
Xenon tetrafluoride is a chemical compound having the chemical formula XeF4. It is formed by the reaction of xenon with fluorine. Xenon tetrafluoride is a white-colored solid compound with a square planar shape. Xenon tetrafluoride is non polar and has zero dipole moment.

According to VSEPR theory in xenon tetrafluoride, the xenon center has two lone pairs. White crystalline colorless solid and sublimes at 115.7 ℃.
Xenon tetrafluoride is formed by the reaction of Xe with fluorine.
Xe + 2F2 → XeF4
This is an exothermic reaction releasing 251 KJ/mole energy.
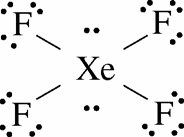
Lewis Structure of XeF4 :

Xenon tetrafluoride consist of Xenon noble gas. As we know that noble gases do not share electrons to any other atom. But xenon is different. The electronic configuration of xenon is ;
1s2 ,2s2, 2p6, 3s2, 3p6, 4s2, 3d10, 4p6, 5s2, 4d10, 5p6
5th energy level of the orbital consists of s,p, d, and f. Xenon has only completely filled s and p. The coming force from outside the electrons from p moves into the d orbital. And then these moving electrons participate in bond formation. So as for drawing Lewis’s structure, Xenon is a noble gas and is expected to have more than 8 electrons from outside.
For this problem.
We will use the number of valence electrons and valence shell electronic configuration of electrons of each atom to help in drawing the right Lewis structure.
By obtaining the following:
- valence electrons
- Electron configuration
Valence electrons of XeF4 :
This can be given out by sum up of valence electrons of each atom in the compound. First of all list down the atoms present and the total number of atoms in the compound. AS we know that in xenon tetrafluoride there is 1 xenon and four fluorine. And from the periodic table, we get the valence electrons of each atom.
| Atom | No.of atom | valence e– | sub-total |
| xenon | 1 | 8 | 8 |
| Fluorine | 4 | 7 | 28 |
So xenon has 8 electrons and fluorine has 7 electrons so the total number of electrons is 36 electrons.
Electron Configuration:
The electronic configuration of xenon tetrafluoride is;
1s2 ,2s2, 2p6, 3s2, 3p6, 4s2, 3d10, 4p6, 5s2, 4d10, 5p6
In xenon tetrafluoride, the xenon is a less electronegative atom so it is considered a central atom. First of all, we draw single bonds between the central atom and terminals fluorine atoms. Then we draw the remaining unpaired electrons of each atom according to their number of valence electrons. Now after this we have to check the stability of xenon tetrafluoride the fluorine must follow the octet rule. Fluorine must have 8 electrons. There is no lone pair.
Dipole moment in XeF4 :

By knowing the dipole moment helps to determine the position of atoms attached to the central atom is correct or not. For dipole moment between Xenon and Fluorine, it helps us pull xenon towards fluorine because fluorine is more electronegative in nature. Between the central atom and its lone pair direction of the dipole moment will favor pointing to the lone pair. The geometry is square planar.
Imagine a plane on the y-axis perpendicular to the plane on the x-axis. The lone pairs are present in the plane on the y-axis. Fluorine atoms are in the perpendicular plane on the x-axis. The lone pair pulls xenon atoms towards them at equal force. Fluorine atoms also pull the xenon atom towards them in opposite direction and with the same amount of force.
This results in zero dipole moment.
Hybridization:
Xenon tetrafluoride contains xenon as the central atom and it is the place where hybridization takes place. The valence shell of an atom contains 6 electrons in a 5p orbital. Whereas there are 2 electrons in the 5s orbital. There are no electrons present in f and d orbitals.
AS we discussed earlier that in xenon tetrafluoride the two electrons in the 5p orbital becomes excited and move to 5d orbitals in order to fill empty or vacant space. And after this, the remaining 4 unpaired electrons form sp3d2 hybridization in which there are two unpaired electrons in the 5p orbital and 2 in the 5d orbital. A sigma bond is formed so xenon tetrafluoride has sp3d2 hybridization.
Points to be noted:
- In xenon tetrafluoride, the central atom constitutes 6 electron pairs in which 2 are lone pair electrons.
- So hybridization is because 2 electrons shifted from p to d which results in the formation of a sigma bond with fluorine.
Molecular geometry:
As xenon tetrafluoride has two lone pair electrons. According to VSEPR theory, the net electronic repulsion will be decreased. And in this way, they get a stable state. For this, the lone pairs lie in a perpendicular plane in an octahedral arrangement that is opposite from each other. And the bond angle is 90 degrees. So that’s why xenon tetrafluoride has square planar geometry.

Polarity of XeF4 :
Xenon tetrafluoride is non polar molecule. As we discussed that there is square planar geometry and the net dipole moment is zero across the molecule. So there is uniform charge distribution and no polarization across xenon tetrafluoride.
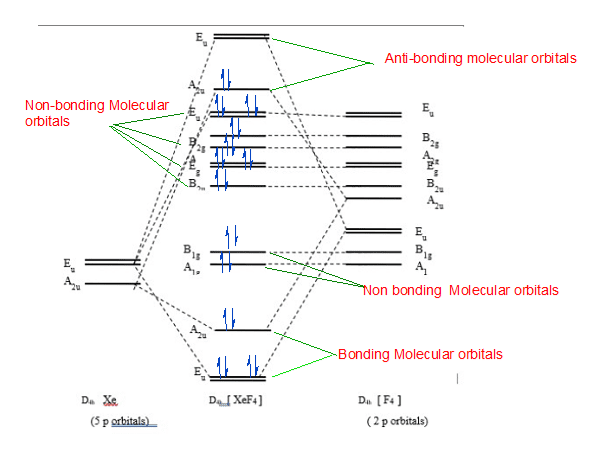
Mo diagram of XeF4 :
First of all the MO diagram of any molecule explain chemical bonds in a given molecule with the help of molecular orbital theory. MO diagram helps to determine the existence of molecules. This determines the strength of bonds and electronic transitions.
In xenon tetrafluoride, there is square planar geometry. And there is a 1.95A° distance between xenon and fluorine. It is explained through valence bond theory that there are 2 nonbonding electrons that jump from p orbital to d orbital in order to fill vacant space. 4 half-filled orbitals form covalent bonds with half-filled 2p orbitals of the fluorine atom in the structure.
Xenon tetrafluoride there is 2 lone pairs and four bond pairs.
Uses of XeF4:
It is used in finding traces of silicon metal that is contaminated with rubber.



Leave a Reply