Is it NaOH acid or base? Before we answer the question, we explain the concept of acid and base. According to Arrhenius’s concept, acids are those which give only H+ ions in an aqueous medium and Base gives only OH- ions in an aqueous medium. We have a NaHCO3, when it dissociates with its ions, it gives Na+, H+, and CO32-.
NaHCO3 → Na+ + H+ + CO32-
What do you think about NaHCO3, is it an Arrhenius acid or not? It gives Na+ and H+, so it is not an Arrhenius acid because, according to Arrhenius, acids are those that only give H+ ions in an aqueous solution. But when NaHCO3 is ionized, it gives H+ and Na,+ bo, so it is a salt. Similarly, When Cu(OH)Cl is ionized,
Cu(OH)Cl → Cu+ + OH– + Cl–
Cu(OH)Cl is not an Arrhenius base because it gives both OH- and Cl- but according to Arrhenius, base are those which gives only OH- ions in an aqueous solution.
Is it NaOH acid or base?
NaOH is an Arrhenius base that gives OH- ions in an aqueous solution.
NaOH → Na+ + OH–
Sodium hydroxide is an inorganic compound also known as lye and caustic soda with the chemical formula NaOH. It is the white solid ionic compound having sodium cations and hydroxide anions. The question is here,
Why Sodium Hydroxide Is Also Called Caustic Soda?
Caustic soda is the common name for NaOH and this name is given due corrosive nature on plants and animal tissues.
Properties of Sodium Hydroxide
- Sodium hydroxide is a white ionic solid.
- Molar mass: 39.997 g/mol
- Density: 2.14 g/cm3
- Melting point: 318 °C
- Boiling point: 1388 °C
- Soluble in: Water, Ethanol, Methanol
- It has a soapy feel to touch and is bitter to taste.
- It is strongly alkaline.
- Chemical formula: NaOH
- IUPAC ID: Sodium hydroxide, Sodium oxidanide
Preparation of Sodium Hydroxide
Sodium hydroxide can be prepared by different methods. Here we study in detail the mechanism of Castner-Kellner, Nelson Diaphragm cell, and Loewig’s process.
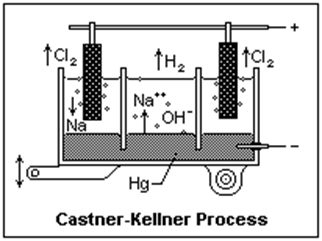
1. Castner-Kellner Process
Principle
To obtain sodium hydroxide, electrolysis of brine solution is performed.
Castner-Kellner cell
It is a rectangular steel tank. The hard rubber contains 25% to 80% sulfur, and linseed oil is known as ebonite. Ebonite is lined inside the rectangular tank. A layer of mercury at the bottom of the rectangular tank acts as a cathode, and titanium acts as an anode.
The brine solution is ionized according to the following reaction:
2NaCl → 2Na+ + 2Cl–
When electricity is passing through the brine solution, the positive cation Na+ moves toward the cathode, and the negative anion OH- moves toward the anode. Sodium ions get deposited at the Hg cathode forming a sodium amalgam that reacts with water where NaOH, H2, and Hg are produced. Chloride ions move toward the anode and exist in the cell from the top.
2. Nelson Diaphragm Cell
The anode (positive electrode) is titanium, carbon (graphite) coated with Ru-Ti oxide. The anode and cathode compartments are separated by the porous diaphragm of asbestos or metal oxide with polymer. The porous diaphragm prevents chloride ions from entering the cathode compartment and prevents hydroxide ions from entering the anode compartment. The saturated brine solution enters the anodic compartment where the chlorine gas is produced.
Anode reaction (oxidation):
2Cl–(aq) → Cl2 (g) + 2e–
Cathode reaction (reduction):
2H2O (l) + 2e– → H2 (g) + 2OH–(aq)
Sodium-ion Na+ moves across the diaphragm to the cathode compartment, combining with hydroxide ion OH– to form NaOH.
Overall cell reaction:
2H2O (l) + 2Cl–(aq) + 2Na+(aq) → 2Na+(aq) + 2OH–(aq) + H2 (g) + Cl2 (g)
The product contains sodium hydroxide, NaOH, and sodium chloride, NaCl. Sodium hydroxide can be crystallized out.

3. Loewig’s Process
Loewig’s Process for sodium hydroxide preparation depends on the production of sodium ferrate, Na2Fe2O4 which is then decomposed with H2O.
How do we prepare Sodium Ferrate?
The soda liquors are combined with ferric oxide, and the mass is evaporated to dryness, and heated at a bright red heat, usually in a rotating furnace. By heating at a high temperature, a reaction between the iron oxide and the sodium carbonate is occur, sodium ferrate is formed and CO2 escapes.
Until all the soluble matter is eluted, the mass is washed with cold water. After the elution of all soluble matter, water at 90 °C is passed over the sodium ferrate and it is decomposed. Sodium hydroxide is formed and iron oxide is regenerated that is returned to the calcining process. The ferric oxide must be very clean and free from silica or other impurities and it is used in natural iron ore.
What are the Uses of Sodium Hydroxide?
- Sodium hydroxide is used as a cleansing agent.
- It is used in the manufacturing of sodium carbonate, also known as washing soda.
- It is used as a reagent in laboratories.
- It is also used in the manufacturing of soda lime.
- Caustic soda is used in the extraction of aluminum.
- 16% used in pulp and paper.
- 17% used in alumina.
- 17% used in organic pharma polycarbon.
- 10% used in inorganics: sodium silicate, STPP
- 7% used in soap and detergents.
- 9.5% are used in textiles.
- It is also used in water treatment, food, and other industries.
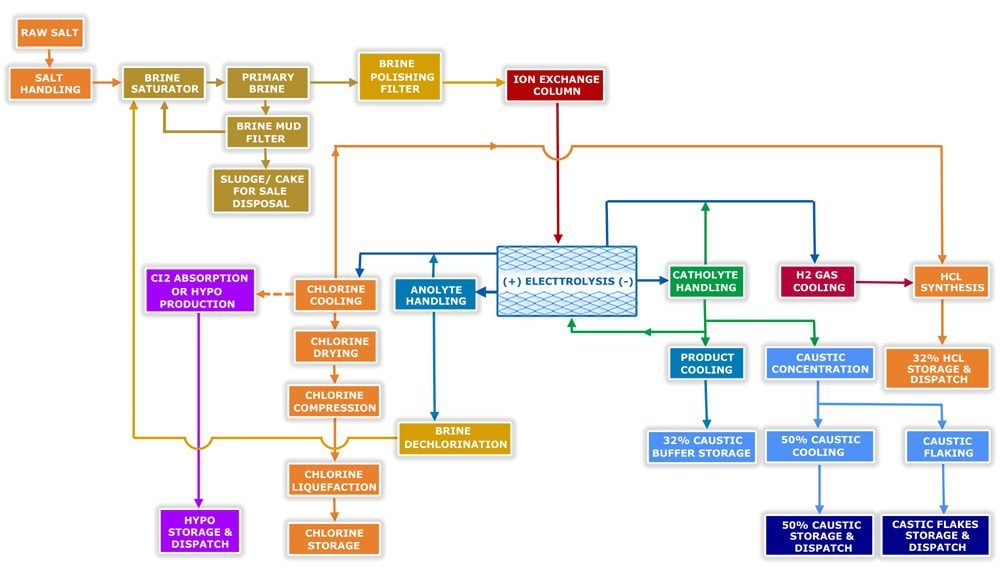
Caustic Soda Plant Process Flow Diagram