
Here we draw the N2 lewis structure, molecular geometry, and hybridization of nitrogen molecule. To draw the lewis structure of N2, first to find out the valance electron of nitrogen. The atomic number of nitrogen is 7 having an electronic configuration of 1s2, 2s2, 2p3. How do we find the valance electron of an atom? Actually, the valance electron is the electrons of the outermost shell. That’s why we have to find out the highest value of the principal quantum number. In N2, the highest value of the principal quantum number is 2. As (2s2, 2p3) there are two electrons in the s-subshell and three electrons in the p-subshell, so the nitrogen atom has a total of 5 valance electrons.
There is another way to find the valance electron for nitrogen. Actually, the group number tells us the valance electrons for the elements. Nitrogen belongs to the 5A group in the periodic table. So it has 5 valance electrons. The total number of valance electrons of N2, the one nitrogen atom have 5 valance electron so two nitrogen atom will have 10 valance electrons totally.
How do you draw the N2 Lewis Structure?
After determining the total number of valance electrons of the N2 molecule, we have to connect two nitrogen atoms by a single bond. One single bond contains two valance electrons. By connecting two nitrogen atoms via a single bond, we used just two valance electrons so we have 8 left. These 8 electrons will be used to satisfy the octet of nitrogen atoms.

As we know that every atom wants to complete its octet and gets stability. In the above structure, one of the nitrogen has 8 electrons (1 bond pair, 3 lone pairs) and completes its octet but another atom has 4 electrons (one bond pair, 1 lone pair) and an incomplete octet. Remember that the lone pair electrons are used for both lone pairs as well as a double bond or triple bond. So, the nitrogen atom shares its lone pair electrons with another nitrogen atom to satisfy the octet.
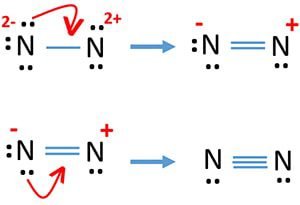
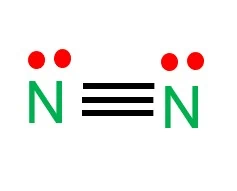
The two nitrogen atoms contain 3 bond pair electrons (triple bond) and two lone pair electrons. There is another method to draw the N2 lewis structure by using the formula.
How do you Calculate Lewis Dot Structure?
First of all, we calculate Q (total electrons), equal to the valence electron of all-atom plus the number of negative charges minus the number of positive charges.
Q = Valance electron of all-atom + no of -ve charge – no of +ve charge
Then we find the bond pair electrons equal to the number of bonds multiplied by two.
B.P e– = 2 × no of bonds
Then we find lone pair electrons by subtracting the bond pair electrons from total electrons Q.
L.P e– = Q – B.P e–
Lone pair electrons are used for both lone pairs as well as a double bond or triple bond.
How do you Calculate the N2 Lewis Structure?
Now we learn how to draw a N2 lewis structure, first, by using the formula of Q, we find the total number of electrons.
Q = Valance electron of all-atom + no of -ve charge – no of +ve charge
Q = 10 + 0 – 0
Q = 10
B.P e– = 2 × no of bonds
B.P e– = 2 × 1
B.P e– = 2 e–
L.P e– = Q – B.P e–
L.P e– = 10 – 2
L.P = 8
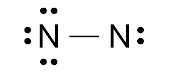
These lone pair electrons are also used as a double bond or triple bond by sharing the lone pair with another atom to complete the octet.
