of molecule | Xenon hexafluoride |
| Hybradization type | sp3 d3 |
| Geometry | Distorted Octahedral |
| Bond pair | 6 |
| Lone pair | 1 |
Hybradization of XeOF2 :
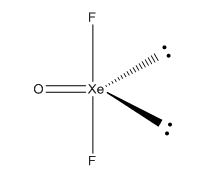
| Name of molecule | Xenon Oxydifluoride |
| Hybradization type | sp3 d |
| Geometry | Trigonal Bipyramidal |
| Shape | Triangular |
| Bond pair | 3 |
| Lone pair | 2 |
Hybradization of XeO2F2 :
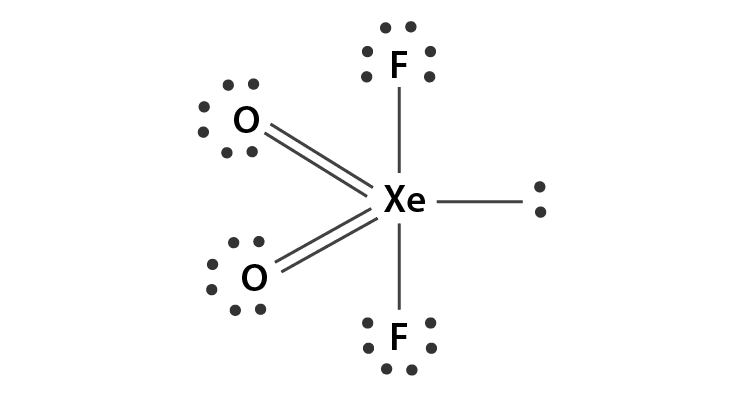
| Name of molecule | Xenon dioxide difluoride |
| Hybradization type | sp3 d |
| Geometry | Trigonal Bipyramidal |
| Shape | See-saw |
| Bond pair | 4 |
| Lone pair | 1 |
Electronic configuration of noble gases:
Electronic configuration of Helium:
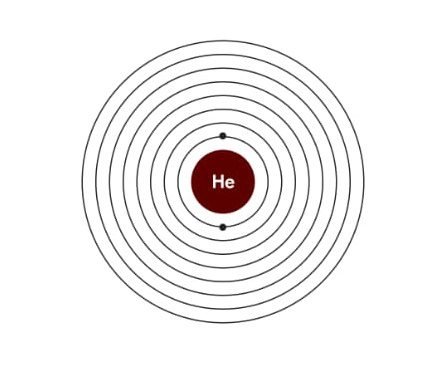
| Electrons | 2 |
| Protons | 2 |
| Netrons | 2 |
Electronic configuration of Neon:
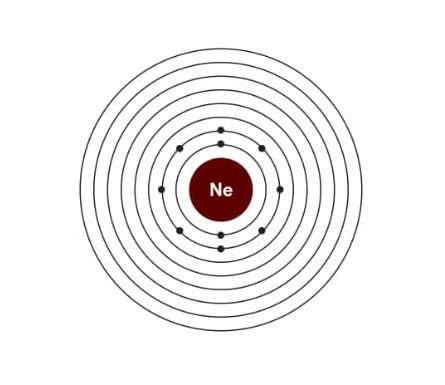
| Electrons | 10 |
| Protons | 10 |
| Neutrons | 10 |
Electronic configuration of Argon:
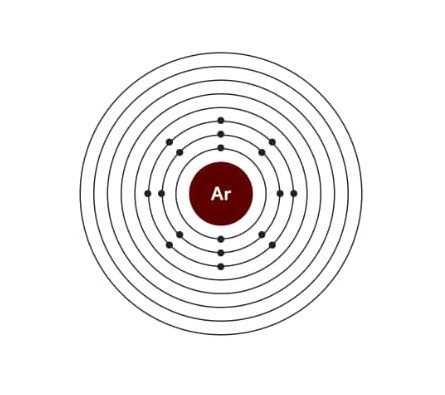
| Electrons | 18 |
| Protons | 18 |
| Neutons | 22 |
Electronic configuration of Krypton:

| Electrons | 36 |
| Protons | 36 |
| Neutrons | 48 |
Electronic configuration of Xenon:

| Electrons | 54 |
| Protons | 54 |
| Neutons | 77 |
Electronic configuration of Radon:
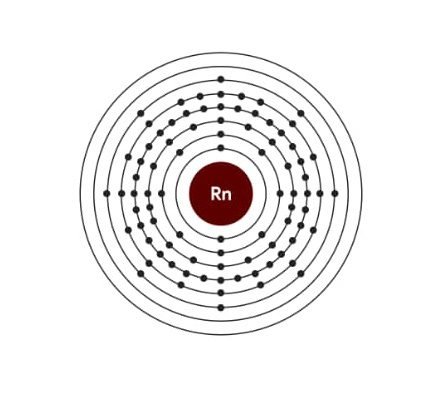
| Electrons | 86 |
| Protons | 86 |
| Neutrons | 136 |



Leave a Reply