
Chemical Reactions are defined as :The change of one or more chemicals into other chemicals this change is called a chemical reaction.
As we know that matter can combine or break up to produce new things or a new type of matter. and this type of change of matter into the new matter we called this is a chemical reaction.
Chemical reaction:
The chemical reaction is defined as ;
The chemical reaction is a process that is the transformation or change of chemical substances into another.
A chemical reaction is the conversion of reactants(substances that react to form new products)into products(substances that are formed by a chemical reaction).
- Chemical reactions relate to chemical change
- When a chemical change happens there starts a chemical reaction.
- A chemical change is a permanent change in the composition of chemical substances.
Evidence of a chemical reaction to occur:
If the following changes occur we can say that a chemical reaction has occurred;
- change in color
- evolution of gas.
- change in temperature
- change in state.
- these are the conditions or symptoms of a chemical reaction.
What is a chemical equation?
A chemical equation can be defined as ;
A short scientific representation of chemical reaction by using symbols or formulas of given substances participating in a chemical reaction.
A chemical equation is the simplest display of a chemical reaction.
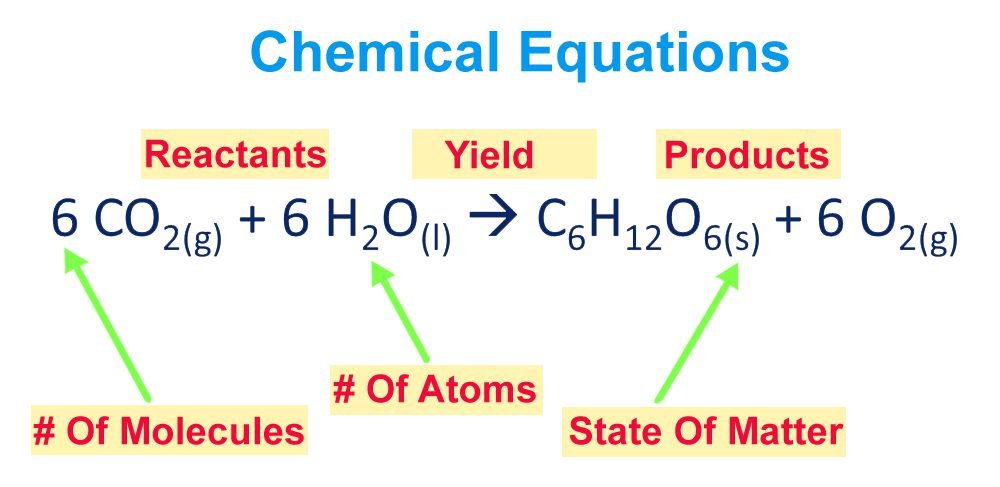
How to write a chemical equation?
The following steps are kept in mind to write a chemical equation:
- The substances that react to form products (reactants) are always written on the left side of the reaction.
- Products are written on the right side of a chemical reaction.
- Arrow sign is used between reactants and products in a chemical equation.
- The coefficients represent that how many molecules are participating in a chemical reaction.
common symbols that we used in writing a chemical equation are;
‘ +’ the plus sign is used between reactants and products to represent that how reactants combine to form products.
‘ → ‘ this arrow sign is used to represent yields of products and separate reactants and products in a chemical equation.
‘s‘ that is used to represent that the substance participating in reaction has a solid-state.
‘I’ is used to indicate that the substance has a physical state of liquid.
‘g‘ represents the gaseous state of a substance participating in a chemical reaction.
‘aq’ is written after a symbol or formula to show that substances are dissolved in water.
‘↓’ if precipitates are formed in a chemical reaction then an arrow showing downward direction is used that is written after the formula.
‘↑’ if products are in a gaseous state then the arrow shoeing upward direction is used and written after the formula of the substance.
Example of a chemical equation:
Magnesium reacts to form magnesium oxide.
2Mg(s) + O2 (g) → 2MgO (s)
Reactants products
limestone decomposed to give calcium oxide and carbon dioxide gas.
CaCO3 (s) → CaO(s) + CO2 ↑(g)
Reactant products
Balanced chemical equation:

According to the law of conversation of mass, a balanced chemical equation is that in which the mass of reactants is equal to the mass of products.
The unbalanced equation is as ;
H2 + O2 → H2O
Balanced equation of the above example is
2 H2 + O2 → 2H2O
Types of chemical reactions:
As we know there are lots of changes that occur daily chemically all the changes that occur chemical and form new products are called chemical changes and there is a lot of chemical reactions here are some categories of reactions that are discussed.
Main types of chemical reactions are
- Combination or synthesis reactions
- Decomposition reactions
- Single replacement(or single displacement) reactions
- Double replacement (or displacement) reactions
- Acid-base neutralization reactions
- Precipitation reactions
- Oxidation-reduction reactions
- Combustion reaction
Combination (synthesis) reactions:
It is a type of chemical reaction when two or more elements or compounds combine to make a single compound is called a combination or synthesis reaction.
General equation:
The general equation for this type of reaction is;
A + B → AB
Example:
4Fe(s) + 3 O2 (g) → 2Fe2 O3 (s)
- Combination reactions also combine to form larger compounds such as;
Fe2 O3 (s) + 3SO3 (s) → Fe2 (SO4 )3 (s)
Decomposition reactions:
It is the opposite of combination reactions. in this type of reaction single substance is transformed or broken down into two or more products.
General equation:
The general equation for decomposition reaction is:
AB → A + B
For example
Sodium bicarbonate when heated;
2NaHCO3(S) → Na2 CO3 (s) + CO2 (g) + H2O(l)
Another reaction for a decomposition reaction is:
2KClO3 (s) → 2KCl(S) + 3 O2 (g)
As we see in the above reaction a single substance is converted to two or more substances hence it’s a decomposition reaction.