Hybridization of Nitrogen
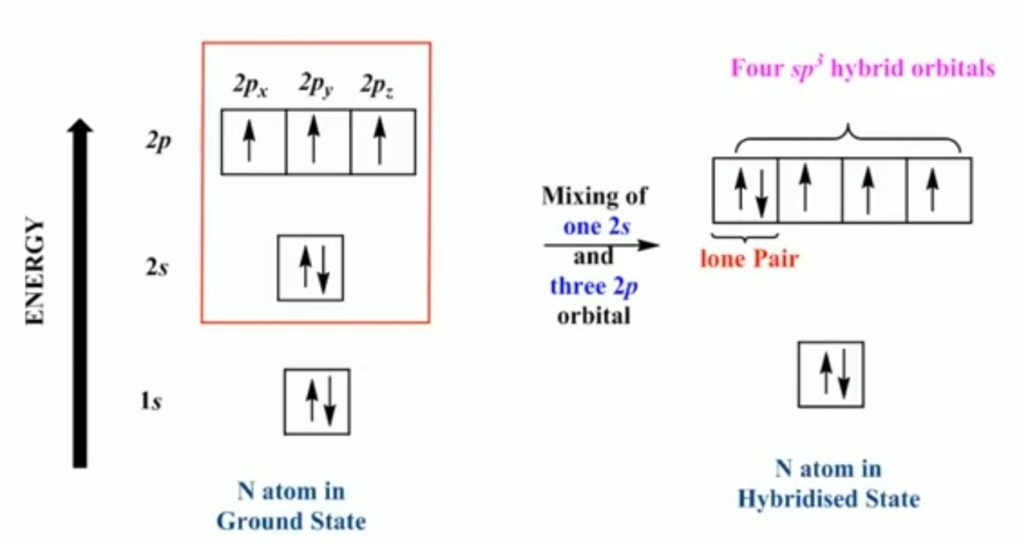
Hybridization is the intermixing of atomic orbitals having different shapes and energy and forming new hybrid orbitals of the same shapes and the same energy and suitable for the pairing of electrons to form chemical bonds in valance bond theory.
Sp3 Hybridization of Nitrogen
The atomic number of nitrogen is 7 having a ground state electronic configuration is 1s2, 2s2, 2p3. In sp3 hybridization, one of the 2s and three 2p atomic orbitals intermix to form equivalent sp3 hybrid orbitals. The 2s orbital electrons contain lone pair of electrons.
Example – Ammonia
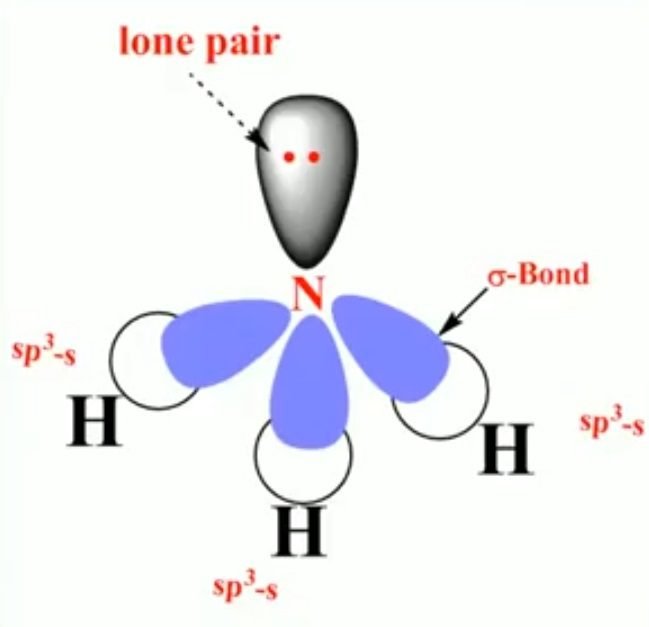
- The three sp3 hybrid orbitals of nitrogen are overlapped with the half-filled s-orbital of 3 hydrogen atoms.
- There remains a full sp3 hybrid orbital.
- The geometry of ammonia is trigonal pyramidal or distorted tetrahedral due to the presence of lone pair.
- In ammonia, there is a lone pair and bond pair repulsion.
- H-N-H bond angle in ammonia 107°. It is close to the tetrahedral angle which is 109.5°. But in ammonia, the bond angle is 107° because the nonbonding pair occupies more space than the bonding pairs.
- Hybridization of nitrogen = sp3
- Bond angle = 107°
- Geometry = pyramidal
Sp2 Hybridization of Nitrogen
In sp2 hybridization, one of the 2s and two 2p atomic orbitals intermix to form equivalent sp2 hybrid orbitals. The 2s orbital electrons contain lone pair of electrons.
