
Which atom is the smallest in the periodic table? The most common wrong answer is hydrogen. Everyone thinks hydrogen is the smallest atom in terms of size in the whole periodic table because it has only one proton. Helium has two protons so shouldn’t it be double the size no that’s wrong. Helium is the smallest atom. How is it possible?
Which Atom Is The Smallest – Helium, Why?
Hydrogen has one proton in its nucleus and the most common isotope of it has no neutrons. Hydrogen has only one electron in its first shell. Helium on the other hand has two protons in its nucleus and also has two neutrons. Helium has two electrons that revolve around the nucleus in its first shell.
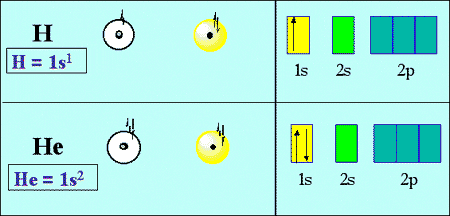
Hydrogen and helium both have electrons in the first shell. But here’s the question is:
Which of these Nuclie (Hydrogen and Helium) Attract the Electrons with Greater Force?
Remember that the electrons have minus charges on them and unlike charges always attract each other so minuses are attracted to pluses (charge on proton). The nucleus is dense and doesn’t take up much space, it’s just like a tiny little charge, chunk in the center of an atom. The nucleus of hydrogen has only a proton nut on the other hand the nucleus of helium has two protons. The protons of helium are doubly as attractive to electrons. The result is that the electrons of helium are more attracted by the nucleus and held closer to the nucleus as compared to the hydrogen atom. That’s why Helium is smaller in size than Hydrogen.
Why the Noble Gases Are the Smallest Atoms in their Entire Row?
The noble gases are the smallest atoms in their entire row for example rubidium and xenon both have electrons in their fifth shell but rubidium only has 37 protons in its nucleus and xenon has 54 protons. The nucleus of xenon is more attractive to electrons so xenon’s electrons are held slightly closer and the xenon atom is smaller than the rubidium atom.
Which Element has the Largest Atomic Radius?
Francium is the largest element in the periodic table.
- Element Symbol: Fr
- Atomic Number: 87
- Atomic Weight: 223
- Discovery: Perey, In 1939
- Electron: [Rn] 7s1
- Group: 1 (Alkali Metals)
- Period: 7
- Block: s-block
- Apperence: Silver metal (predicted)




Leave a Reply