
Helium is the chemical element with the symbol He. Helium has atomic number 2, atomic mass 4 and, an electronic configuration is 1s2. It is colorless, tasteless, odorless, inert, non-toxic, monoatomic gas, the first in the noble gas group in the periodic table.
The boiling point of helium is the lowest among all the elements. Helium is the second lightest and second most abundant element in the universe. (Hydrogen is the lightest and most abundant element)
Helium is the first member of noble gases having 2 electrons in 1s orbit. Due to the least number of electrons in its orbit, it has a more effective nuclear charge, more attractive forces between nucleus and electrons, no shielding effect, the radius of its atom is shortest as compare to other members of its group.
Its abundance is similar to this in both suns and in Jupiter. This is due to the very high nuclear binding energy per nucleon of He-4, with respect to the next three elements after helium. Helium-4 is the most helium in the universe, the vast majority of which was formed during the big bang. The large amounts of new helium are being formed by the nuclear fusion of hydrogen in stars.
Discovery of Helium
Helium was discovered by Frankland and Lockyer in 1868 in a new bright-line, D3 in the yellow region of the son’s atmosphere. Lockyer and Frankland observed a new yellow light in the chromosphere of the sun, during a total solar eclipse. This line did not identical with D1 and D2 lines of sodium and Lockyer called it the D3 line. This shows the presence of a new element. It was named helium. The word helium is originated from the Greek letter “Helios” meaning the sun.
In the earth, it was discovered by Ramsay. William Ramsay’s work led to groundbreaking advances in thermodynamics and nuclear physics. While searching for Argon, Ramsay found helium, which had been previously thought to exist only in the sun. In 1898, Ramsay isolate Helium from Uranium ore. He was convinced that there were other unreactive gases and they were all members of one group in the periodic table. Sir William Ramsay was a Scottish chemist who discovered the noble gases (Argon, Neon, Krypton, Xenon) and received the Noble Prize in Chemistry in 1904
Physical Properties
- Helium is a colorless gas
- It is tasteless, odorless, toxic, monoatomic, and inert gas.
- Atomic Number: 2
- Atomic Mass: 4.003
- Density: 0.0001785
- Melting Point: -272℃, -458℉, 0.95K
- Boiling Point: -269℃, -452.07℉, 4.216K
- Charges: A helium nucleus has a charge +2.
- Atomic Radius: 31
- Covalent Radius: 32
- Ionization Potential: 24.59
- Electronegativity: —
- Electron Affinity: 0 KJ/mol
- Oxidation States: 0
Prevalence
| Universe | 23.00% |
| Sun | 23.00% |
| Oceans | 7.2×10-10% |
| Earth Crust | 5.5×10-7% |
| Human | N/A |
| Meteorites | N/A |
How Helium is made
On earth, helium officially called helium-4 comes from radioactive decay. When a radioactive element like uranium or thorium breaks down over time or decays it occasionally loses two protons and two neutrons. This is called alpha decay.
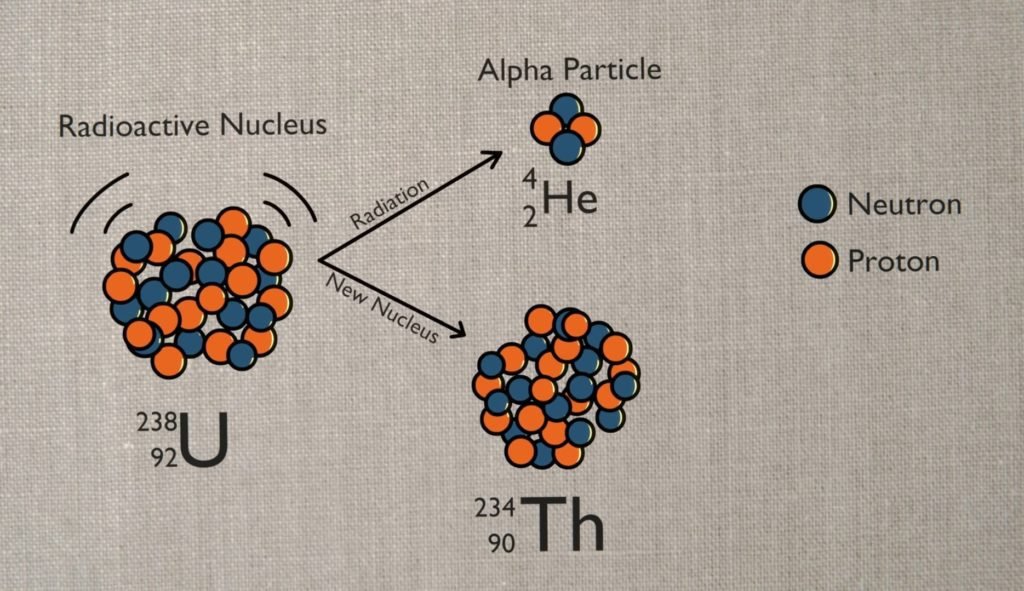
If that alpha particle grabs a couple of electrons it forms helium-4. Helium is usually produced as a by-product of natural gas processing. Natural gas contains methane and other hydrocarbons which are the principal sources of heat energy when natural gas is burned. Most natural gas deposits also contain smaller quantities of nitrogen, water vapors, carbon dioxide, helium, and other non-combustible materials. When the gas contains more than about 0.4% helium by volume, a cryogenic distillation method is often used in order to recover the helium content.