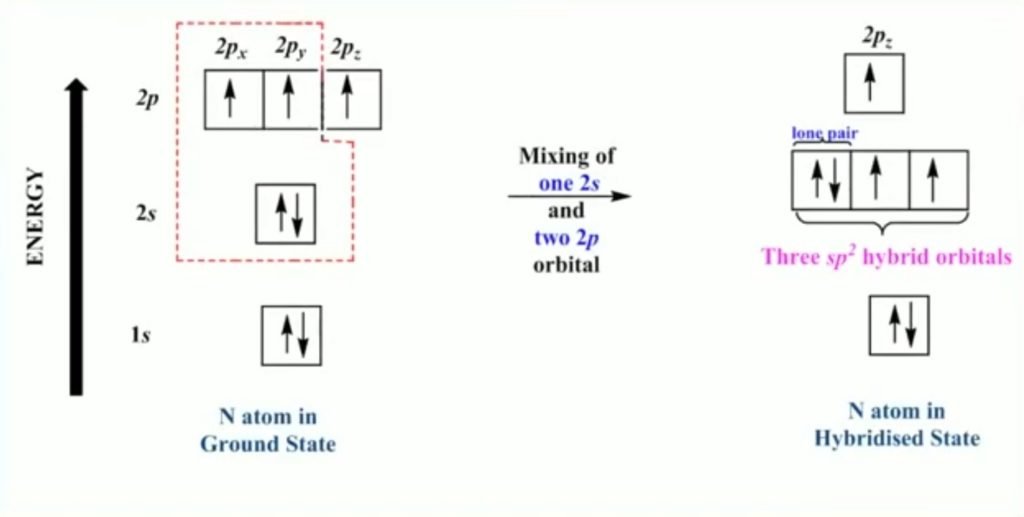
Example – Imine
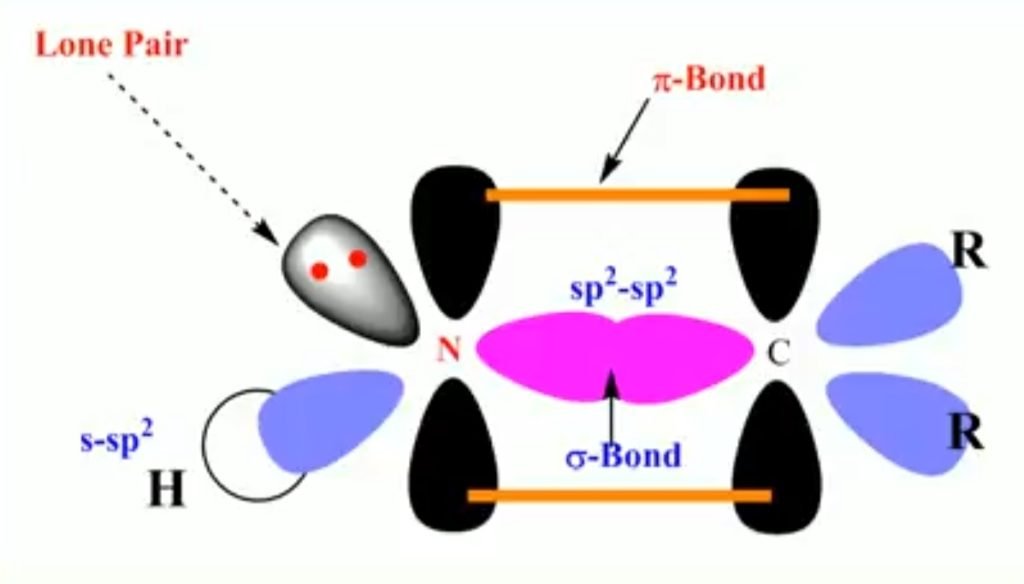
- Hybridization of nitrogen = sp2
- Bond angle = 120°
- Geometry = Trigonal planar
Sp Hybridization of Nitrogen
In sp hybridization, one of the 2s and one 2p atomic orbitals intermix to form equivalent sp hybrid orbitals. The 2s orbital electrons contain lone pair of electrons.
