
Titanium is a periodic element with the chemical symbol “Ti”. In the periodic table, titanium is present in the fourth horizontal row or period and in the fourth group. It is grouped as a transition element with numbers of other elements. Periodic element Titanium is the fourth most common metal on the earth after aluminum, iron, and magnesium. It is found in rocks, soil, and deposits deep under the sea.
Titanium atom:
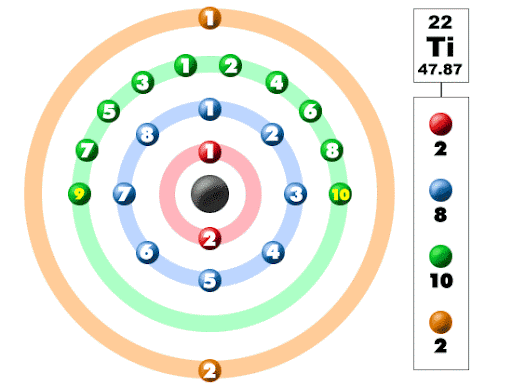
Titanium is present in 4th group of the periodic table and it is categorized as transition element. Titanium atom consists of 22 positively charged protons in the nucleus. And we can say that titanium has an atomic number of 22.The number of neutrons in the titanium is 26. As we know that total sum of neutrons and protons is called atomic mass. So the atomic mass of titanium is 48.Titanium has 22 electrons revolves around the nucleus in 4 shells. There are 2 electrons in the first shell. 8 electrons in the second shell. 10 electrons in the third shell. 2 electrons in the outermost shell. Titanium has a very unusual arrangement of electrons comparable to other elements.
Discovery:
People think that titanium is a modern and high-tech material but it is true. Titanium has been widely used since the 1950s.
William Gregor:
It was first discovered 200 years ago almost in 1791, English clergyman and chemist William Gregor (1761-1817) found a sample of black sand at the beach in Menaccan in Cornwall England.
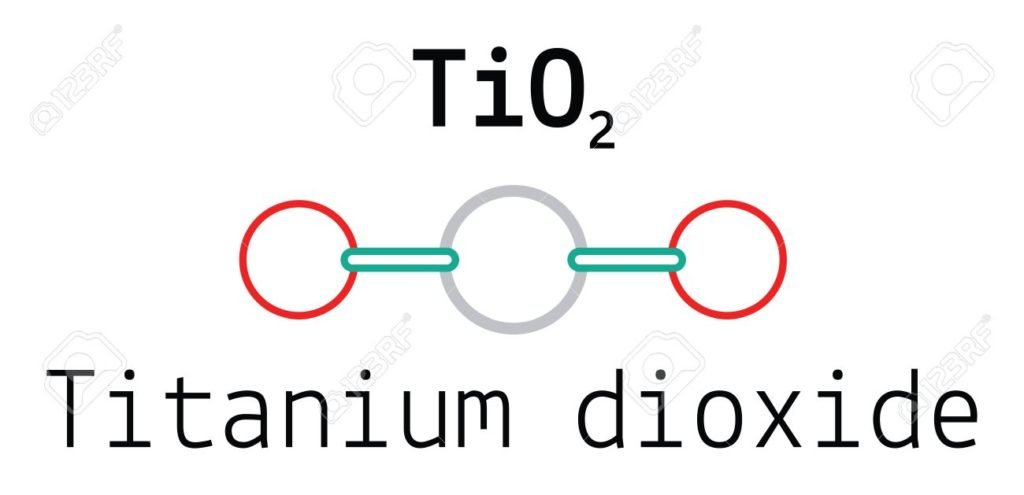
After analyzing the sample Gregor found a reddish-brown compound known as titanium dioxide. Gregor called this new substance menacchanine after the name of the town where it was found.
Martin Heinrich Klaproth:
After five years of Gregor’s analysis. A German Chemist Martin Heinrich Klaproth (1743-1817) located same substance in rocks. He observed that the substance was metal oxide. And he named this substance Titanium.