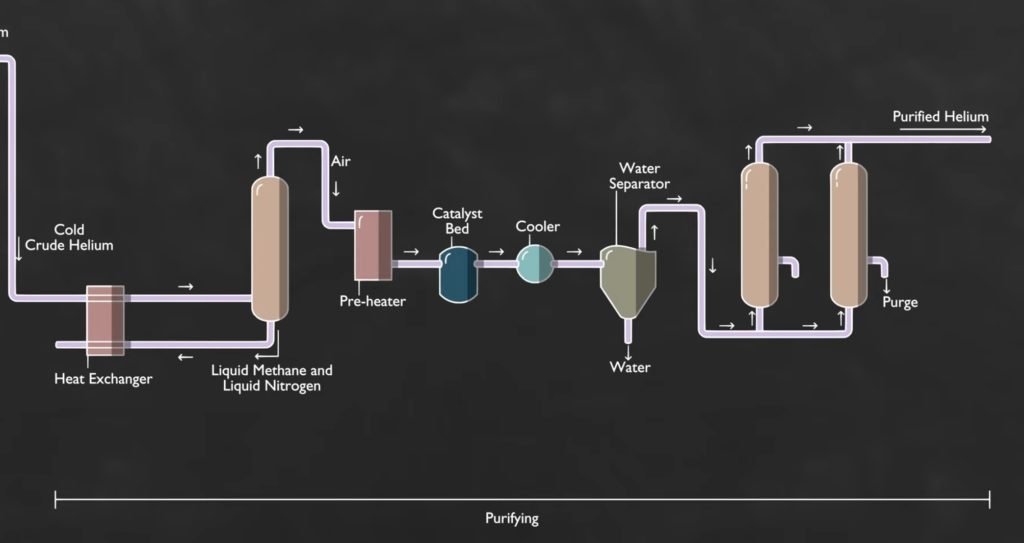
Now we discuss three main parts and ten steps of making helium:
Part 1: Pre-treating
This method utilizes an extremely cold cryogenic section as a part of the process. All these impurities that might solidify such as water vapors, carbon dioxide, and certain heavy hydrocarbons must first be removed from the natural gas.
Step 1
In the pre-treating process, the first step is removing carbon dioxide. The crude natural gas is pressurized to about 800 psi. It then flows into a scrubber where it is subjected to a spray of monoethylamine which absorbs the carbon dioxide.
Step 2
Step two in the pre-treating process is removing water molecules. The gas stream passes through a molecular sieve which strips the larger water vapor molecules from the stream while letting the smaller gas molecules pass. The water is backflushed out of the seal and removed.
Step 3
Step 3 is removing heavy hydrocarbons. Any heavy hydrocarbons in the gas stream are collected on the surface of a bed of activated carbon. As the gas passes through it, the gas stream now contains mostly methane and nitrogen with smaller amounts of helium, hydrogen and, neon.
Part 2: Separating
Natural gas is separated into its major components through a distillation process known as fractional distillation. In this process, the nitrogen and methane are separated in two stages leaving a mixture of gas containing a high percentage of helium. The gas stream passes through one side of a plate-fin heat exchanger while very cold methane and nitrogen from the cryogenic section pass through the other side. The incoming gas stream is cooled while the methane and nitrogen are warmed.