Single replacement (or displacement) reactions:
In this type of chemical reaction, a single element replaces another element from a compound. This type of reaction is most frequently given by metals.
- These reactions appear in two forms first one is the cationic exchange and the second is anionic exchange.
- As the name suggests that in cationic exchange cation(+ve ion) it replaces and in anionic the anion(_ve ion) will replace.
The general equation for cationic replacement is:
A + BC → AC + B
The following example represents the cationic replacement reaction.
2Ag(s) +2HCl(aq) → H2 (g) +2AgCl(s)
In the above-given
reaction, the silver replaces the hydrogen with HCl from silver chloride in the
Final product.
General equation for anionic replacement:
D + BC → BD + C
Example:
The following example helps to understand anionic replacement as
3F2(g) + 2FeCl3 (aq) → 2FeF3 (aq) +3Cl2(g)
In the above reaction, fluorine replaces the anion Cl in iron 3 chlorides to give iron 3 fluorides.
Double replacement (displacement ) Reactions:
In this type of chemical reaction two groups exchange or replaces the compounds.
General Equation:
The general equation for double displacement or replacement reaction is as “
AB + CD → AD + CB
There are two types of double displacement or replacement reactions
- Acid-base neutralization reactions
- Precipitation reactions
Precipitation reactions:
First of all what is a precipitate?
An insoluble ionic solid is called a precipitate.
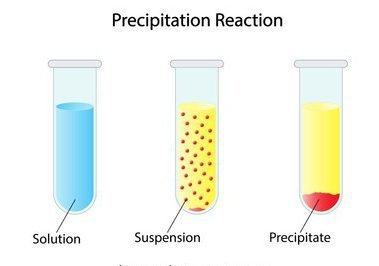
- In these types of chemical reactions negative or positive ions combine in an aqueous solution to form a precipitate this type of reaction is called a precipitation reaction.
- These reactions occurred in an aqueous solution and these reactions are determined by solubility rules. Because all the reactions occurring in aqueous solutions are not forming precipitates.
Example:
The following example helps to understand precipitation reaction.
AgNO3 (aq) + NaCl(aq) → AgCl(s) +NaNO3(aq)
Applications of precipitation reactions:
- These reactions are used in qualitative analysis for example silver nitrate is used to test halides.
- Limewater test used for carbon dioxide is also qualitative analysis.
- These reactions occur in the human body between antibodies and antigens.
Acid-base neutralization Reactions:
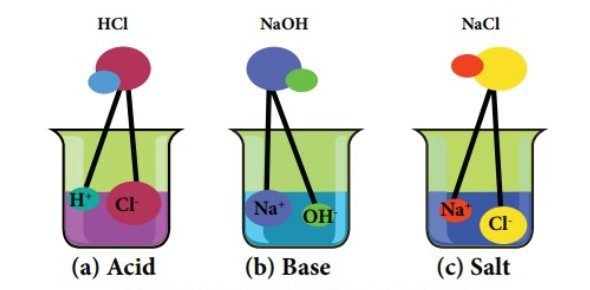
In this type of chemical reaction, a reaction is held between an acid and a base the net products of a neutralization reaction are water and salt.
General equation:
The general expression for this reaction is;
Acid + Base → Salt + water Example:
The following reaction is an example of an acid-base neutralization reaction:
HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l)
Oxidation-Reduction Reactions:
Oxidation-reduction reactions are the types of chemical reactions in which the oxidation state of one or more substances changes. These are also called redox reactions.
What is oxidation?
Oxidation is defined as the addition of oxygen or removal of hydrogen during a chemical reaction is called oxidation.
Oxidizing agent:
The oxidizing agent is a substance that oxidizes itself and reduces the other one.
What is reduction?
Reduction is defined as the removal of oxygen and the addition of hydrogen during a chemical reaction is called reduction.
Reducing agent
Reducing agents are the substances that reduce themselves and oxidize others.
Both these take place simultaneously. so we can say that where there is oxidation there is reduction.
So oxidation-reduction reactions can be defined as loss or gains of electrons is called oxidation-reduction reactions.
| Oxidation | Reduction |
| Addition of oxygen | Removal of oxygen |
| Removal of hydrogen | Addition of hydrogen |
| Loss of electron | Gain of electron |
Example
According to this concept :Oxidation is the loss of electrons by an atom or an ion.e.g:
Zn(s) → Zn2+ (aq) +2e–
Fe2+ (aq) → Fe3+ (aq) + e–
Reduction is the gain of electrons by an atom or ion.
e.g;
2H+ (aq) + 2e– → H2 (g)
Cl2 (g) + 2e– → 2Cl– (aq)
Overall redox reaction :
it is the sum of both reactions:
Zn(s) + 2H+ (aq) → Zn2+ (aq) + H2 (g)
Combustion Reactions:
- In this type of chemical reaction, substances react from the air.
- These are exothermic in nature produce heat.
- combustion reactions are also redox reactions.
- In combustion reactions, hydrocarbons are oxidized by molecular oxygen and produce carbon dioxide and water.
- The reaction of alkanes and alkenes.
The combustion reaction of alkanes:
Burning of alkane in the presence of oxygen is known as combustion.
Complete combustion of alkene produces CO2, H2O, and heat.
CH4(g) + 2O2(g) → CO2 (g) + 2 H2O (g) + 891kj/mol.
The combustion reaction of alkenes:
Alkenes burn in air with luminous flame and produce CO2, H2O vapors.
C2 H4 +3O2 → 2 CO2 + 2 H2O +Heat
Summary:
So that’s the complete overview of types of a chemical reactions. All the types are described completely with examples. There is a lot of reactions happening daily but the above-discussed types are some main types of chemical reactions.



Leave a Reply