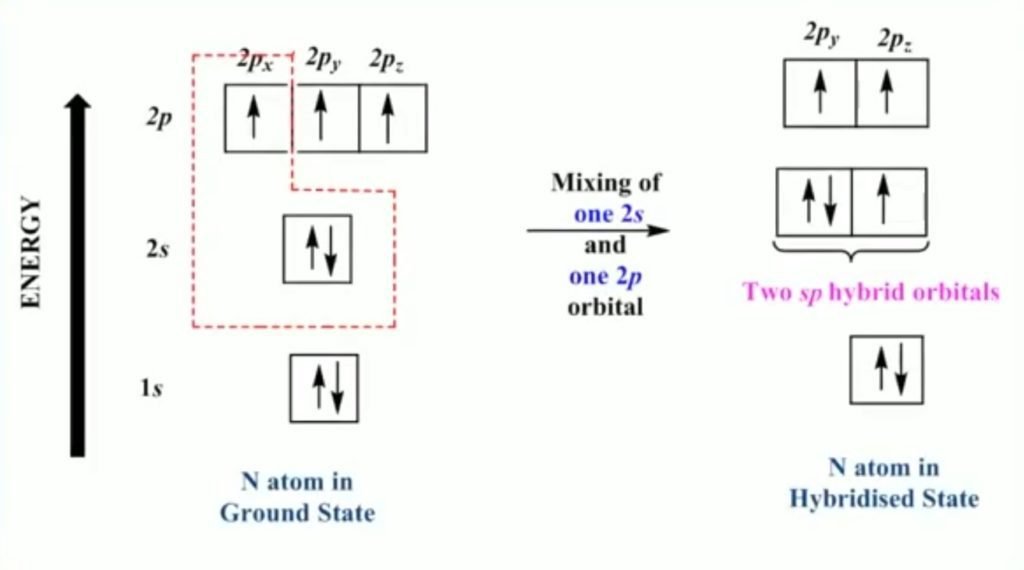
Example – Nitrile
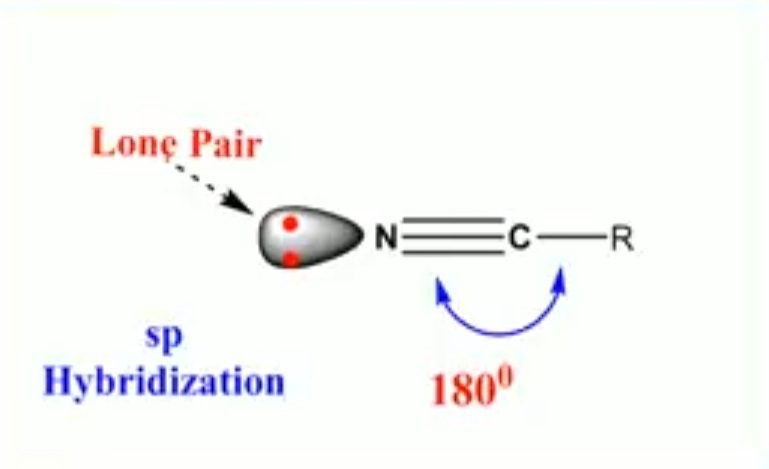
- Hybridization of nitrogen = sp
- Bond angle = 180°
- Geometry = Linear
What is the MO Diagram for N2?
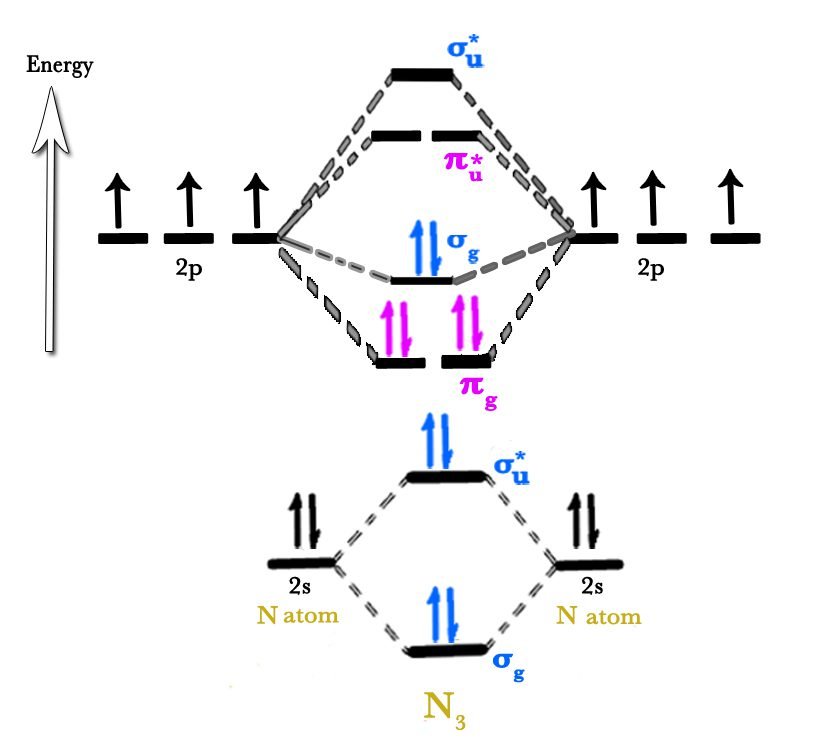
Now we calculate the bond order of nitrogen molecule, N2.
Bond Order of N2
Bond order is the number of covalent bonds present in a molecule.
Formula of Bond Order
Bond order = 1/2 (Number of electrons in BMO – Number of electrons in ABMO)
Bond order = 1/2 (8 – 2)
Bond order = 3
- All electrons are paired, so N2 is diamagnetic in nature.



Leave a Reply