Step 4
The gas stream then passes through an expansion value which allows the gas to expand rapidly while the pressure drops to about 145 to 360 psi, this rapid expansion cools the gas stream to the point where the methane starts to liquefy.
Step 5
The gas stream is now part liquid and part gas enters the base of the high-pressure fractionating column as the gas works its way up through the internal baffles in the column it loses additional heat. The methane is continuous to liquefy forming a methane-rich mixture in the bottom of the column where most of the nitrogen and other gases flow to the top.
Step 6
The liquid methane mixture called crude methane is drawn out of the bottom of the high-pressure column and it is cooled further in the crude sub-cooler then it passes through a second expansion wall which drops the pressure to about 22 psi. Before it enters the low-pressure fractionating column, as the liquid methane works, it’s way down to the column. Most of the nitrogen is separated leaving a liquid that is no more than 4% nitrogen and the remaining methane. This liquid is pumped off warmed and evaporated to become upgraded natural gas.
Step 7
Meanwhile, the gases from the top of the high-pressure column are cooled in a condenser. Much of the nitrogen condenses into a vapor and is fed into the top of the low-pressure column. The remaining gas is called crude helium. It contains approximately 50 to 70% helium, 1 to 3% unliquefied methane, smaller quantities of hydrogen, and neon and, the remaining nitrogen.
Part 3: Purifying
Crude helium must be further purified to remove most of the other materials. This is usually a multi-stage process involving several different separation methods.
Step 8
The crude helium is first cooled to about -193℃. At this temperature, most of the nitrogen and methane condenses into a liquid and are drained off the remaining gas mixture is now 90% pure helium.
Step 9
Air is added to the gas mixture to provide oxygen. The gas is warmed in a pre-heater and then it passes over a catalyst which causes most of the hydrogen in the mixture to react with the oxygen in the air and forms water vapor. The gas is then cooled and the water vapor condenses and is drained off.
Step 10
The gas mixture enters a pressure swing absorption unit consisting of several absorption vessels operating in parallel. Within each vessel there are thousands of particles filled with tiny pores as the gas mixture passes through these particles under certain gases are trapped within the pores of the particle, the pressure is then decreased and the flow of the gas is reversed towards the trapped gases. This method removes most of the remaining water vapors, nitrogen, and methane from the gas mixture. The helium is now about 99.99 pure.
How the Sun Shines: The Nuclear Reactions that Power the Sun
The sun was always a bit of a mystery. No one could figure out what was powering it. In the 19th century, some scientists thought it could all be explained by gravity. The sun began as a faint cloud of dust and gas. And this cloud collapsed under the force of gravity releasing a lot of heat. This might explain why the sun is hot, but it doesn’t explain why the sun has been hot for so long.
A clear picture didn’t emerge until we had a better model of the atom. At the center of each atom is a tiny nucleus made of protons and neutrons. Protons have a positive electric charge and this is a problem because their charges are pushing them apart. The electric force is trying to rip apart the nucleus. But there is a strong force holding it together: the nuclear force.
The nuclear force brings protons and neutrons together. It doesn’t really affect pairs of protons or pairs of neutrons. To hold everything together you need to have just the right combination of protons and neutrons. Too many protons and it fall apart. Too many neutrons and falls apart. But when you get just the right combination, you produce a nuclear reaction.
Nuclear reactions are similar to chemical reactions in many ways. There are certain combinations of chemicals that are more stable. When you form stable compounds like CO2, H2O, salt NaCl, etc. you make energy. For example, if you combine hydrogen and oxygen to make H2O, this is a more stable combination and you make energy.