Method for Isolation of titanium :
Due to the vast applications of titanium in different fields. Its demand is increasing day by day so it is important to extract more titanium. It is necessary to extract more titanium from its ores.
In 1910, U.S Chemist Matthew Hunter isolated titanium in the laboratory by reacting titanium tetrachloride ( TiCl4 ) with sodium.
In 1937, German Chemist William A. Kroll extended or modified Hunter’s technique. Kroll’s method is now the main commercial method for the isolation of titanium from its ores.
The Kroll Process:
In the Kroll process firstly titanium dioxide ( TiO2 ) reacts with chlorine (Cl2 ) . A light yellow liquid material titanium tetrachloride is produced. Then this liquid material is passed into large stainless steel or carbon vessel containing molten magnesium metal.
This process is used to form pure titanium metal. The titanium tetrachloride consist of one atom of titanium joined to four atoms of chlorine.
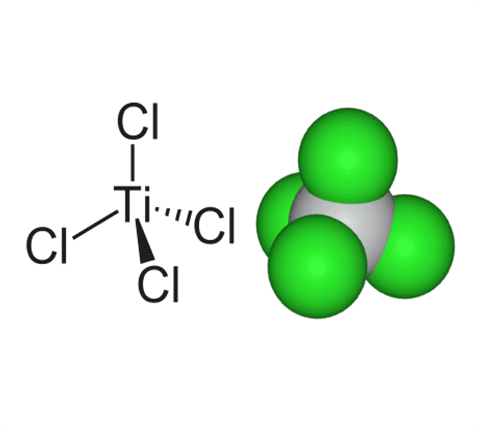
In the first step of the Kroll process, titanium tetrachloride and magnesium are heated to 1800°F (1000°C). High temperatures break up the bond between titanium and chlorine atoms to leave behind pure titanium metal.
Then four chlorine atoms are free to form new bonds. The chlorine atoms react with magnesium atoms to make magnesium chloride. Two chlorine atoms join with one magnesium to produce magnesium chloride. After this titanium atom was left on its own.
Reaction:
The following reaction takes place during the Kroll process.
2Mg + TiCl4 → Ti +2MgCl2
Occurrence:
The black particles found in beachy sand contain titanium. Metal titanium is found in rocks, soil, and deposits deep undersea. Titanium is the fourth most abundant metal found on earth after aluminum, iron, and magnesium.
Titanium found in the ground:
Naturally titanium is not found in purely. It occurs in the form of ores. The two most common ores of titanium are rutile and ilmenite. Pure titanium has a silvery gray color due to titanium dioxide formed as a thin layer on the surface when metal reacts with oxygen.
Rutile:
Rutile is the black particles present in the sand. Rutile consists of a compound called TiO2. Along with traces of other elements are also present.
Ilmenite:
It is made up of compounds consisting of iron, titanium, and oxygen (FeTiO3 ). The name ilmenite is taken from the Ilmen Range of the Ural Mountains in Russia. Where ilmenite is found in abundant quantity.

Many of the countries produce pure forms of titanium from rutile and ilmenite. Including Australia, Brazil, Canada, Norway, and the United States.
When rutile or ilmenite is heated with carbon in the stream of chlorine at 900 °C gives volatile tetrachloride TiCl4 having a boiling point of 136 °C. That is purified by fractional distillation. And reduced by magnesium at 800 °C in an atmosphere of argon. The magnesium is sublimed away and silvery-white metal is fused under argon and helium and cast into ingots.
Atomic Properties of Titanium:
The atomic properties of titanium are as follow:
| Atomic number | 22 |
| Period | 4th period |
| Group | 4th group |
| Electronic configuration | 1s2 , 2s2, 2p6, 3s2, 3p6, 3d2, 4s2 |
| Standard atomic weight | 47.867 |
| Electrons per shell | 2, 8, 10, 2 |
| Block | d-block |
| Ionization energies First I.E Second I.E Third I.E | 658.8 kJ / mol 1309.8 kJ / mol 2652.5 kJ / mol |
| Atomic Radius | 147 pm |
| Electronegativity values | Pauling scale:1.54 |
| Oxidation states of Ti | -2,-1, +1, +2, +3, +4 |
Physical properties of Titanium:
As we know that titanium is the most abundant metal found on earth surface. The physical properties of titanium are discussed below.