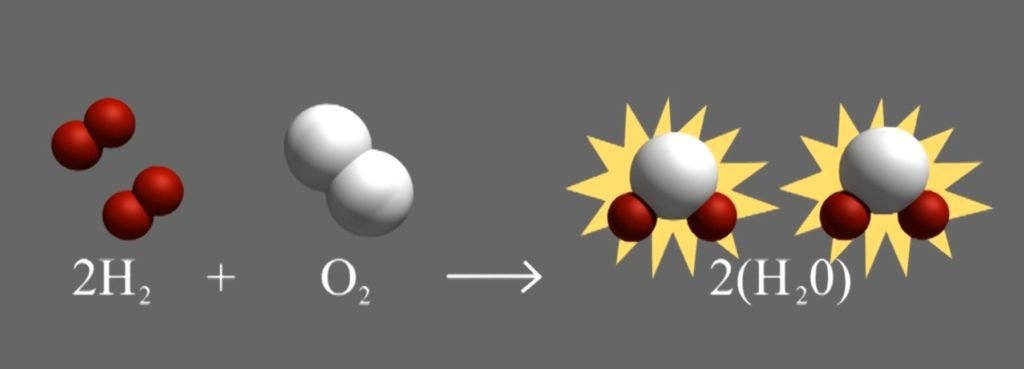
Similarly, if you combine two hydrogen nuclei into helium, this is a more stable combination and you make energy.
Nuclear Reactions
Nuclear reactions are different from chemical reactions. They’re much harder to produce, they’re much more powerful, and they involve neutrons. Chemical reactions are part of our everyday life, but nuclear reactions are not. The reason is that nuclear force only works over very short distances.
The protons are all positively charged. So if you bring hydrogen nuclei together, they just repel each other. If you bring them more closely, they repel each other more. The nuclear force only kicks in when they are extremely close. Normally, the nuclei never get near each other. It is only under conditions of extreme pressure and extreme heat that the nuclei will collide. And this only happens at the center of a very massive object like our sun or inside a nuclear bomb.
Neutrons are very important in nuclear reactions, but in chemical reactions they’re irrelevant. If you have protium (10H) and deuterium (12H). For this purpose of chemistry, we don’t care about the neutrons. But for nuclear reactions, we do.

In the above figure, there is a hydrogen-1 nucleus (protium) and a hydrogen-2 nucleus (deuterium). The number at the end is the total number of protons and neutrons.
- Two protons and a neutrons make Helium-3
- Two protons and two neutrons make Helium-4
Nuclear Power
Nuclear reactions are very powerful. Let’s look at two reactions in given:
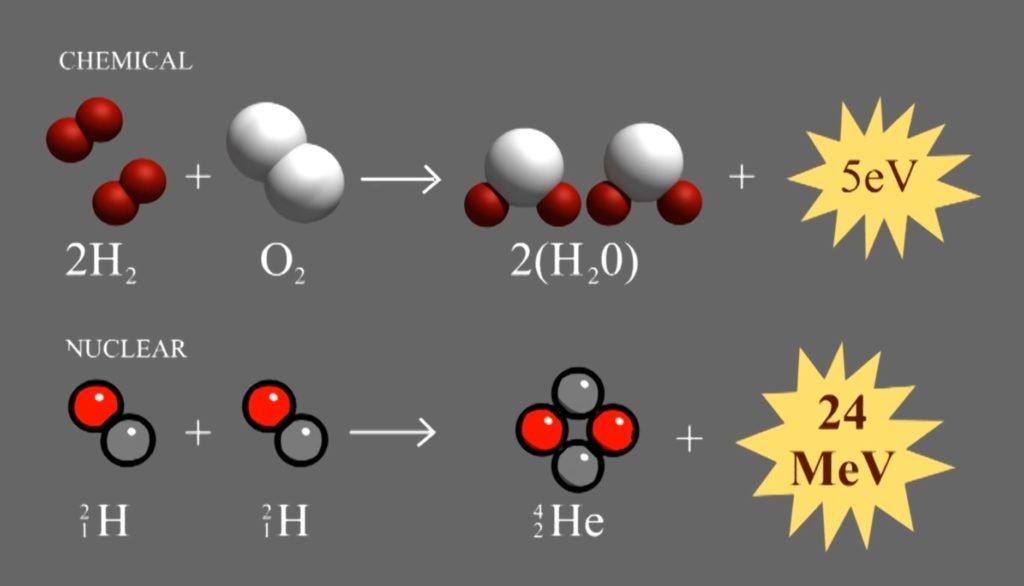
The chemical reactions release 5 eV of energy. The nuclear reaction release 24 million eV of energy. So, the nuclear reaction is 5 million times more powerful. There’s enough energy in just a pound of deuterium to power 10,000 homes for a year. This is an insane amount of energy. It’s why the sun is so bright, why nuclear bombs are so scary, and why scientists have been working on fusion power for decades with little success.
Nuclear Forces
We’ve been talking about the strong nuclear force, but there’s also a weak nuclear force. The strong force is what holds the nucleus together. The weak force causes radioactivity. And it’s called the weak force because it acts slowly.
The weak force can cause protons and neutrons to switch identities. A proton can turn into a neutron or a neutron can turn into a proton. This is called beta decay.
Neutrons are heavier than protons just barely. But what this means is that a free neutron can lose some mass and turn into a proton. But a proton cannot turn into a neutron unless it’s near other particles. The reason this process is called beta decay is that it’s one of the ways that a radioactive element can decay.
Radioactivity-A Slow Process
Now it turns out that radioactivity can be slow. Imagine you have a box full of Uranium-235 atoms. Eventually, the Uranium atoms will all turn into Lead, but they’re not in any hurry to do this. If you want to wait until half your Uranium atoms have to turn into Lead, you’ll be waiting 700 million years. Radioactivity involves more than just beta decay. But the point is that beta decay can be a slow process and it’s a random process.
Now we finally have covered enough background to explain how the sun shines.
How the Sun Shines
This was worked out in the 30s by Hans Bethe. The sun is mostly made of Hydrogen-1 which is just a proton. The sun is very hot and very dense so these protons collide. But this doesn’t cause a nuclear reaction because there are no neutrons.
Most of the time when two protons collide they immediately fly apart producing no energy. But it is possible, very very rarely, that sometimes else happens: beta decay. At the moment the two protons collide, one of them undergoes beta decay and turns into a neutron. This gives us hydrogen-2 and release tons of energy. The Hydrogen-2 nucleus continues moving around and when it bumps into a proton it turns into Helium-3 and these releases tons of energy. As this process continues, the sun produces more and more Helium-3. When two Helium-3 nuclei collide, they can form into an even better combination, Helium-4, and this releases tons of energy.
Hydrogen is converted into Helium in three steps:
- First, two protons are com,bined into deuterium through beta decay.
- Secondly, a proton is added to make Helium-3.
- Finally, two Helium-3 nuclei collide creating Helium-4 plus two leftover protons.
The first step is by far the slowest because you have to wait for beta decay. For a typical proton in the center of the sun, the first step takes an average of one billion years. Compare that to the second step which takes about 4 seconds. Now in the final step, you have to find another helium nucleus which is rare. This takes about 400 years. These nuclear reactions are the source of the sun’s tremendous power.

Importance of Sun
The sun is the ultimate energy source. The sun heats the air which causes wind. The sun heats water which causes rain giving us hydroelectric power. Sunlight is the foundation of life. The energy from ancient sunlight was deposited into the ground giving us our fossil fuels: coal, oil, and natural gas. Practically, all the world’s energy ultimately comes from either the sun or nuclear power.
Interesting Facts about Helium
- Helium was the first element that was not discovered on earth. It was sun’s spectroscopy data that first revealed that existance of this element.
- Helium is an inert gas. It is non-toxic, completely odorlss, tasteless and, has no color.
- Helium is only about 0.005% in earth atmosphere.
- Helium is the second least reactive noble gas.
- Earth gravity is not strong enough to attract helium.
- Of all elements that exist in this universe, Helium has the lowest boilinh point and lowest melting point. (Melting point: 0.95 K = -272.2℃ and Boiling point: 4.216 K = -268.93℃)
- At normalpressure, when the temperature is droped to absolute zero, Helium will remain in liquid state. It is the only known element to do so.
- When the temperature of helium is dropped a few degrees below its boiling point (which is -452.070℉) (dropped below -456℉, that is slight above zero kalvin), it becomes superfluid. In superfluid state, Helium has zero friction and its density becomes 1/8th of that of water.
- Though hydrogen is lighter than helium, Helium is used for making things float (for example, balloons). The for this is that hydrogen is high combustible and hence, dangerous.
- All other elements become solid on cooling but cooling only produce helium liquid.
- The richest source of helium is south-west USA, where the natural gas contains 0.5-0.8% helium.
- Helium is too light and can escape our planet’s atmosphere. Then, how come it is present in our atmosphere? That’s because it is constantly replenished. There are two sources:
- Cosmic Rays which contain high-energy helium nuclei (about 9% of cosmic rays is made of helium)
- Radioactive decay of element present on earth.
Helium Balloons
What do party balloons, neon signs, and certain light bulbs have in common they are all filled with a noble gas in this.