Melting point : 1941 k
Boiling point : 3560 k
Heat of Fusion: 14.15 Kj/mol
Heat of Vaporization : 425 Kj / mol
Density : 4.11 g/cm3
Molar heat capacity : 25.060 J(mol.k)
Magnetic ordering: paramagnetic order
Thermal Expansion : 8.6 μm / (m.k) at 25 ℃.
Thermal conductivity : 21.9 w / (m. k)
Poisson Ratio : 0.32
CAS Number : 7440-32-6
Special characteristics of Titanium metal:
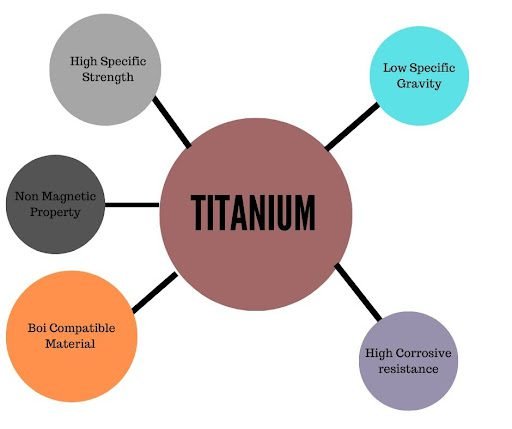
Titanium has many of attractive properties or characteristics some are as:
It is resistant to corrosion. Titanium has high strength to density ratio. Titanium has good fracture resistance. Titanium is a strong metal but exhibits low density. Titanium has two allotropic forms that is high-temperature beta phase and other is low temperature alpha phase. Titanium react easily with oxygen. In pure form this metal is soft and weak but it is strong when mixed with other metals to form alloys. The high melting point of titanium makes it a ideal metal for the construction of high speed aircraft and space vehicles.
Isotopes of Titanium:
Naturally occurring titanium has total five stable isotopes. These isotopes are discussed following with their uses in different fields.
Titanium 46 ( Ti 46):
Titanium 46 isotope of titanium metal is ideally suited to environment for used in high neutron fluxes for example : fission reactors.
Titanium 47 ( Ti 47):
Titanium 47 the isotope of titanium is used as a precursor for the production of V-48.
Titanium 48 (Ti 48):
This isotope of titanium is actively used for the formation of radioactive isotope V-48 .
Titanium 49 (Ti 49):
This isotope of titanium is used in the production of radioisotope V-49.
Titanium 50 (Ti 50):
Titanium 50 is used for formation of heavy elements and also used for bombardment of elements such as Lead, Bismuth, Berkelium . These elements are bombarded with Ti 50.
Chemical Reactions of Titanium:
Following are some of the chemical reactions of Titanium:
Reaction of Titanium with air:
Coated titanium metal has an oxide layer that makes it inactive. When titanium is burned in air it burns with white flame and form titanium dioxide, TiO2 and titanium nitride. Titanium also burns in pure nitrogen to give titanium nitride.
Ti (s) + O2 → TiO2 (s)
2Ti (s) + N2 → TiN (s)
Reaction with Water:
The coated titanium metal has an oxide layer makes it inactive. Titanium react with steam and give dioxides, titanium (iv) oxide , TiO2 , and hydrogen.
Ti (s) + 2H2O(g) → TiO2 (s) +2H2(g)
Reaction of titanium with Halogens:
Titanium reacts with halogens to form titanium (IV) halides. Titanium reacts with fluorine at 200 ℃ to form TiF. Also reacts with chlorine, Bromine and iodine to give Titanium (iv) bromide ,titanium (iv) chloride and titanium (iv) iodide respectively.
The reactions of titanium with above mentioned halogens are as follow.
Ti (s) + 2F2(g) → TiF4(s)
The TiF4(s) formed is in white color.