Helium is often used in balloons as it is lighter than air and unreactive so a good choice for balloons. It has many uses in balloons such as weather balloons, party balloons, blimps and, airships.
Isotopes of Helium
- Helium most common isotopes is Helium-4 which has 2 protons and 2 neutrons in the nucleus and is stable.
- Helium-3 is the other stable isotopes and has 2 protons and 1 neutron in the nucleus. This one is very rare however.
- There is also Helium-5, Helium-6 and Helium-8 which are all rare radioactive isotopes.
Uses of Helium
- Helium (80%) with oxygen (20%) is used for respiration by sea divers as it is insoluble in blood even at high pressure.
- A mixture of helium and oxygen is used for artificial respiration as He has a high diffusion rate so diffusion occurs freely even through a contracyed respiratory tube.
- It is used for creating inert atmosphere for fusion of some metals like Aluminium and titanium which are easily oxidizable.
- It is filled in balloons and in tyres of aeroplanes.
- It is used as a filler in electric transformer.
- Helium nuclei (alpha-paricles) are used as projectiles in nuclear reactions.
- It is used for cooling in nuclera reactors.
- It is also used in welding of metals.
Some Interesting Questions about Helium
How does Helium makes your Voice High?
It is a very interesting fact that why does helium make your great sound funny? Let’s find out.
In order to understand, how helium makes our voice sound so strange. We’ll have to learn the basics about how our voice works? Every person fully starts in a little organ in our necks called a larynx or more commonly our voice box. Your larynx or voice box contains vocal cords which are muscles that stretch across the larynx as rubber bands think of them like guitar strings. Every time when you speak, you’re pushing air from your lungs through your vocal cords. This causes them to vibrate which is what makes the sound we call our voice.
Normal air contains 78% nitrogen, 28% oxygen, and 2% other stuff but helium is a lot lighter than oxygen and nitrogen. When you breathe in a heaping helping of helium that fast air makes your vocal cords vibrate faster than normal and just like guitar strings, if your vocal cords are vibrating faster that means your voice gets higher.
In small amounts, helium is hugely harmless and almost always hilarious. When you breathe in helium from a balloon, you’re not breathing in oxygen which means that your lungs and blood are not getting the fresh oxygen they need so. If you keep breathing only in helium, the lack of oxygen over time could lead to brain injuries or even death.
But despite its dangerous demeanor, Helium is actually a highly important element used in all sorts of high-tech ways scuba gears, MRI machines, nuclear reactors, rockets, and even our smartphones. I guess that’s helium always be the number one element but number two technically.

How does Helium Induced Dipole to Create Intermolecular forces?
Helium is non-polar so it has induced dipole forces. In Helium, electrons of an atom influence each other. When they come closer, they repel each other due to the negative charge of electrons, pushing away from each other. So, the temporary dipole is produced to move the negative charge on one side of the other.
Why is Helium closed of being Ideal?
Electronegativity plays a crucial role in determining the chemical bond. It is the ability to polarize electron density toward itself. In the case of helium, nuclear charge +2 and two electrons in the electronic shell cause the atomic radius smaller. Thus, helium has high ionization energy.
Having the least number of electrons, the intermolecular van der Waal forces are least so, it is closed of being ideal.
According to Pauling’s electronegativity values, they are not mentionable about relative attraction for electrons in the physical properties of the noble gas element because according to the above discussion in the case of helium, intermolecular forces are weaker. They want to come close to each other to form a liquid until the temperature drops to -269℃. These forces are much weaker than other chemical bonding so the interaction of dipole-dipole bonding with each other combines with weaker dipole-induced dipole and dispersion forces, result to condense into a liquid. The molecule that wants to attract each other until the temperature becomes -269℃.
Why Fusion reaction cannot be Used at Commercial level?
Energy is released during the splitting of nuclei. Energy can also be released specific nuclei join together. Helium is the most important example of fusion reaction but the question is here why fusion reactions have not been used commercially. The reason is that it is difficult to control the reaction. Reacting particles in the form of gaseous ions have to be accelerated to high energy for successful collisions. The second reason is the temperature becomes extremely high due to the release of energy. Thirdly, the mixture of particles changes into plasma, magnetic field of sufficient strength and accuracies is needed to control plasma.
Do You Know
Which type of force is present in helium?
Why does helium have weak intermolecular forces?



Leave a Reply