Ti (s) + 2Cl2(g) → TiCl4 (l)
The TiCl4 (l) formed is colorless.
Ti (s) + 2Br2(g) → TiBr4 (s)
The TiBr4 (s) formed is in orange color.
Ti (s) + 2I2(g) → TiI4 (s)
The TiI4 (s) formed is in dark brown color.
Reaction of Titanium with Acids:
Titanium does not react with mineral acids and hot hydrochloric acid. Titanium react with aqueous hydrofluoric acid, HF and form complex anions [TiF6 ]3- and also form hydrogen H2 .
2Ti (s) +12HF(aq) → 2 [TiF6 ]3- (aq)+3H2(g) +6H+ (aq)
Reaction with Bases:
Titanium don’t react with bases under normal conditions.
Applications of Titanium:
Titanium is alloyed with other elements such as Al, Vanadium, iron, etc. This results in a strong low density alloys used in jet engines, missiles, spacecrafts . Also used in military ,industrial applications such as chemicals of various kind and petrochemicals, desalination plants, pulp and papers. Also used in agricultural food ,medical and dental uses jewelry, cellular phones, sporting goods and auto-motives . All in all it is used many industries and it has vast applications . Some applications are discussed here below.
Auto-motive use :
Auto-motive and motorbike use of titanium in racing applications provide the decreased weight and high electricity with tension provide racer’s aspects. Also decreased weight in auto-motive family enhance the fuel economy. Also the metal is too expensive to use in auto-motive applications but some niche uses has been occurred now.
Sporting Goods:
Titanium has another consumer application of sporting goods. Titanium is used in manufacturing of many sporting goods such as lacrosse stick shafts, cricket ,hockey, lacrosse ,football helmet grills ,bicycle frames and components .We see that titanium bikes are used in racing events.
Architecture use:
Titanium is extensively used in architecture. The architectural use of titanium is based on coloring and inertness of titanium. Some of the worlds beautiful or historical places framed with titanium are mentioned here as; The 42.5 m monument of Yuri Gagarin , who was the first person traveled in space, His statue is made up of titanium and it is located in Moscow Russia.

Some other examples of buildings are Cosmonaut Museum in Moscow also made of titanium. The sheathed titanium building in Europe and North America are the Guggenheim Museum in Bilbao, Cerritos Millennium Library respectively. Titanium is also used in roof tiles.
Firearms and Laptop Computers:
Titanium has high strength to low density as compared to other metals that makes it use in firearm productions. Such as rifles and pistols . This ability of titanium makes it used in body of laptop computers. For example in Apple’s power Book line .
Kitchenware and tools:
Titanium is increasingly use in low density, high end and corrosion resistant tools. For examples flashlights ,knife handles, hammers, shovels etc. Bolts are made up of titanium and titanium alloys. High end kitchenware is also made of titanium.
Backpacking:
Titanium appliances like lanterns, cooking utensils, tent stake has been extensively used by backpackers. As compared to other metals appliances titanium components are lighter weight so they are preferable.
Jewelry including eyeglasses and watches:
Durability of titanium makes it extensively used for designer’s jewelry .Inertness of titanium makes it to be good choice for those with allergies and to be used in environments like swimming pools. Ti-Au alloy has approximately hardness of 14 carat gold with durability greater than pure 24 carat gold.

Due the durability ,low density , corrosion resistant abilities of titanium makes it increasingly used in watches.
Other uses:
- Titanium is also used in making pictures, decorative objects ,sculptures and furniture also.
- Titanium plated to change the thickness of oxide layer and give a bright color . Due to this coloring property or inertness it is used in body piercing.
- Titanium is also used in making some noncirculating coins and medals.
Conclusion:
Titanium belongs to 4th group of the periodic table . It is the fourth most abundant metal found on earth. Due to its inertness , low density, high strength it has vast applications .Titanium don’t react with mineral acids and has no effect of alkalis. It does not found pure in nature. Four isotopes above mentioned are stable isotopes.



Leave a Reply